Neoceratitis asiatica (Becker), BeCker
|
publication ID |
https://doi.org/10.11646/zootaxa.4363.4.7 |
|
publication LSID |
lsid:zoobank.org:pub:C5E158E8-4516-4F7B-B7A1-AC2AFAD1F02A |
|
DOI |
https://doi.org/10.5281/zenodo.5621902 |
|
persistent identifier |
https://treatment.plazi.org/id/407A6A1F-FFAD-FFD8-4A93-FA83FBEBFBD4 |
|
treatment provided by |
Plazi |
|
scientific name |
Neoceratitis asiatica (Becker) |
| status |
|
Neoceratitis asiatica (Becker) View in CoL
belongs to the family Tephritidae , and the damage it causes has seriously affected the production of wolfberry in recent years, especially organic wolfberry in Ningxia, China. Within China, N. asiatica is mainly distributed in Ningxia, Tibet and Xinjiang, which contain areas for special production of wolfberry ( Lycium barbarum L.). Under the combined effect of many factors, including climate change, field management and irrigation mode, the regional and localized occurrence of N. asiatica becomes more obvious ( Zheng, 2015). N. asiatica occurred seriously especially in an organic wolfberry garden without sprayed pesticides. N. asiatica adults lay eggs in the peel of wolfberry and larvae feed on flesh until pupation in the soil. The surface of the wolfberry in the early stage shows no obvious symptoms, but white curved stripes appear on the surface in the late stage, indicating the flesh is gone and the fruit is full of frass ( Peng et al., 2000; Wang, 2015; Xue, 2009) ( Fig. 1 View FIGURE 1 ). Wolfberry in this condition lost commodity or medicinal value, which are important influences in the production of wolfberry in China. The percentage of fruit trees that are infested by N. asiatica was nearly 70% in severe areas, and the damage caused by N. asiatica seriously affected the production of wolfberry in China; therefore, it has become a top priority requiring highly efficient, ecologically and environmentally safe technology ( Zheng, 2015). N. asiatica has three generations per year, and the average number of days required for a generation is 41.4 days. The eclosion of adults begins in mid-May every year, and after mid-September the pupae overwinter in 1 to 3 inch deep soil ( Wang, 2015; Xue, 2009). Normally, N. asiatica lays one egg per fruit; occasionally 2 or 3 eggs are laid in one fruit, but in these cases only one larva survives ( Wu & Meng, 1963; Zhang, 2016).
Chinese wolfberry is well known both domestically and overseas as a valuable medicinal and edible special economic plant resource and a traditional export agricultural product. Ningxia is the main base for cultivation of wolfberry in China. In recent years, there has been development of wolfberry production, not only in the traditional cultivation area of Ningxia, but also in Inner Mongolia, Xinjiang, Hebei, Gansu, etc. Chinese wolfberry plays an important role in improving the ecological environment and implementing the sustainable development strategy of agriculture ( Xue & Lin, 2009). Investigations into the production of wolfberry in the 1960s found that the pest problem, especially the occurrence of N. asiatica , affected the yield and quality of wolfberry seriously (Wu et al., 1963), but there were few reports of this pest in the nearly half a century following. However, there have been outbreaks of N. asiatica in recent years in Ningxia. In most of the new planting areas, people are inexperienced in the identification of N. asiatica , and larvae often need to be reared to the adult stage for identification. Since species identification is the basis of field monitoring and scientific governance, grass-roots technical staffs and farmers require identification results timely in order to guide field management. Therefore, simple and rapid identification of N. asiatica is necessary in wolfberry planting areas.
With the rapid development of molecular biology, many of its techniques are now used in the identification of insects. DNA barcoding is popular among researchers because of its simplicity and precision. The Barcode of Life Data system (BOLD) is an integrated web platform that helps researchers to analyze DNA barcode data. It is convenient and available for researchers to accomplish the identification of many biological individuals ( Ratnasingham & Hebert, 2007). By August 18, 2017, there were 5,622,724 specimens with barcodes, including 172,506 insect species, of which 858 are Tephritidae species (http://www.boldsystems.org/). Hebert et al. (2003a) first proposed the concept of DNA barcoding and established the mitochondrial gene cytochrome c oxidase I (COI) as a taxonomic tool for animals. COI gene can generally distinguish closely allied species in all animal phyla and the results indicating that it has a high degree of accuracy ( Hebert et al., 2003b).
In the present study, we identified 15 samples of fruit flies from Ningxia using morphological and molecular methods. Morphological features helped us identify the adult specimens and DNA barcoding based on COI gene was used for larval and pupal identification. This study fills in the blank of DNA barcode sequences for N. asiatica , and discusses the significance of rapid identification of this species.
Materials and methods
Specimen collection. Five adults, five pupae and five larvae were collected in wolfberry trees from Zhongning, Ningxia during August 2015. All specimens were preserved in 100% ethanol and stored at -80? before DNA extraction.
Morphological identification. All specimens were observed using a UV-C optical confocal microscopy image system (Olympus, Tokyo, Japan) and all are conserved in the Plant Quarantine Laboratory in the Department of Entomology of China Agricultural University. Images were taken before DNA extraction.
DNA extraction, COI amplification and sequencing. Total genomic DNA was isolated from whole individuals of N. asiatica using the commercial TIANamp Genomic DNA Kit (TIANGEN, China) and following the manufacturer s protocol. Polymerase chain reaction (PCR) was completed in a final volume of 25µl containing 12.5µl 2×Taq PCR MasterMix, 9.5µl sterilized distilled water, 1µl DNA as a template, 1µl forward and reverse primer (10µM), respectively. PCR was performed with a pair of universal primers, LCO1490 (forward, 5’- GGTCAACAAATCATAAAGATATTGG-3’) and HCO2198 (reverse, 5’-TAAACTTCAGGGTGACCAAAAAATCA- 3’) ( Folmer et al., 1994). The following thermal cycling profile was used: 94? for 3 min; followed by 35 cycles of 94? for 1 min, 50? for 1 min and 72? for 1 min; then 72? for 10 min. The reaction was performed on Veriti TM 96- well Thermal Cycler (ABI, USA). The amplified PCR products were separated on 1.5% (w/v) agarose gel (1×Tris Acetate-EDTA buffer), stained with GeneGreen Nucleic Acid Dye (TIANGEN, China) and visualized under UV light. The PCR products were purified and bi-directional sequencing using the same amplification primers commercially through Beijing Genomics Institute (BGI, Beijing, China).
Data analyses. All sequences obtained by sequencing were analyzed with BioEdit software (Borland, CA, USA) for testing sequence peak plot quality and proofreading. After sequences assembly and multiple sequences alignment using DNAMAN version 6.0.3.99, we deleted the primer sequences and low-quality base pairs and obtained the COI gene partial sequence for each specimen; the sequences were then submitted to GenBank for accession numbers. The pairwise genetic distances for COI gene based on Kimura 2-Parameter method ( Kimura M., 1980) were computed using Molecular Evolutionary Genetics Analysis version 7.0 (MEGA 7.0, PA, USA) ( Kumar et al., 2016). The minimum, maximum and mean genetic distances of the intra-specific and inter-specific divergence values were calculated using Excel. A barcoding gap analysis was performed to evaluate the utility of COI gene for N. asiatica identification. In the phylogenetic analysis, neighbour-joining (NJ) and maximum likehood (ML) trees were graphically displayed with third codon positions removed. The NJ tree was conducted in MEGA version 7.0 and the percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (1000 replications) were shown next to the branches ( Felsenstein, 1985). The ML tree was constructed using PhyML (http://www.atgc-montpellier.fr/phyml/) with GTR substitution model.
Results
Morphological characteristics of N. asiatica . Based on the morphological characteristics observed by microscopic examination, we identified all adult samples collected as N. asiatica ( Fig. 2 View FIGURE 2 ), in accordance with the description and illustration of Wang (1996). The body colour and wing patterns, particularly the small dark circle inside the subbasal triangular hyaline indentation from the costa on the wing are the most obvious characteristics distinguishing this species.
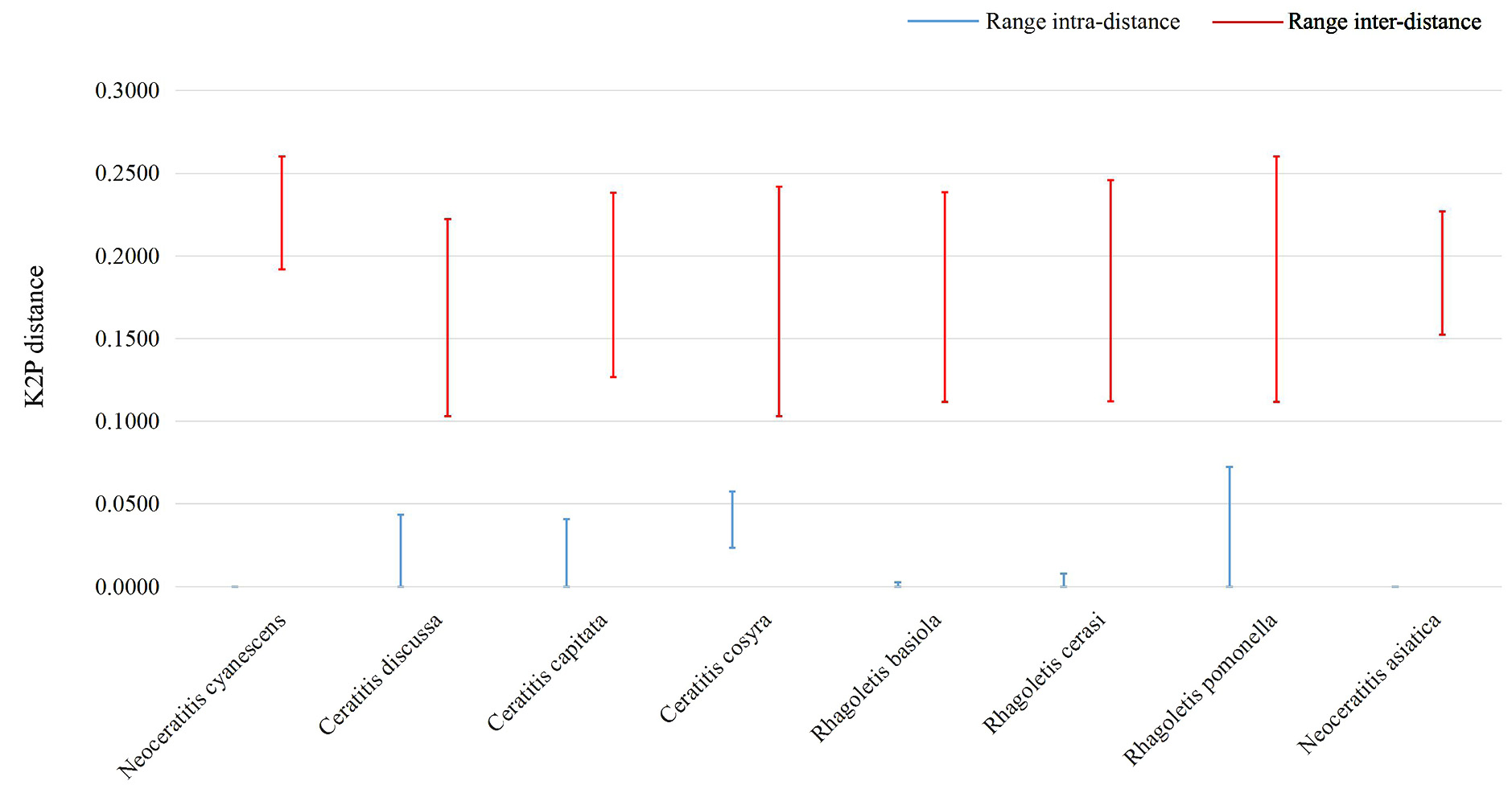
Genetic distance and barcoding gap analysis. We obtained 15 COI gene sequences from known N. asiatica adults, unknown pupae and unknown larvae. All sequences were deposited in GenBank and are available under accession numbers listed in table 1. The COI gene sequence Kimura 2-parameter distance between unknown samples and known N. asiatica adults was 0 (table 2). The minimum inter-specific genetic distance of each species was significantly higher than the maximum intra-specific genetic distance and there is an obvious gap between the intra- and inter-specific divergence ( Fig. 3 View FIGURE 3 ).
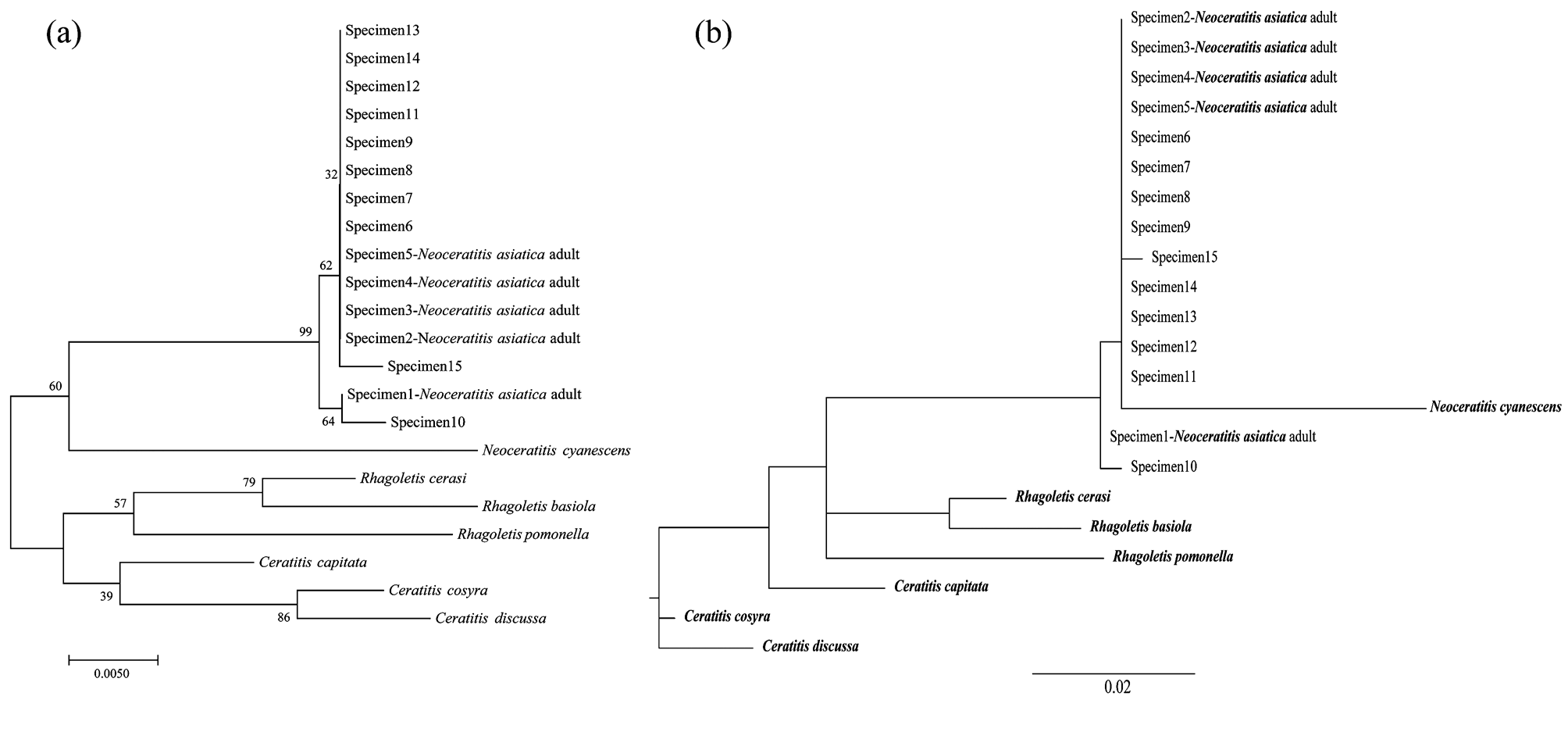
Phylogenetic analysis. The NJ tree based on mtDNA COI gene sequences showed that unknown samples were within the same cluster as N. asiatica adults ( Fig. 4a View FIGURE 4 ). They formed sister cluster to N. cyanescens , which belongs in the same genus. The ML tree displayed specimen 1 (a known N. asiatica adult) and specimen 10 as two individual branches in the cluster; the other 13 specimens formed a sister cluster to N. cyanescens ( Fig. 4b View FIGURE 4 ). Both NJ and ML trees suggested that the samples can be distinguished clearly.
Discussion
From morphological identification, N. asiatica can be readily discriminated from other Tephritidae species. The small circle inside the right angle to the costa is the obvious characteristics to distinguish this species. In the present study, the five N. asiatica adults were successfully identified by microscopic observation.
Many molecular biology techniques have been applied to insect identification, such as restriction fragment length polymorphisms (RFLP) ( Botstein et al., 1980; Qin et al., 2013), random amplification of polymorphic DNA (RAPD) ( Wilkerson et al., 1995; Zhang, 2004), single-strand conformational polymorphisms (SSCP) ( Boge et al., 1994; Li et al., 2003), DNA barcoding ( Jiang et al., 2014a; Liu et al., 2011), species-specific primer PCR ( Jiang et al., 2013; Jiang et al., 2014b; Yang et al., 2013) and real-time PCR ( Bai et al., 2011; Xu et al., 2010). Among them, DNA barcoding shows a prominent advantage because of it is general, accurate and fast. The DNA barcodes method has become a useful common method for identifying different insect species of Lepidoptera ( Hajibabaei et al., 2006; Janzen et al., 2005), Hymenoptera ( Smith et al., 2005), Diptera ( Armstrong & Ball, 2005; Cywinska et al., 2006; Cywinska et al., 2010), etc. BOLD, as an informatics workbench of DNA barcodes, is a species identification tool that, when submitting sequences from the barcode region, will return taxonomic matches and the rapid identification of species based on BOLD can be accomplished. To achieve rapid identification without the long time needed for rearing to adults for morphological identification, the larvae and pupae we collected were successfully identified by use of DNA barcoding in 2 days. The submitted sequences have been matched to N. asiatica with 100% similarity in BOLD. Our work make the sequence of Neoceratitis asiatica publically available.
The combination of morphological and molecular identification in our study indicated that COI-based DNA barcode data were useful and effective for identifying N. asiatica . The barcoding gap is an important indicator of the evaluation of DNA barcode. The comparison of intra- and inter-specific genetic distance indicated that barcoding gaps based on COI sequence can be used for molecular identification at the species level. The NJ and ML trees could distinguish the specimens from other species clearly.
The integrated approaches of morphological and molecular identification provide helpful support in the monitoring and rational control of insect pests ( Bertin et al., 2010). The utility of the COI barcode system as a taxonomic tool will play a significant role in monitoring unknown species in the field. For instance, the occurrence of N. asiatica can be monitored in other wolfberry planting areas using molecular identification in the shortest time, thus facilitating the selection of suitable control methods for pest management.
The present study serves as a contribution to the development of a rapid, accurate and practical molecular approach of N. asiatica identification. This will also be helpful for identifying small organisms, plus poorly-studied and other species without obvious morphological characteristics ( Armstrong & Ball, 2005; Ball et al., 2005; Greenstone et al., 2005). Due to the use of pesticides in most of the wolfberry orchards, we could only collect samples in an organic orchard in Zhongning, Ningxia. Future work based on increasing sample sizes and geographic populations for further verifying the identified effectiveness of DNA barcodes will enable access to a convenient and efficient way to carry out species identification in practical situations.
| DNA |
Department of Natural Resources, Environment, The Arts and Sport |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |