Microphilypnus ternetzi Myers, 1927
|
publication ID |
https://doi.org/ 10.5281/zenodo.201744 |
|
DOI |
https://doi.org/10.5281/zenodo.5632262 |
|
persistent identifier |
https://treatment.plazi.org/id/B222878F-070A-FF9C-FF60-3CD1FC0EE9CD |
|
treatment provided by |
Plazi |
|
scientific name |
Microphilypnus ternetzi Myers, 1927 |
| status |
|
Microphilypnus ternetzi Myers, 1927 View in CoL
( Figs. 1 View FIGURE 1 , 5–9 View FIGURE 5 View FIGURE 6 View FIGURE 7 View FIGURE 8 View FIGURE 9 Table 2 View TABLE 2 )
Microphilypnus ternetzi Myers, 1927: 134 View in CoL (type locality “ Venezuela, Caño de Quiribana, near Caicara”); Weitzman & Vari, 1988: 449 (miniature Neotropical fishes; Table 1 View TABLE 1 ; listed); Mago-Leccia, 1970: 108 (checklist of fishes of Venezuela); Taphorn et al., 1997: 97 (checklist of freshwater fishes of Venezuela); Kullander, 2003: 662 (checklist of fishes of South and Central America); Lasso et al., 2004: 191 (freshwater fishes of Venezuela; rio Negro and Orinoco; listed); Thacker et al., 2006: 495, Fig. 2 View FIGURE 2 (taxonomic comments); Kullander in Vari et al. 2009: 51 (fishes of Guiana shield; listed); Eschmeyer, 2010: unpaginated (catalog of fishes); Prada-Pedreros et al., 2009: 178 (fishes of Puerto Carreño; listed).
Microphilypnus amazonicus Myers, 1927: 134 View in CoL (type locality Brazil, Manaus, Igarapé da Mãe Joana); Fowler, 1954: 321 (catalog of the freshwater fishes of Brazil; listed); Weitzman & Vari, 1988: 449 (miniature Neotropical fishes; Table 1 View TABLE 1 ; listed, Tab. 1 View TABLE 1 (miniature Neotropical fishes); Taphorn et al., 1997: 97 (freshwater fishes; zoological catalogue of Venezuela; listed); Menezes, 2003: 97 (checklist of marine fishes of Brazil); Kullander, 2003: 662 (checklist of fishes of South and Central America); Lasso et al., 2004: 191 (freshwater fishes of Venezuela; rio Negro and Orinoco; listed); Carvalho et al., 2006: 221 (in part; ecology); Thacker et al., 2006: 495, Fig. 2 View FIGURE 2 (taxonomic comments); Ferreira et al., 2007: 186 (fishes of Rio Branco; listed); Montag et al., 2008: 24 (fishes of Caxiuanã, Pará; listed); Montag et al., 2009: 617 ( Table 1 View TABLE 1 ; same as in Montag et al., 2008); Oliveira et al., 2009: 156 -7, 161 ( Table 1 View TABLE 1 ; fishes of the Reserva Catua-Ipixuna; listed); Eschmeyer, 2010: unpaginated (catalog of fishes).
Microphilypnus View in CoL sp. 1; Goulding et al. ¸ 1988: 127 (fishes of Rio Negro; listed); 132 (fish community; Anavilhanas, Rio Negro): 148, 150 (diet; Cladocera, mosquito larvae).
Microphilypnus View in CoL sp.; Mago-Leccia, 1971: 10 (fishes of Casiquiare; listed as “ Microphilynus sp. cerca de M. amazonicus View in CoL ”).
Microphilypnus View in CoL sp. 2; Goulding et al. ¸ 1988: 127 (fishes of Rio Negro; listed).
Microphilypnus View in CoL sp.; Sabaj-Perez in Vari et al., 2009: 92 –93, Plate 16, Fig. E (photograph; ANSP 180643; Venezuela, Rio Negro).
Doubtful references for this species
Microphilypnus cf. macrostoma View in CoL ; Bergleiter, 1999: 64, Fig. 52 (ecology; fishes of Igarapé Nazaré, Rio Xingu). Microphilypnus macrostoma View in CoL ; Py-Daniel et al., 2007: 97, 116 (fish fauna from the Middle Madeira river; listed).
Type material examined. Lectotype (by present designation): SU 18080, 15.2 mm SL, coll. by C. Ternetz in Venezuela: Caño de Quiribana near Caicara, May 1925. Paralectotypes: CAS 76818, 3, 11.2–14.1 mm SL, MCZ 31710, 1, 12.1 mm SL, same data from the lectotype; Holotype of Microphilypnus amazonicus : CAS 76819, 1 male, 18.1 mm SL, coll. by C. Ternetz in Brazil, Amazonas: Manaus, igarapé da Mãe Joana, 23 December 1924.
Non-type material examined. 415 specimens 8.1–23.2 mm SL.
BRAZIL. INPA 4683, 3, 16.5–18.4 mm SL, Amazonas: rio Negro basin, Igarape Anatiranga; INPA 6890, 1, 15.7 mm SL, Pará: rio Tapajós basin, Itaituba, Pimental, downstream of the mouth of river Jamanxim, 04º33’41” S
56º15’50” W; INPA 7271, 8, 14.8–16.1 mm SL, Pará: rio Tapajós basin, Mucum, near Aveiro, offshore the sand
beach; INPA 9627, 1, 16.9 mm SL, Amazonas: rio Negro basin, Lago Panema, nearby the mouth of river Jaú,
01º53'51" S 61º25'39" W; INPA 11628, 1, 17.6 mm SL, Amazonas: rio Negro basin, rio Cuieiras; INPA 11629, 20, SL 12.9–22.8 mm, Amazonas: Barcelos, rio Caures; INPA 11896, 3, 13.3–15.7 mm SL, Amazonas: rio Negro
basin, Ilha Camaleão, in front of Hotel Tropical, 03º08'43"S 60º01'23"W; INPA 15474, 39921, 10, 13.9–19.1 mm SL, Rondônia: rio Aripuanã basin, Jenipapo, 06º07'59" S 60º13'27" W; INPA 27536, 12, 11.3–19.0 mm SL,
Amazonas: Coari, Igarapé do Boinha, upstream from community Sta. Luzia do Boia, Catua, 03º47'52" S 64º10'32" W; INPA 28869, 10, 12.9–15.6 mm SL, Amazonas: rio Purus basin, Beruri, Ayapuá, Rio Itauba; INPA 29168, 10, 12.3–15.1 mm SL, Amazonas: rio Purus basin, Beruri, Lago Uauaçu 04º22'58" S 62º40'35" W; INPA 29222, 8,
11.1–14.8 mm SL, Amazonas: rio Purus basin, Beruri, Lago Uauçu, igarapé Ilhinha; INPA 34819, 5, 8.1–9.2 mm SL, Amazonas, Parauari, Maués, nearby Santo Sinai community, 04º21'39" S 57º35'01" W; INPA 34849, 5, 9.4–
15.7 mm SL, Amazonas: Parauari, Maués, Igarapé São Salvador nearby Ebenezer community, 03º59'58" S
57º35'20" W; MCP 13601, 2, 14.1-16.1 mm SL, Pará: rio Tapajós basin, near Santarém, 0 2o 26' S 54o44' W; MCP 30361, 30368, 5, 16.7-20.7 mm SL, Amazonas: rio Solimões basin, Alvarães, 03º06'44"S 64º47'32" W; MCP
38047, 44, 13.6-16.3 mm SL, Rondônia: rio Madeira basin, Igarapé das Araras, 10º00'49" S; MCP 38871, 2, 15.2- 18.4 mm SL Rondônia, rio Madeira basin: Rio da Lage, 10º26´00” S 65º20'40" W; MCP 38888, 4, 19.1–19.7 mm SL, Amazonas: rio Madeira basin, Rio Maicimirim, 07º37'56" S 62º39'44" W; MPEG 663, 2, 15.0- 16.1 mm SL,
Amazonas: rio Negro basin, near mouth of Rio Urubaxi; MPEG 666, 10, 16.4-21.3 mm SL, Amazonas: rio Negro basin, Anavilhanas, Lago do Prato; MPEG 10421, 3, 13.7-16.0 mm SL, Pará: Rio Caxiuanã; MPEG 10690, 2, 12.9- 13.5 mm SL, Pará: rio Amazonas basin: Tucuruí, 03º38'13" S 49º37'11" W; MPEG 14530, 1, 15.7 mm SL, Pará: rio Trombetas basin, Rio Saracá; MPEG 15326, 1, 16.2 mm SL, Pará: rio Amazonas basin, Nhamundá, 01º42'23" S
57º12'10" W; MZUSP 5753, 65531, 14, 12.2–22.0 mm SL, Amazonas: Urucará, Lago Carauaçu; MZUSP 6366, 2, 14.6-14.8 mm SL, Amazonas: rio Purus basin, Lagoa Castro; MZUSP 6440, 10, 12.6–16.2 mm SL, Amazonas: rio Purus basin, igarapé of Lago Beruri; MZUSP 8428, 5, 8.1–16.6 mm SL, Pará: Santarém, Alter do Chão, Igarapé
Jacundá; MZUSP 9698, 5, 11.7–13.8 mm SL, Brazil, Amazonas: Lago Supiá, in front of Codajás; MZUSP 22078, 4, 12.5-13.4 mm SL, Pará: rio Tapajós basin, Monte Cristo, Lago de Santa Clara; MZUSP 27022, 42734, 31,
10.9–14.6 mm SL, Amazonas: Lago Miuá, upstream from Codajás; MZUSP 28056, 2, 16.0– 18.6 mm SL, Roraima: Caracaraí, mouth of rio Branco; MZUSP 28058, Brazil, 2, SL 13.1, 14.2 mm SL, Roraima: Caracaraí, rio Jufari, Baía Grande; MZUSP 31176, 2, 22.1–23.2 mm SL, Amazonas: rio Negro basin, Anavilhanas, Lago do Prato; MZUSP 42725, 1, 12.5 mm SL, Pará: lakes along bank of rio Tocantins, near Tucuruí; MZUSP 42726, 1, 15.1 mm SL, Amazonas: Ati-Paraná, Igarapé Tucuxi; MZUSP 42730, 1, 14.1 mm SL, Pará: rio Capim, Lago Maria Preta; MZUSP 42731, 8, 11.8–15.9 mm SL, Pará: rio Capim, Lago Jurunundéua; MZUSP 42735-6, 19, 13.5–19.4 mm SL, Amazonas: lake near igarapé in Lago Janauacá, right bank of rio Solimões; MZUSP 43500, 3, 15.7–19.4 mm SL, Amazonas: rio Negro basin, Paricatuba; MZUSP 47992, 64912, 18, 4 c & s, 13.9–19.6 mm SL, Amazonas: rio Negro basin, Rio Urubaxi; MZUSP 54565, 62260, 62422, 5, 15.2–18.2 mm SL, Amazonas: Paricatuba, lake near bank of rio Negro; MZUSP 56950, 1, 14.1 mm SL, Amazonas: rio Purus basin, 1.1 km downstream Lagoa de São Tomé; MZUSP 59158, 5, 14.0– 18.2 mm SL, Amazonas: rio Negro basin, Cantagalo; MZUSP 74269, 3, 8.0– 18.9 mm SL, Amazonas, Manaus, left bank of rio Cuieiras, approx. 40 km from the mouth;; MZUSP 75563, 19, 17.5-22.0 mm SL, Amazonas: rio Solimões basin, Janauacá, Lago Murumuru; MZUSP 88728, 16, 13.0– 16.5 mm SL, Amazonas: Rio Preto da Eva, near Igarapé Tauari, 02º47'25" S 59º38'11" W; MZUSP 92262, 4, 13.0– 18.5 mm SL, Amazonas, rio Negro basin, Rio Tiquié, between São José "do meio" and Floresta, 00º13'00" N 69º36'00" W; MZUSP 92523, 3, 14.0– 16.3 mm SL, Amazonas, rio Negro basin, igarapé Castanha, near mouth of Rio Tiquié, 00º12'00" S 69º35'00" W; MZUSP 92835, 4, 12.6–19.8 mm SL, Pará: Santarém, Igarapé Juá, 7 km from Santarém along airport road, 02º26’00” S 54º46’52” W; MZUSP 92849, 2, 13.3–14.4 mm SL, Brazil, Pará: rio Tapajós basin, Alter do Chão; MZUSP 92978, 9 (1 c & s), 13.7–18.8 mm SL, Amazonas, rio Negro basin, Rio Tiquié, between São José and Bela Vista, 00º13'00" N 69º36'00" W; MZUSP 93062, 3, 13.5–17.0 mm SL, Amazonas, rio Negro basin, Rio Tiquié, igarapé Castanha, nearby Farm São Pedro, 00º11'00" N 69º35'00" W; MZUSP 93385, 4, 13.1– 15.1 mm SL,Amazonas, rio Negro basin, Rio Tiquié, between Fronteira and São Luís, 00º13'00" N 69º35'00" W; MZUSP 100240, 1, 17.6 mm SL, Amazonas: rio Solimões basin, left margin, Lago Itacaré, 03º21'00" S 60º19'00" W; MZUSP 100412, 20 (3 c & s), 12.2–17.0 mm SL, Pará: Santarém, rio Tapajós basin, 02º28'05" S 54º55'34" W; MZUSP 102966, 2, 13.4-15.2 mm SL, Amazonas: rio Solimões basin, Lago do Castanho; MZUSP 104927, 6, 14.6-19.6 mm SL, Amazonas: rio Negro basin, left margin, Igarapé Demuriari, 00º15´51" S 67º10'16" W; MZUSP 104928, 3, 9.2-17.4 mm SL, Amazonas: rio Negro basin, Igarapé Baxiria, 00º07´24" S 67º07'54" W; NRM 18376, 5, 11.3–14.0 mm SL, Pará, rio Tapajós basin, Rio Arapuins at at Vila Brasil, 60–100 km from Santarém; UFRO-I 1310, 1, 16.2 mm SL, Rondônia: rio Madeira basin, Igarapé Arara, 10º00'51" S 65º19'05" W; UFRO-I 1363, 13, 12.3–17.1 mm SL, Rondônia: rio Madeira basin, rio Jaciparaná, 09º12'59" S 64º38'56" W; UFRO-I 1944, 1 male, 12.9 mm SL, Rondônia: rio Madeira basin, rio Jaciparaná, Lago Madalena, 09º17'07" S 64º23'59" W; UFRO-I 2624, 4, 10.2–13.8 mm SL, Rondônia: Porto Velho, Igarapé Belmont, stream on right margin of river Madeira, 08º39'43" S 63º51'42" W; UFRO-I 2913, 2, 12.3-13.5 mm SL, Rondônia: rio Madeira basin, Igarapé Belmont, 09º12'59" S 64º38'56" W; UFRO-I 3057, 6389, 10, 12.5–15.4 mm SL, Rondônia: Porto Velho, foz do igarapé Arara, 10º00'50"S 65º19'04" W; UFRO-I 6388, 5, 12.6-14.8 mm SL, Rondônia; rio Madeira basin, Rio São Lourenço, 09º22'03 " S 64º51'00 " W; UFRO 6390, 1, 12.1 mm SL, Rondônia: rio Madeira basin, Rio Sotério, 11º36' 03" S 65º13'44" W; UFRO-I 6391, 10, 11.6-14.7 mm SL, Rondônia: rio Madeira basin, Humaitá, Lago do Puruzinho, 07º22'11" S 63º03'33" W; UFRO-I 6392, 7, 10.6-13.8 mm SL, Rondônia: rio Madeira basin, rio Machado, 08º20'30" S 62º41'39" W; UFRO 6393, 10, 11.9-15.6 mm SL, Rondônia: rio Madeira basin, Porto Velho, Vila Teotônio, Igarapé Jatuarana, stream in left margin of rio Madeira; UFRO-I 6394, 6, 9.9-11.9 mm SL, Rondônia: rio Madeira basin, Lago do Cuniã, 08º18'56" S 63º 29'43" W; USNM 317427, 1, 16.9 mm SL, Rondônia: rio Machado, Lago Antonio.
COLOMBIA. CAS 70618, 1, 17.0 mm SL, rio Negro basin, Cucuhy, Santo Antonio; NRM 17954, 1, 13.6 mm SL, Puerto Narino, Quebrada Najarito
VENEZUELA. USNM 269999, 1, 14.6 mm SL. Department Rio Negro: Caño Chola, where crossed by road from San Carlos de Rio Negro basin to Solano, 01º58' N 67º00' W; USNM 270002, 4, 12.8–15.3 mm SL, Department Rio Negro: large rock in middle of river between Santa Rosa and Santa Lucia, 0–1.5 m depth, 01º20' N 66º52' W; USNM 270003, 8, 13.2–15.9 mm SL, Department Rio Negro: Caño Manu, tributary of Casiquiare River approx. 250 m upstream of Solano, 02º00' N 66º57' W; USNM 270004, 6, 12.1–16.2 mm SL, Department Ature: caño ca. 15 km south of El Burro on road from Puerto Ayacucho to El Burro, 0–1 m depth, 06º02' N 67º23' W; USNM 270005, 16, 13.0– 16.1 mm SL, Orinoco River: bank upstream of town landing, 01º55' N 67º03' W, 0–1.5 m depth.
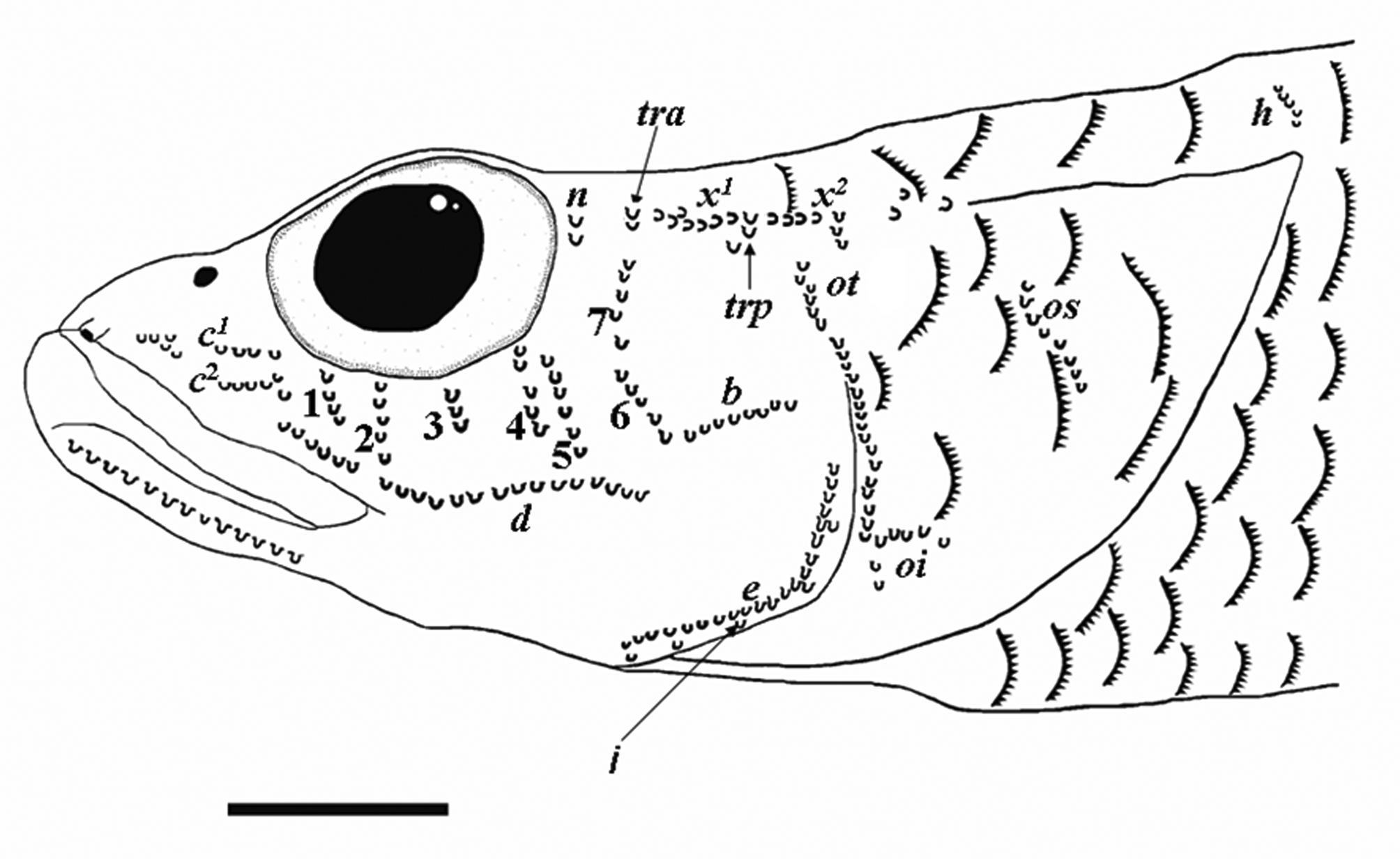
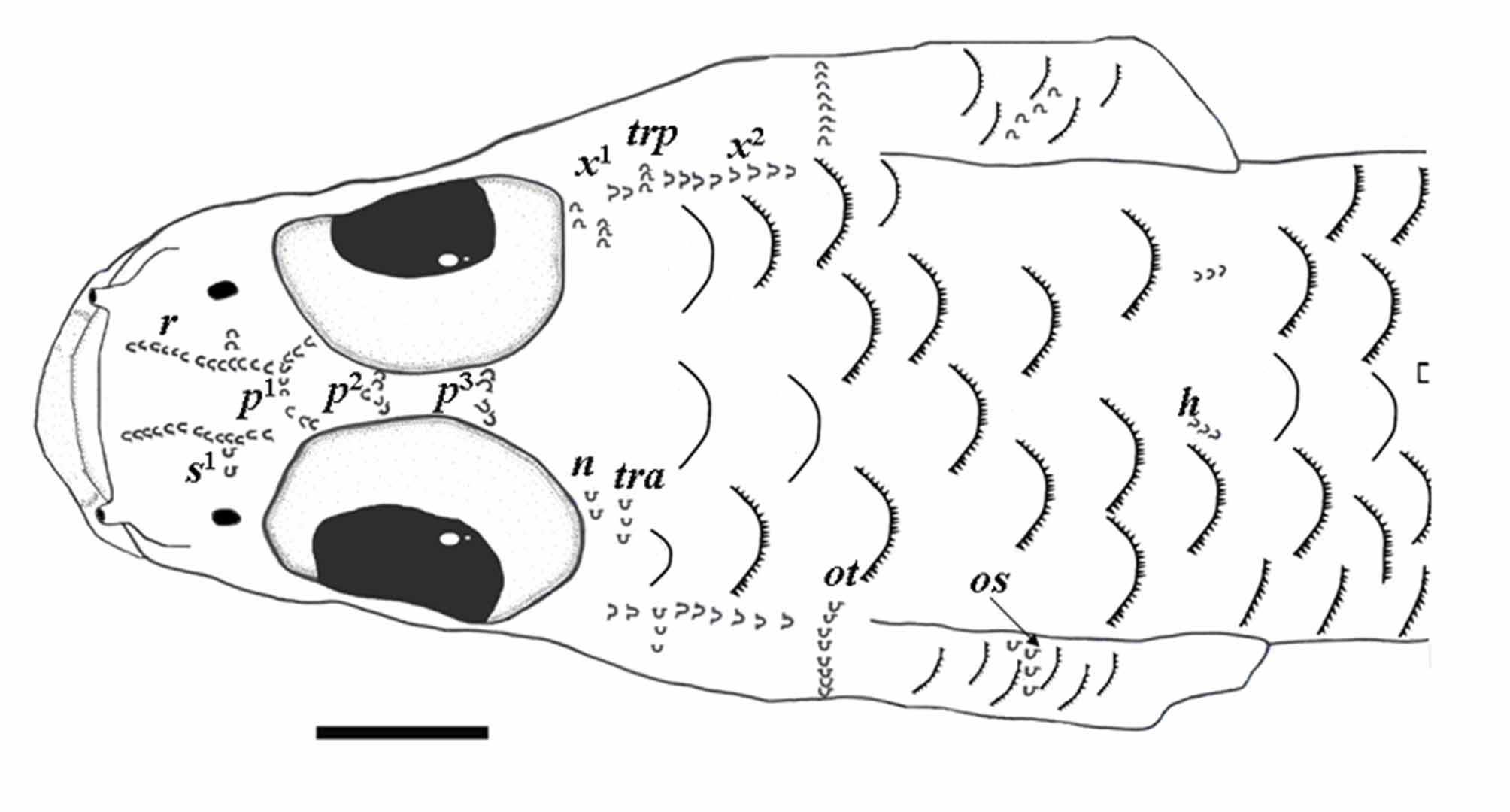
Diagnosis. Microphilypnus ternetzi can be identified by the following combination of characters: snout pointed but not depressed; upper jaw not reaching vertical through middle of eye; D. III–VII + 6–10; A. 6–10; P. 11–15; 23–30 scales in longitudinal row (usually 26 or more); opercle covered with cycloid or ctenoid scales; predorsal region partially to fully covered with cycloid or ctenoid scales; no head pores, 6–7 transverse rows of infraorbital sensory papillae below the eye; three long rows of interorbital sensory papillae; one predorsal longitudinal row of sensory papillae (row h) visible, short.
Description. Morphometric data in Table 2 View TABLE 2 . Body elongated and moderately compressed, with caudal peduncle long, slender. Head long, snout short with blunt to slightly pointed tip, anterior naris small, tubular, posterior naris a small circular slit; eye larger than snout, placed higher on head; interorbital region narrow. Upper jaw inclined, barely surpassing anterior border of eye; lower jaw protruded, inclined; lower lip stiffened. Tongue short, emarginated. Gill opening large, anterior margin on vertical that passes through anterior eye border; gill rakers 0– 8+6–10 (66 specimens examined).
Head with 6–7 transversal rows of sensory papillae below eye, penultimate row usually the shortest; two horizontal rows of papillae below eye, one below transverse rows (row d), other behind sixth transverse row (row b), both usually long. Three rows of papillae on opercle: one vertical (row ot); one short, joined to row ot (row oi), other long and oblique (row os) in most specimens, short, curved in some individuals from Rio Negro. Preorbital region with one longitudinal row on each side (r) and a short transverse row (s 1); interorbital region with three long rows of papillae (p rows), first row with 4–11 elements, remaining ones with 4–9 elements; postorbital transverse row n and tra present, short; oculoscapular longitudinal row x 1 short and x 2 long, transverse oculoscapular row trp short, between x 1 and x 2; predorsal longitudinal row h present, row m not visible ( Figs. 6–7 View FIGURE 6 View FIGURE 7 ).
Dorsal fin elements III + 8 (2), IV + 8 (1), IV + 9 (1), V+ 6 (1), V + 7 (12), V + 8 (28), V + 9 (5), VI + 6 (5), VI + 7 (62*), VI + 8 (209), VI + 9 (92), VI + 10 (4), VII + 8 (3) or VII + 9 (1); anal fin rays 6 (16), 7 (109*), 8 (227), 9 (60) or 10 (2). Pectoral fin rhomboid, medial rays the longest, rays: 11 (5), 12 (117), 13 (173*), 14 (123) or 15 (12); pelvic fins long, separate from base; first pelvic ray the shortest, second and third the largest; caudal fin margin convex; vertebrae 12+16 (type-material), 12–13+15–17=27–29 (17 specimens examined).
Trunk covered with ctenoid scales; scales absent on snout, chin (sometimes with one cycloid scale in largest specimens), interorbital region, belly, and pectoral fin base; trunk scales on longitudinal row 23 (6), 24 (39), 25 (86), 26 (98), 27 (79), 28 (47), 29 (20*), or 30 (4); scales on transverse row 6 (13), 7 (136), 8 (144*), or 9 (9); circumpeduncular scales 10 (1), 11 (56), 12 (214), or 13 (28); 3–10 large cycloid or ctenoid scales on opercle, in 3–4 transverse rows. Predorsal region covered with cycloid or ctenoid scales, partially naked in young specimens: scales on predorsal region 2 (1), 3 (11), 4 (26), 5 (18), 6 (16), 7 (7), 8 (29), 9 (58), 10 (76), 11 (56), 12 (36), 13 (7), or 15 (1). Lectotype of M. ternetzi and holotype of M. amazonicus only with scale pouches on trunk and without visible scales on head; one paralectotype (MCZ 31710) with ctenoid scales on opercle
Coloration in alcohol ( Fig. 5 View FIGURE 5 ) yellowish, with small aggregations of black or brown chromatophores on posterior margin of lateral trunk scales, sometimes with irregular rows of small spots along flank; rows of slender darkbrown markings along belly; inconspicuous, oblique dark stripe from anterior border of eye to upper jaw, 1–2 vertical stripes of same shape below eye; small dark-brown spots on opercle and pectoral-fin base; fins hyaline, with rows of small dark chromatophores along rays, rarely forming oblique stripes, diffuse spot on caudal fin base.
Remarks. In the original description ( Myers, 1927), M. amazonicus is distinguished from M. ternetzi by the number of scales on the longitudinal series (27 scales in M. amazonicus , compared with 29 in M. ternetzi ), dorsal fin elements (VI + 8 in M. amazonicus and V + 8 in M. ternetzi ), anal fin rays (8 in M. amazonicus and 7 in M. ternetzi ) and presence, in M. amazonicus , of a bony ridge in front of the orbit and a spiny bony projection on the ventral margin of the pre-operculum. Nonetheless, it should be emphasized that Myers examined few specimens of the genus he had created; one of the characters provided by him (bony point on pre-operculum) actually is not correct, since it is not found in any of the hundreds of specimens of Microphilypnus we have examined (see also Thacker et al., 2006). Furthermore, many of such characters could not be verified neither in the holotype of M. amazonicus nor in the syntypes of M. ternetzi , which are in poor condition. The solution was to compare specimens in good condition from areas near the type localities of both species.
The name Igarapé da Mãe Joana is given for many localities along the lower Amazon; however, it is known that Dr. Ternetz was in “the mouth of rio Negro” in the end of 1924 (when the types of M. amazonicus and M. macrostoma were taken), traveling along this river in January 1925 and then reaching Casiquiare and Orinoco ( Stehlin, 1929, p. 31), so the locality where the holotype of M. amazonicus was collected is indeed in rio Negro.
A comparison between the specimens from rio Negro and those from Orinoco basin and Casiquiare revealed that they are practically identical: individuals of both localities have short, slightly pointed snout, maxilla short, not reaching vertical that passes through the middle of eye, and bear ctenoid scales on opercle, which are also visible in one paralectotype of M. ternetzi , as detailed above. Moreover, specimens collected in the same area, whether from rivers Negro or Orinoco, possess either 27 or 29–30 scales in longitudinal series, five or six spines on the first dorsal fin, and 7 or 8 anal rays. These facts strongly suggest that characters used by Myers to diagnose M. amazonicus and M. ternetzi are uninformative or incorrect, and the species should be recognized as synonyms. The name Microphilypnus ternetzi is here used as the valid name based on Articles 24.1 and 24.2 of the International Code of Zoological Nomenclature (ICZN, 1999), because it represents the type species of this genus. Microphilypnus amazonicus Myers is its junior synonym.
Goulding et al. (1988) reported two species of Microphilypnus from the Rio Negro, which they identified as “sp. 1” and “sp. 2”, but provided no data to justify this classification. All the specimens of Microphilypnus collected by Michael Goulding and deposited in MZUSP have been examined during this study, and it could not be distinguished two species in his material; furthermore, all the specimens of Microphilypnus that he collected possess large ctenoid scales on the predorsal region and opercle, as the lectotype. For these reasons, the two forms recognized by Goulding et al. (1988) are herein regarded as Microphilypnus ternetzi .
Body width at pectoral base 44.2 33.3–38.4 38.5 29.9 59.2 Body width at 2nd dorsal 34.3 26.7–31.3 28.8 17.3 44.2 origin
Bergleiter (1999), in his monograph on the fish community of the Xingu River, provided some information and an illustration (Fig. 52) of the head of a species of Microphilypnus collected in the Xingu and identified by him as Microphilypnus sp. cf. macrostoma . Although the abovementioned image is not detailed, we observed that it depicts a specimen with short, blunt snout, and thus could not be M. macrostoma . Surprisingly, however, there is no material or additional literature about the occurrence of Microphilypnus in the Xingu, so it cannot be confirmed at this time if these specimens by Bergleiter are M. ternetzi or represent an undescribed species.
Py-Daniel et al. (2007) report the occurrence of Microphilypnus amazonicus and M. macrostoma in the middle river Madeira. They further state that the first species was taken only in rio Aripuanã, whereas M. macrostoma was found in other localities. A further examination of much material from Madeira reveals that, in spite of the variation among samples from this locality, as will be detailed below, M. macrostoma has not been currently encountered in this drainage. Although the real identity of M. macrostoma of Py-Daniel et al. can not be established, we rather suspect that it is M. ternetzi .
Geographical variation. Most specimens collected near the type localities of M. ternetzi and M. amazonicus present fully scaled predorsal region, ctenoid scales on opercle (sometimes also on predorsal region), three long series of papillae in introrbital region and two longitudinal ones on predorsal region, and series of papillae os on opercle short, curved. However, many specimens from other localities (e.g. most of individuals collected in rivers Solimões, Purus and Tapajós) have cycloid scales on opercle and series os long, straight, and in some specimens (mainly from Rio Madeira) the predorsal region is naked in posterior half (see Figs. 8 View FIGURE 8 a, d).
Given the striking difference in the disposition of head scales between the morphs from different localities, it could be suggested that they represent distinct species. However, some facts must be taken into account. Firstly, these morphs are virtually identical between them in all other characters, particularly body pigmentation and head shape (but see Figs. 8 View FIGURE 8 a, d). Additionally, scale morphology in fishes changes during the ontogeny, beginning always as cycloid scales and then becoming ctenoid (Rosen, 1910; Hase, 1911); moreover, the regeneration of ctenoid scales in cycloid scales is common in gobioids, and specimens with cycloid or ctenoid scales but identical in other aspects were found in the same site (e.g. Madeira, W. Ohara, pers. commun.). Also, cycloid or ctenoid scales were detected on different sides of the same specimen, so this character is not very reliable for discriminating species.
A comparison of M. ternetzi populations with Principal Component Analysis was made (see Methods and Materials above) and the results are presented in Fig. 9 View FIGURE 9 . Again, no detectable difference was found among the samples from different localities, either with cycloid (e.g. Purus) or ctenoid (e.g. Negro) scales on opercle. Finally, although specimens with ctenoid scales on opercle appear to have more scales in longitudinal series than those with cycloid opercular scales (27–28 vs. 24–26), the superposition among samples is yet too large to warrant clear distinction between morphs from different localities, so these populations are here identified as M. ternetzi , regardless of the fact that more refined analyses may reveal that some of these populations represent undescribed species.
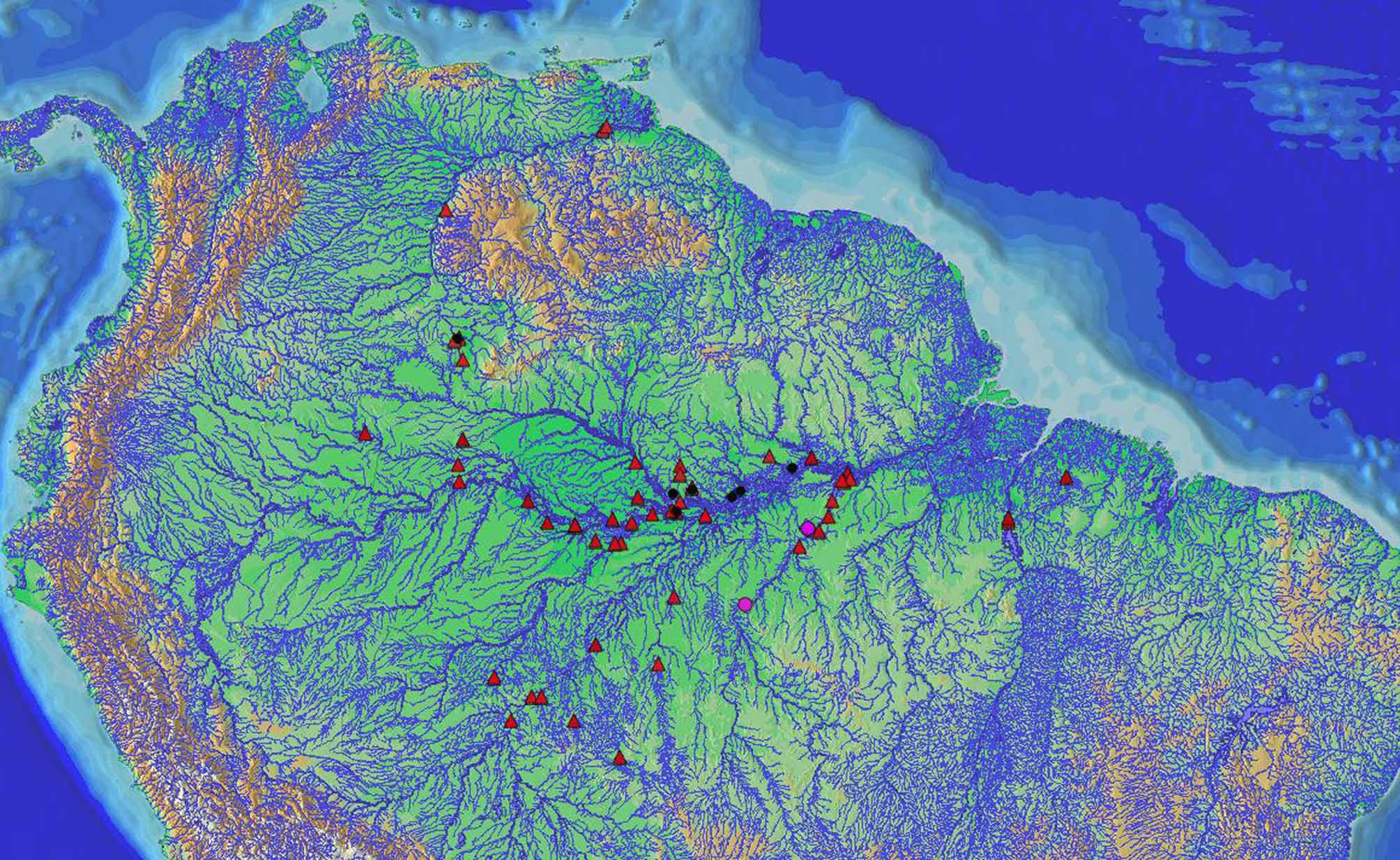
Distribution. Microphilypnus ternetzi is found in the tributaries of the following basins: Negro, Orinoco, Tapajós, Capim, Madeira, Purus, Solimões, Trombetas and Tocantins ( Fig. 1 View FIGURE 1 ).
TABLE 2. Morphometric data for Microphilypnus ternetzi, given as percentages of the standard length (% SL) and of head length (% HL).
| Lectotype | Paralectotypes (N=3) | Holotype of M. amazonicus | Non-type specimens (N=164) Minimum Maximum | |
|---|---|---|---|---|
| Standard length (mm) | 15.2 | 11.2–14.1 | 18.1 | 8.0 23.2 |
| %SL | ||||
| Head length | 30.6 | 30.0–31.7 | 30.1 | 27.4 36.0 |
| Preanal distance | 53.5 | 50.0–55.1 | 51.7 | 47.4 61.5 |
| Postanal distance | 45.9 | 41.2–46.2 | 37.2 | 38.2 54.2 |
| Caudal peduncle length | 26.0 | 23.1–32.9 | 25.8 | 21.5 42.0 |
| Pectoral fin length | 19.3 | 16.9–27.9 | 21.2 | 15.7 31.0 |
| Pelvic fin length | damaged | 11.4–19.8 | 20.5 | 11.8 32.8 |
| Caudal fin length | damaged | damaged | 14.2 | 13.3 32.4 |
| %HL | ||||
| Snout length | 18.7 | 20.5–21.9 | 17.6 | 10.5 30.5 |
| Orbital diameter | 22.3 | 23.1–23.7 | 23.7 | 15.6 42.7 |
| Interorbital width | 3.9 | 4.0–5.7 | 3.5 | 2.6 9.7 |
| Upper jaw length | 27.0 | 25.0–32.2 | 27.5 | 20.4 37.6 |
| Lower jaw length | 35.8 | 38.4–40.5 | 34.1 | 25.7 47.7 |
| Head width | 45.3 | 49.1–49.8 | 40.0 | 33.3 62.4 |
| Head depth | 45.7 | 43.3–47.2 | 46.6 | 40.5 64.6 |
| Predorsal distance | 115.6 | 111.9–112.3 | 112.5 | 100.2 142.2 |
| CAS |
California Academy of Sciences |
| MCZ |
Museum of Comparative Zoology |
| INPA |
Instituto Nacional de Pesquisas da Amazonia |
| MCP |
Pontificia Universidade Catolica do Rio Grande do Sul |
| MPEG |
Museu Paraense Emilio Goeldi |
| MZUSP |
Museu de Zoologia da Universidade de Sao Paulo |
| NRM |
Swedish Museum of Natural History - Zoological Collections |
| USNM |
Smithsonian Institution, National Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Microphilypnus ternetzi Myers, 1927
| Caires, Rodrigo A. & Figueiredo, José Lima De 2011 |
Microphilypnus
| Vari 2009: 92 |
Microphilypnus
| Goulding 1988: 127 |
Microphilypnus
| Goulding 1988: 127 |
Microphilypnus
| Mago-Leccia 1971: 10 |
Microphilypnus ternetzi
| Vari 2009: 51 |
| Prada-Pedreros 2009: 178 |
| Thacker 2006: 495 |
| Lasso 2004: 191 |
| Kullander 2003: 662 |
| Taphorn 1997: 97 |
| Weitzman 1988: 449 |
| Mago-Leccia 1970: 108 |
| Myers 1927: 134 |
Microphilypnus amazonicus
| Montag 2009: 617 |
| Oliveira 2009: 156 |
| Montag 2008: 24 |
| Ferreira 2007: 186 |
| Carvalho 2006: 221 |
| Thacker 2006: 495 |
| Lasso 2004: 191 |
| Menezes 2003: 97 |
| Kullander 2003: 662 |
| Taphorn 1997: 97 |
| Weitzman 1988: 449 |
| Fowler 1954: 321 |
| Myers 1927: 134 |