Enosteoides habibi, Anker & Benzoni, 2023
|
publication ID |
https://doi.org/10.11646/zootaxa.5278.1.3 |
|
publication LSID |
lsid:zoobank.org:pub:7B68BAD4-FA98-45B3-8267-F2652F03CDE9 |
|
DOI |
https://doi.org/10.5281/zenodo.7900230 |
|
persistent identifier |
https://treatment.plazi.org/id/15405E79-EB05-FF9C-FF7E-C00AC3B2CC12 |
|
treatment provided by |
Plazi |
|
scientific name |
Enosteoides habibi |
| status |
sp. nov. |
Enosteoides habibi sp. nov.
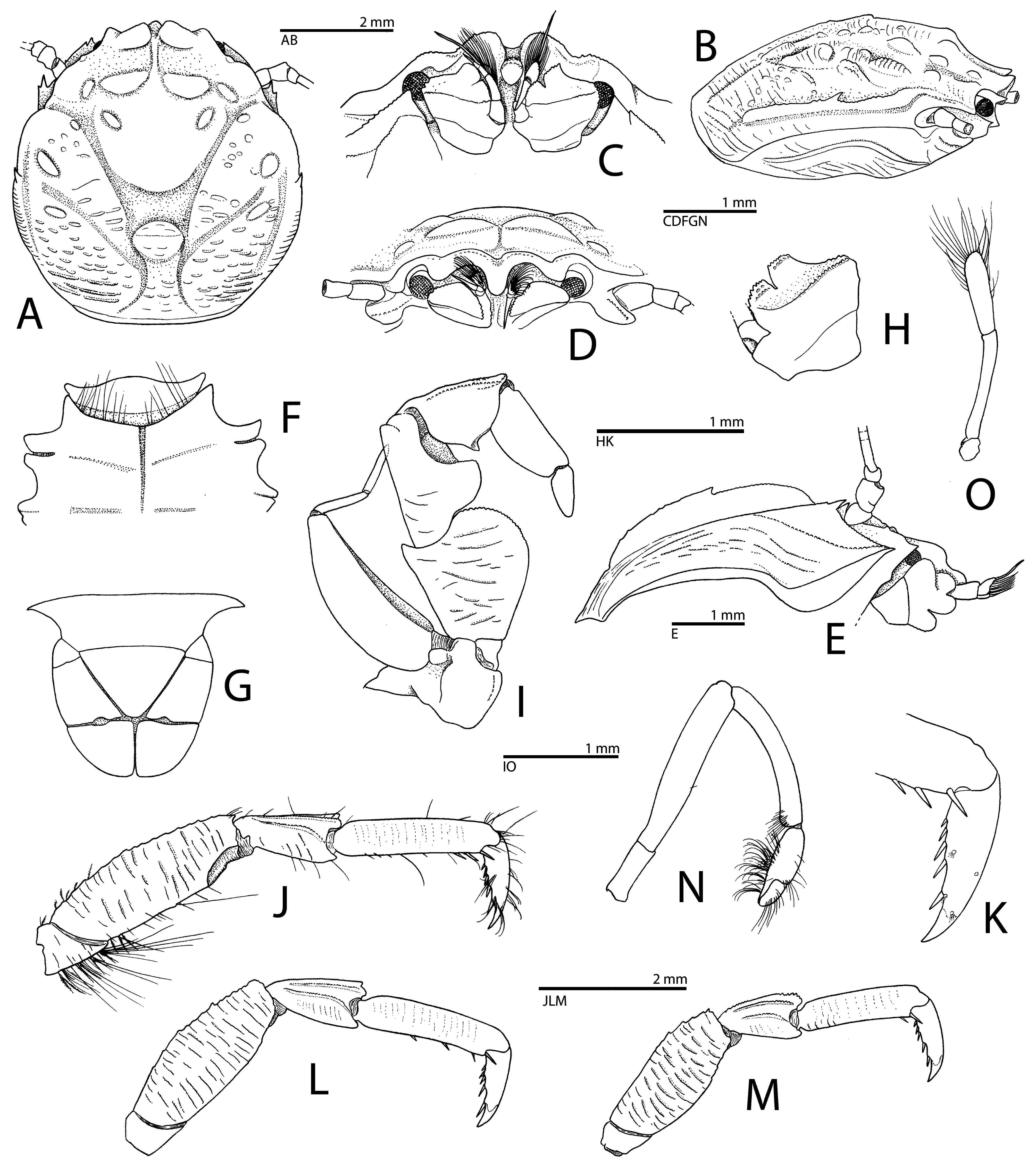
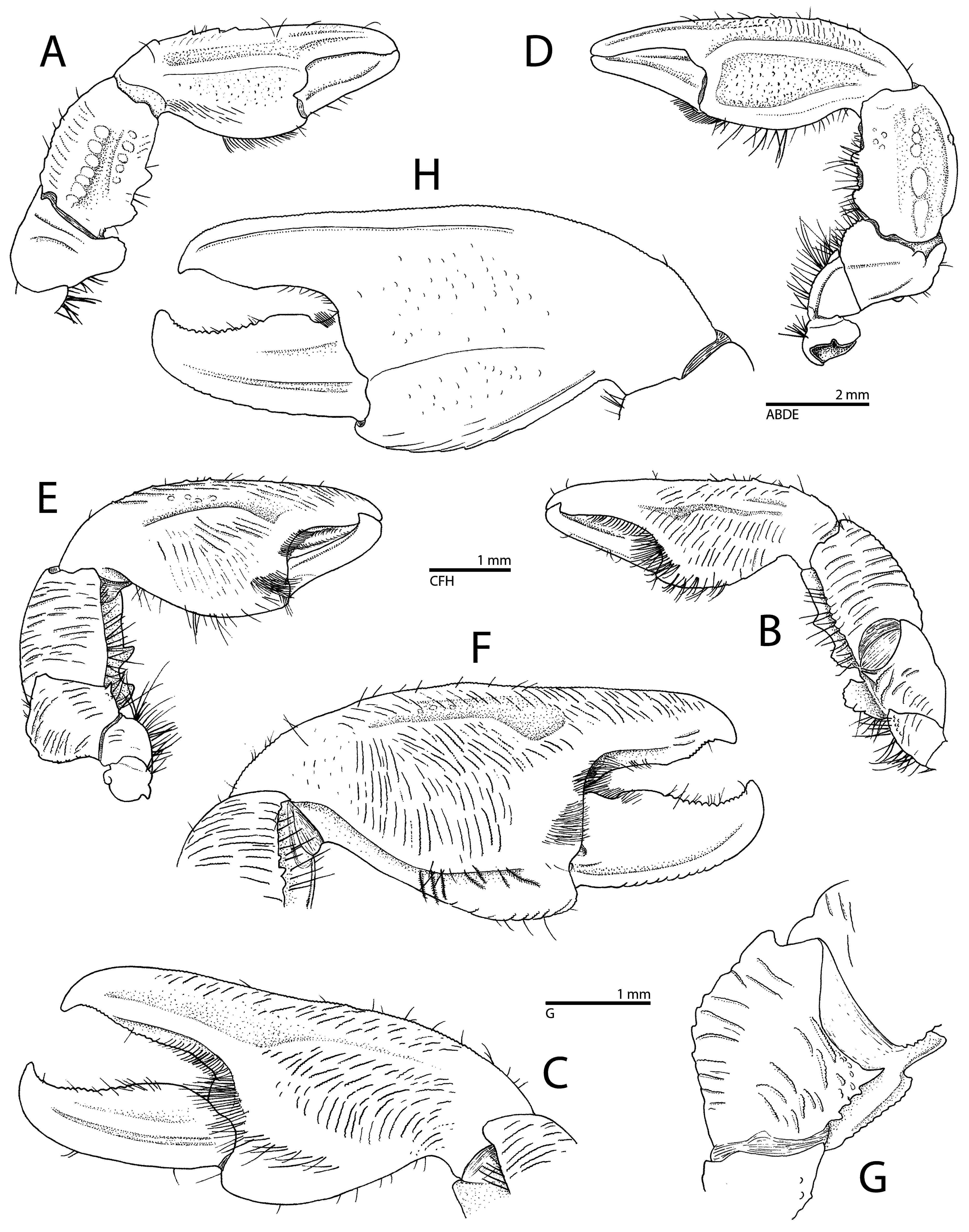
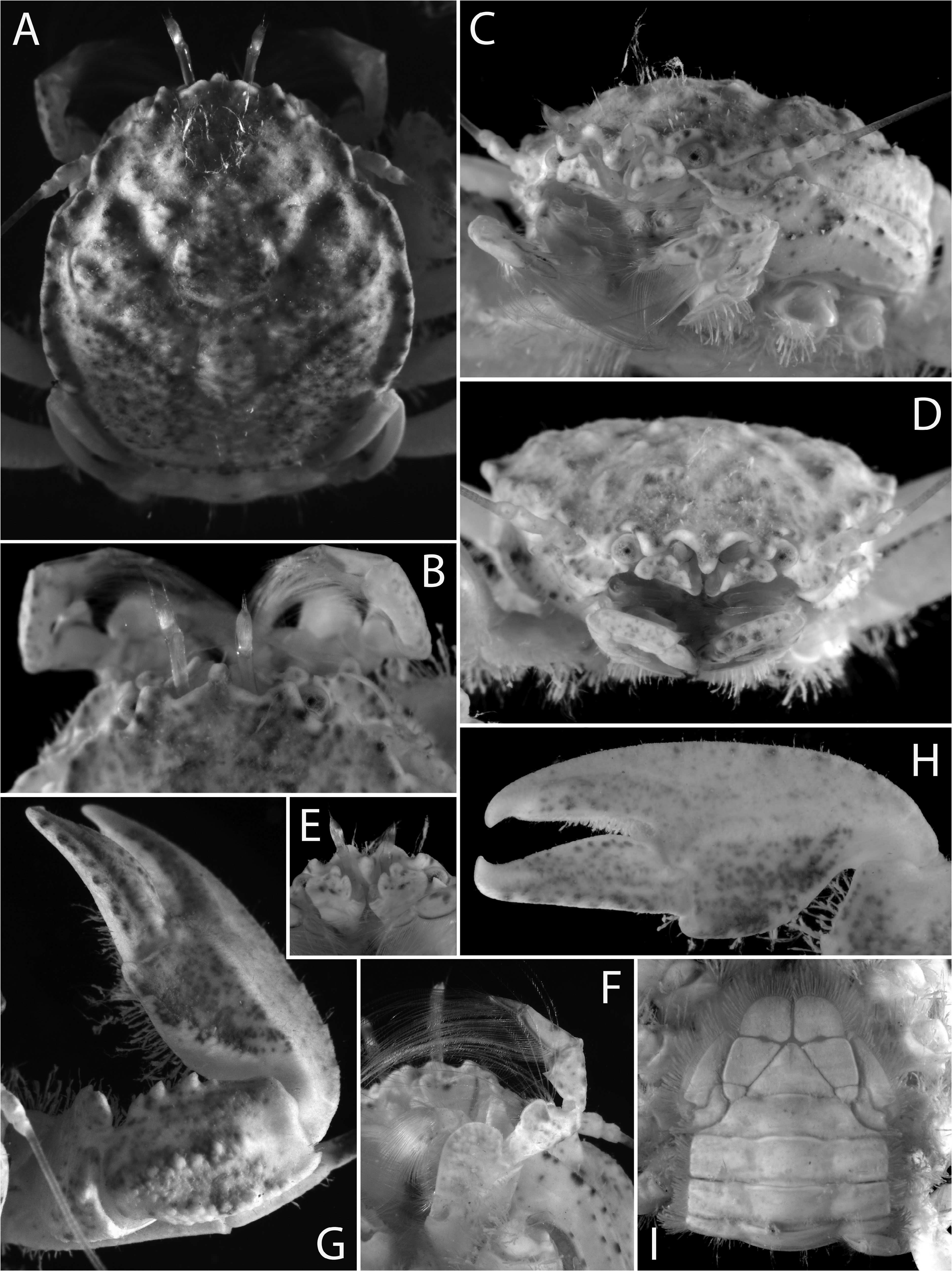
( Figs. 6–11 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 View FIGURE 10 View FIGURE 11 )
Type material. Holotype: male, cl 8.1 mm, cw 7.5 mm, FLMNH UF 71320 , Saudi Arabia, Makkah Province, Thuwal, King Abdullah University of Science and Technology ( KAUST), near King Abdullah Monument , 22°20’26.2”N 39°05’15.1”E, shallow sand-rubble flat between small mangrove and deeper channel, in burrow of Alpheus sp. , suction (yabby) pump, depth: 0.3 m, leg. A. Anker & A. Assayie, 29.12.2022 [fcn AA-22-436] GoogleMaps . Paratypes (not deposited, specimens lost, see text): 1 male, cl 5.5 mm, cw 5.2 mm, 1 female, cl 3.8 mm, cw 3.6 mm, Saudi Arabia, Makkah Province, Thuwal, King Abdullah University of Science and Technology ( KAUST), near King Abdullah Monument , 22°20’26.2”N 39°05’15.1”E, shallow sand-rubble flat between small mangrove and deeper channel, in burrow of Alpheus sp. , suction (yabby) pump, depth: 0.5 m, leg. A. Anker, 24.09.2022 [fcn AA-22-224] GoogleMaps ; 1 male, cl 5.8 mm, cw 5.5 mm, same collection data as for previous paratypes [fcn AA-22-222] GoogleMaps ; 1 female, cl 6.2 mm, cw 6.3 mm, same collection data as for previous paratypes [fcn AA-22-223] GoogleMaps .
Description. Carapace ( Figs. 6A, B, D View FIGURE 6 , 8A View FIGURE 8 ) subcircular, approximately as long as wide to 1.1 times longer than wide; dorsal surface somewhat convex from side to side, uneven, patchily covered with numerous short rows of granules and rugosities; postrostral area with groups of long setae; rest of carapace with scattered short setae; regions well demarcated; protogastric area with one pair of large, blunt, submedian lobes, one pair of smaller, more lateral swellings and one pair of small, more posterior swellings; anterior gastric region slightly elevated, with low, rounded median elevation; posterior gastric region with short granular ridges; anterior and median-posterior branchial regions separated by shallow sulcus, with numerous short and long granular ridges; anterior branchial regions moderately elevated, each with low bump laterally, smaller tubercles anteriorly, and some elevated granular ridges more posteromesially; median branchial regions each with one small swelling; median and posterior branchial regions with numerous short and moderately long granular ridges; cervical grooves moderately deep; lateral branchial and posterior cardiac regions with short, more or less elevated, granular ridges.
Rostrum ( Figs. 6A, C, D View FIGURE 6 , 8B–D View FIGURE 8 ) relatively broad, about 0.4 carapace width (measured between inner orbital angles), trilobate, slightly descendent; median lobe longer but narrower than lateral lobes, deeply grooved on dorsal mid-line; anterior median apex only partly visible in dorsal view, strongly bent ventrally, terminating bluntly; lateral lobes subtriangular or rounded, obtuse. Orbit shallow; supraorbital margin weakly concave; each outer orbital angle with small obtuse protuberance. Hepatic margin undulated, with some rugosities. Branchial margin ( Figs. 6A, B View FIGURE 6 , 8A View FIGURE 8 ) broadly convex, somewhat irregular, elevated from 0.3 to 0.7 of carapace length, forming distinct marginal crest, sometimes armed with small subacute tooth or obtuse protuberance at about mid-length of carapace; posterior branchial margin with short, blunt, transverse, granular ridges. Pterygostomial flap ( Figs. 6E View FIGURE 6 , 8C View FIGURE 8 ) with longitudinal or obliquely running, minutely tuberculate ridges, anteriorly terminating in acute tooth; dorsal margin with subacute protuberance below base of first article of antennal peduncle.
Third thoracic sternite ( Fig. 6F View FIGURE 6 ) with ventral surface slightly concave medially; median lobe weakly produced anteriorly; anteromedian margin broadly rounded; lateral lobes narrow, each with bluntly rounded apex. Fourth thoracic sternite ( Fig. 6F View FIGURE 6 ) distinctly separated from third sternite; anterior margin evenly convex, furnished with row of long submarginal setae; ventral surface slightly convex medially, apparently without marked transverse ridges; posterior lateral lobes strongly produced, blunt or subacute distally. Remaining thoracic sternites concave on ventral surface, with distinct longitudinal groove on midline.
Telson ( Figs. 6G View FIGURE 6 , 8I View FIGURE 8 ) as long as wide to slightly wider than long, composed of seven plates; each posterior plate subtriangular, usually with small anterior convexity.
Ocular peduncles ( Figs. 6A, B, D View FIGURE 6 , 8B, C View FIGURE 8 ) short, with relatively small, normally pigmented cornea.
Antennular peduncle ( Figs. 6C, D, H View FIGURE 6 , 8B, E View FIGURE 8 ) with basal (= first) article of peduncle approximately square-shaped (exclusive of anteromesial lobes); anterior surface with scattered small tubercles, separated from ventral surface by transverse ridge of small tubercles; anteromesial surface with two lobes subequal in size and shape, each covered by rows of small tubercles, including on distal margin. Antennal peduncle ( Figs. 6D, E View FIGURE 6 , 8C View FIGURE 8 ) short, slender; first article largest; anterior margin subacutely produced towards eye and basal article of antennular peduncle in lateral view, and largely in contact with ventral part of orbital margin; lateral surface with wellmarked, longitudinal, granular ridge along ventral margin; second article somewhat longer than third article, with some granules; third article subrectangular, somewhat wider distally, with row of granules; fourth article shortest, rounded.
Third maxilliped ( Figs. 6I View FIGURE 6 , 8F View FIGURE 8 ) with coxa bearing rounded lobe distally on ventral side, submedian surface with row of small granules; basis small, not fused with ischium, subtriangular; endopod broad, robust; ischium very broad, with lateral surface covered with numerous transverse granular ridges, distodorsal margin bluntly projecting, distoventral margin strongly convex; merus with broad, subrectangular lobe on ventral margin, lateral surface with short granular ridges, distolateral portion with short ridge at constriction; carpus with acutely triangular projection on ventral margin, lateral surface with few longitudinal ridges along dorsal margin, one running almost entire length of article; propodus slender, tapering distally, smooth; dactylus about half-length of propodus, tapering distally, smooth; exopod with proximal article short, rounded; distal article inflated proximally, narrowing distally, reaching half-length of merus, almost smooth on lateral surface; flagellum well developed, slender, with two almost equal subdivisions.
Chelipeds (first pereiopods) ( Figs. 7A–H View FIGURE 7 , 8G, H View FIGURE 8 , 9A View FIGURE 9 , 10 View FIGURE 10 , 11 View FIGURE 11 ) slightly unequal in size but similar in shape and sculpture, flattened, sparsely setose on surfaces and margins, more densely setose on ventral surface. Ischium with short rows of minute granules or fields of granules on ventral surface. Merus somewhat cup-shaped in dorsal view, with blunt transverse crest submedially on dorsal surface; anterodorsal margin with large, protruding, marginally rugose and tuberculate lobe; posteroventral margin distally with two blunt lobes; ventral surface with several short and long granular ridges; anteroventral margin distally with strong subacute tooth. Carpus robust, about 1.7 times as long as wide (exclusive of teeth on anterodorsal margin); anterodorsal margin with two large blunt teeth on proximal half, proximal tooth subdivided into two or more smaller lobes distally; dorsal surface rough, with small granules (isolated or arranged in rows), larger bump-like tubercles, blunt longitudinal crest marked by row of larger rounded protuberances along midline, and series of transverse granular ridges near posterior margin; posterior margin crenulated, rugose, ending in subacute or blunt projection distally; ventral surface with numerous, long, transversal, granular ridges. Chela moderately broad, 1.9–2.1 times as long as carpus, about 2.1– 2.3 times as long as broad. Palm with anterior margin almost straight, thin, slightly crenulated, and bearing one row of up to five spaced, low, distally blunt teeth; dorsal surface flattened, with weak ridge on midline, covered with numerous small granules and short granular ridges; ventral surface with shallow longitudinal depression in its middle, most of ventral surface ornamented with numerous short and long, transverse, granular ridges; posterior margin crenulated, slightly elevated dorsally, forming blunt crest. Pollex (= fixed finger) with anterior margin straight, slightly crenulated, without marked teeth; dorsal surface with low, blunt, longitudinal crest along midline, extending on about half of palm; ventral surface with some granular ridges, mainly proximally and anteriorly; tip short, blunt, not markedly curved; cutting edge unarmed or with row of small, obtuse teeth subdistally, concave on proximal half. Dactylus about 0.5–0.6 length of chela, somewhat twisted, i.e., opening at slightly oblique angle; posterior margin slightly elevated, forming distinct, blunt, longitudinal crest, with row of short granular ridges resulting in crenulation; dorsal surface bluntly elevated on midline; tip strongly curved, blunt, crossing with tip of pollex; ventral surface with shallow groove along posterior margin; cutting edge with small, obtuse teeth on distal half and low, broadly rounded tooth on proximal half fitting into shallow concavity of pollex, or with obtuse teeth along entire margin (e.g., in holotype). Distoventral surface of palm near pollex and dactylus with two small, dense fields of setae, sometimes merged into single, densely pubescent area (in holotype); cutting regions of fingers with rows of short, more or less dense, fine setae.
Ambulatory legs (= second to fourth pereiopods) ( Figs. 6J–M View FIGURE 6 , 9A View FIGURE 9 , 10 View FIGURE 10 , 11 View FIGURE 11 ) moderately stout, somewhat compressed laterally, generally similar in shape and armature, decreasing in size posteriorly. Second pereiopod ( Fig. 6J, K View FIGURE 6 ) longest of ambulatory legs. Ischium bearing some plumose setae on dorsal and ventral margins, with relatively dense row of long stiff setae on ventral margin. Merus about 2.8 times as long as maximal width, with crenulated dorsal and almost smooth ventral margins; distodorsal margin with blunt tooth; lateral surface with numerous transverse granular ridges; mesial surface with similar granular ridges as on lateral surface. Carpus slightly less than 0.6 length of merus; lateral surface with two granular crests, one on dorsolateral surface and one on lateral midline, and several short, transverse, granular ridges; mesial surface with similar granular ridges as on lateral surface; distodorsal margin with three blunt lobes. Propodus moderately long, about 4.3 times as long as wide, with transverse rows of granules; ventral margin with two or three robust corneous spines on distal third of margin; distoventral margin with pair of stout spines on each side of dactylo-propodal articulation. Dactylus about 0.6 length of propodus, robust, slightly curved, terminating in stout, somewhat curved unguis; ventral (flexor) margin with five corneous spinules increasing in size distally.
Third pereiopod ( Fig. 6L View FIGURE 6 ) generally similar to second pereiopod in shape and proportions. Merus with distinctly crenulated dorsal margin. Carpus similar to that of second pereiopod, slightly shorter. Propodus about 4.5 times as long as wide; ventral margin with three robust corneous spines along distal 0.6 of length; distoventral margin with pair of stout spines on each side of dactyl-propodal articulation. Dactylus about half-length of propodus, with five corneous spinules on ventral margin.
Fourth pereiopod ( Fig. 6M View FIGURE 6 ) slightly smaller than third pereiopod and much smaller than second pereiopod. Merus with bluntly crenulated dorsal margin; carpus with two granular ridges as on carpi of second and third pereiopod, and crenulated dorsal margin, crenulation stronger on distal third; propodus about 4.8 times as long as wide; ventral margin subdistally with one small corneous spine; distoventral margin with pair of stout spines on each side of dactylo-propodal articulation; dactylus about 0.6 length of propodus, with five corneous spinules on ventral margin.
Fifth pereiopod ( Fig. 6N View FIGURE 6 ) short, slender, chelate. Merus about 3.2 times as long as ischium and 1.5 times as long as carpus. Carpus gently arched mesially, with row of simple setae on distoventral margin. Chela almost half-length of carpus; fingers about half-length of palm; palm and pollex with dense tufts of long, distally curved setae on ventrolateral surface; distal portion of palm and dorsal margin of dactylus with simple setae.
Males with one pair of simple gonopods ( Fig. 6O View FIGURE 6 ) on second pleomere; endopod long, slender, distally rounded, with marginal setae; exopod reduced, not visible in posterior view.
Colour pattern. Carapace tan-yellow to pale greyish brown, with some darker red-brown or brown-grey markings (blotches, spots, etc.), especially on tips of tubercles and along crests of lateral margin. Chelipeds tan to pale brown with some pale pink-orange marbling and darker red-brown or grey-brown markings on palm and fingers. Ambulatory legs (second to fourth pereiopods) dorsally pale yellow with conspicuous red-purple patches or broad bands, disposed as following: small patch on merus of second pereiopod (sometimes absent); medium to large patch or band on proximal half of carpus of second and third pereiopods (sometimes absent); larger patch or band near middle of propodus of each second, third and fourth pereiopods (always present). Distal article of antennular peduncles, some parts of third maxillipeds (especially distal articles) and chela of fifth pereiopod with bluish tinge. Ventral side of second to fourth pereiopods, sternum and pleon tan yellowish, without particular pattern or markings ( Figs. 9–11 View FIGURE 9 View FIGURE 10 View FIGURE 11 ).
Etymology. The name of the new species is the Arabic word habibi meaning dear or beloved, but also “buddy” between good friends, alluding to the porcelain crab being a friend of snapping shrimps, gobies and other associates of the burrow; used as a noun in apposition.
Distribution. Presently known only from the type locality on the Saudi Arabian coast of the Red Sea.
Ecology. All specimens were collected from burrows of Alpheus spp. , however, each time without a host. The general habitat at the type locality of E. habibi sp. nov. was described under P. jamila sp. nov. (see above).
Remarks. With the above description of E. habibi sp. nov., the exclusively Indo-West Pacific genus Enosteoides now contains nine species, the others being the type species E. ornatus ( Stimpson, 1858) , E. melissa ( Miyake, 1942) , E. palauensis ( Nakasone & Miyake, 1968) , all three widely distributed in the Indo-West Pacific; E. lobatus Osawa, 2009 , E. lapis Osawa, Nakajima & Hamamoto, 2022 , both known only from southern Japan; E. philippinensis Dolorosa & Werding, 2014 and E. turkayi Osawa, 2016 , both presently known with certainty only from the Philippines, and E. spinosus Hiller & Werding, 2022 , known only from Australia ( Miyake 1942; Nakasone & Miyake 1968; Osawa 2009, 2016; Osawa & Chan 2010; Dolorosa & Werding 2014; Osawa et al. 2022; Hiller & Werding 2022). The photographic record of E. lobatus from Sulawesi in Werding et al. (2016) probably refers to E. turkayi (cf. Osawa 2016; Osawa et al. 2022). Prior to this study, only three species of Enosteoides , viz. E. ornatus , E. melissa and E. spinosus , were known from the Indian Ocean ( Osawa 2009, 2016; Osawa & Chan 2010; Hiller & Werding 2022). The new species from the Red Sea plainly belongs to Enosteoides , as defined by Osawa (2009) and Osawa & Chan (2010), and is morphologically closest to E. melissa , E. philippinensis and E. turkayi (see below).
Enosteoides habibi sp. nov. can be separated from E. melissa , as figured by Miyake (1942) and Nakasone & Miyake (1968), by the presence of only one tooth (sometimes blunt or indistinct) on the lateral margin of the carapace ( vs. two or three teeth in E. melissa ); the third maxilliped carpus projecting ventrally as a subacute tooth ( vs. bluntly triangular in E. melissa ); and the absence of a dense field of setae on the extensor surface of the cheliped chelae ( vs. its presence in E. melissa ) (cf. Figs. 6A, I View FIGURE 6 , 7A, D View FIGURE 7 , 8A, F–H View FIGURE 8 , 9A View FIGURE 9 , 10 View FIGURE 10 , 11 View FIGURE 11 ; Miyake 1942: figs. 25, 26c; Nakasone & Miyake 1968: fig. 2A, B, E). The identity of specimens designated as “cotypes” of E. melissa by Miyake (1942) presently remains uncertain. Nakasone & Miyake (1968) did not consider these specimens, two ovigerous females and one juvenile, to be conspecific with the holotype of E. melissa , likely due to some marked discrepancies in the armature of the second pereiopod and in the shape / armature of the basal article of the antennular peduncle and antennal peduncle (cf. Miyake 1942: figs. 25, 26a, b, 27a, b, d).
The colour pattern of E. melissa was illustrated by Osawa (2009: fig. 4C) and Osawa (2016: fig. 1A) and is generally similar to that of E. habibi sp. nov. ( Figs. 9–11 View FIGURE 9 View FIGURE 10 View FIGURE 11 ), especially in the dark red-brown banding on the ambulatory legs. Interestingly, the specimens from the type locality in Micronesia (Babeldáob Island) and southern Japan (Iriomote Island, Ryukyu Islands) reported by Miyake (1942) and Osawa (2009), respectively, were collected from the underside of intertidal rocks, whereas the specimens from a different Japanese locality ( Okinawa Island, Ryukyu Islands) in Osawa (2016) were collected with a suction pump from burrows of Alpheus rapax . Similarly, a specimen of E. melissa collected by one of us (AA) in Pouébo, New Caledonia ( Fig. 12 View FIGURE 12 ), was also found in a burrow of A. rapax in very shallow water (depth: 0.5–1 m).
Osawa (2007) reported two male specimens identified as E. melissa from a depth of 38 m off New Caledonia and 20–30 m off Loyalty Islands, respectively. As noted by the author, these specimens presented several important differences from the type specimen, including a comparatively wider carapace ( Osawa 2007: fig. 3A); the noticeably stouter first ambulatory legs ( idem.: fig. 3C); and, in the New Caledonian specimen only, the absence of a fringe of plumose setae and a row of small sharp teeth and denticles on the extensor surface of the chelae ( idem.: Fig. 3B View FIGURE 3 ). Importantly, two of these features (slightly wider carapace and absence of setal fringe on the chelae) are also observed in E. habibi sp. nov. from the Red Sea. Whatever the identity of Osawa’s (2007) specimens of E. melissa might be, they appear to be subtly different from the Red Sea species in the dorsal ornamentation of the carapace and the relative width of the merus of the second pereiopod (cf. Fig. 6A, J View FIGURE 6 ; Osawa 2007: 3 A, C). On the other hand, the afore-mentioned New Caledonian specimen ( Fig. 12 View FIGURE 12 ) is generally similar to the specimen from Okinawa illustrated by Osawa (2016) and to the material of E. melissa reported by Miyake (1942) and Nakasone & Miyake (1968). For the time being, the New Caledonian and Loyalty Islands material from below 20 m reported by Osawa (2007) is tentatively considered as not conspecific with the present new species, despite many similarities. However, a molecular analysis of all the material currently assigned to E. melissa from various localities of the Indo-West Pacific and from different habitats / depths is highly desirable.
Enosteoides habibi sp. nov. can be separated from E. philippinensis by essentially the same characters as from E. melissa , i.e., the presence of at most one tooth on the lateral margin of the carapace ( vs. at least three teeth in E. philippinensis ), and the extensor surface of the cheliped chela without dense setal cover ( vs. with dense field of setae in E. philippinensis ) (cf. Figs. 6A View FIGURE 6 , 8A, G, H View FIGURE 8 ; Dolorosa & Werding 2014: figs. 1, 2A, H). In addition, in E. habibi sp. nov., the posterior margin of the cheliped carpus does not bear a stout sharp spine near its mid-length, as in E. philippinensis (cf. Figs. 6A, D View FIGURE 6 , 8G, H View FIGURE 8 ; Dolorosa & Werding 2014: figs. 1, 2H). Furthermore, E. habibi sp. nov. and E. philippinensis can be distinguished by the colouration of the ambulatory legs (second to fourth pereiopods), which have red-purple markings in the former species and are plainly coloured in the latter species (cf. Figs. 9–11 View FIGURE 9 View FIGURE 10 View FIGURE 11 ; Dolorosa & Werding 2014: fig. 4). Finally, the Red Sea species is ecologically different from E. philippinensis , being associated with burrows of snapping shrimps. According to Dolorosa & Werding (2014), E. philippinensis is typically encountered among coral rubble, rocks and decaying wood in the outer-most part of mangrove forests, together with two other, free-living porcellanid species.
Enosteoides habibi sp. nov. can be separated from E. turkayi , for instance, by the more strongly convex lateral margins and dorsal surface of the carapace; the third maxilliped carpus projecting ventrally as a subacute tooth ( vs. bluntly triangular in E. turkayi ); the noticeably stouter propodus of the ambulatory legs, being about four times as long as wide ( vs. almost five times as long as wide in E. turkayi ); and the posterodistal margin of the cheliped carpus ending as a blunt angle or small, subacute tooth ( vs. ending in a prominent, sharp tooth in E. turkayi ) (cf. Figs. 6A, D, I, J, M View FIGURE 6 ; Osawa 2016: figs. 2I, 3, 4A, C, F, G). The two species also differ in the general shape of the third thoracic sternite, which in E. habibi sp. nov. is distinctly separated from the fourth sternite and has rounded lateral lobes and slightly convex anterior margin ( vs. partly fused with the fourth sternite, with square-shaped lateral lobes and concave in E. tuerkayi ) (cf. Fig. 6F View FIGURE 6 ; Osawa 2016: fig. 2E). It is noteworthy that in the shape of the third thoracic sternite, E. habibi sp. nov. ( Fig. 6F View FIGURE 6 ) appears to be intermediate between E. lapis with a sinuous anterior margin (cf. Osawa et al. 2022: fig. 1E) and E. philippinensis and E. melissa , both with a strongly convex anterior margin (cf. Dolorosa & Werding 2014: fig. 2C). However, as for E. philippinensis , the most obvious difference between E. habibi sp. nov. and E. tuerkayi lies in the colour of the ambulatory legs, with typically two distinct, widely separated, red-purple bands, one on the carpus (sometimes missing) and one on the propodus (always present) in E. habibi sp. nov., contrasting to the entire carpus and propodus being deep red in E. turkayi (cf. Fig. 9–11 View FIGURE 9 View FIGURE 10 View FIGURE 11 ; Osawa 2016: fig. 1C, D).
All other species of the genus Enosteoides are morphologically more distinctive from E. habibi sp. nov. than E. melissa , E. philippinensis and E. turkayi , and can also be distinguished from the new species by differences in the colour patterns (e.g., Osawa 2009: figs. 1, 4A, B for E. lobatus and fig. 4D for E. palauensis ; Osawa & Chan 2010: figs. 56, 57 for E. ornatus ; Osawa 2016: fig. 1B for E. palauensis ; Osawa et al. 2022: figs. 1, 4A–D for E. lapis ; Hiller & Werding 2022: figs. 1–3 for E. spinosus ).
Enosteoides habibi sp. nov., E. melissa , E. lapis and possibly E. turkayi (rather than E. lobatus in Werding et al. 2016; see Osawa et al. 2022: 590) form a small ecological group of porcelain crabs associated, permanently or facultatively, with burrows of snapping shrimps ( Alpheus spp. ), which themselves live in partnership with gobiid fishes (see Karplus & Thompson 2011 for the most recent review). At least two species of Alpheus have been so far confirmed as hosts of porcelain crabs, viz. A. rapax ( Osawa 2016; present study) and A. bellulus Miya & Miyake, 1969 ( Werding et al. 2016). The specific snapping shrimp host or hosts of E. habibi sp. nov. and also of Palaemonella jamila sp. nov. (see above) remain to be determined by in situ observations and additional sampling of specimens at the type locality of both species.
In a recent phylogenetic analysis of selected porcellanid taxa ( Hiller & Werding (2022), which included E. ornatus , E. spinosus , E. palauensis , E. turkayi (the only species presumably associated with burrows of Alpheus spp. ) and E. philippinensis , Enosteoides was recovered as non-monophyletic. This result, although preliminary, indicates that a new genus should be established for E. palauensis , E. philippinensis and E. tuerkayi , together with affiliated species not included in the analysis of Hiller & Werding (2022), i.e., E. lapis , E. lobatus , E. melissa and E. habibi sp. nov. The configuration of the thoracic sternum, among other characters, could be used to distinguish the new genus from Enosteoides sensu stricto, represented by E. ornatus ( type species) and E. spinosus .
| FLMNH |
Florida Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |