Tachyglossus aculeatus ( Shaw, 1792 )
|
publication ID |
https://doi.org/ 10.1093/mspecies/sez012 |
|
publication LSID |
lsid:zoobank.org:pub:BF103AF5-86BC-4A71-AB86-51D11EBBF9D0 |
|
persistent identifier |
https://treatment.plazi.org/id/39132C13-C334-B37C-FF58-FECE8C33FD3A |
|
treatment provided by |
Felipe |
|
scientific name |
Tachyglossus aculeatus ( Shaw, 1792 ) |
| status |
|
Tachyglossus aculeatus ( Shaw, 1792) View in CoL
Short-beaked Echidna
Myrmecophaga aculeata Shaw, 1792 : text with pl. 109. Type locality “ New Holland [= Sydney].”
Echidna novaehollandiae Lacépède, 1799:11. Type locality “nouae Hollandiae.”
Ornithorhynchus Hystrix Home, 1802:348 . Replacement name for Myrmecophaga aculeata Shaw, 1792 .
Echidna setosa E. Geoffroy [Saint-Hilaire], 1803:126. Type locality “from the neighborhood of Adventure Bay, Bruny Island, Tasmania;” (see “Nomenclatural Notes”).
E [chidna]. breviaculeata Tiedemann, 1808:592. Type locality “la Nouvelle Hollande, au détroit Bass;” (see “Nomenclatural Notes”).
E [chidna]. longiaculeata Tiedemann, 1808:592. Replacement name for Myrmecophaga aculeata Shaw, 1792 .
Acanthonotus myrmecophagus Goldfuss, 1809:309 . Replacement name for Myrmecophaga aculeata Shaw, 1792 .
Platypus longirostra Perry, 1810: Second page of text associated with Plate 10. Type locality unknown; (see “Nomenclatural Notes”).
[ Tachyglossus View in CoL ] aculeata: Illiger, 1811:114 . First use of current name combination.
Echidna australiensis Lesson, 1827:318. Replacement name for Myrmecophaga aculeata Shaw, 1792 and Echidna setosa E. Geoffroy Saint-Hilaire, 1803.
Ornithorhynchus eracinius Mudie, 1829:180 . Type locality unknown.
Echidna australis Lesson, 1836:460. Replacement name for Myrmecophaga aculeata Shaw, 1792 .
Echidna corealis Krefft, 1872:808. Type locality “Cape York, Queensland.”
Echidna orientalis Krefft, 1872:808. Type locality “eastern Australia.”
Tachyglossus lawesii Ramsay, 1877:32 . Type locality “ Port Moresby, Papua New Guinea.”
Echidna acanthion Collett, 1884:1. Type locality “Gracemere near Rockhampton, Queensland.”
Echidna typica Thomas, 1885:338, pl. 23, fig. B. Type locality “The whole continent of Australia.”
Echidna hystrix multiaculeata Rothschild, 1905:306. Type locality “extreme south of South Australia.”
Tachyglossus aculeatus ineptus Thomas, 1906:2 . Type locality “Parker’s Range, Southern Cross;” (see “Nomenclatural Notes”).
E [chidna]. sydneiensis Kowarzik, 1909:214. Replacement name for Echidna hystrix multiaculea Rothschild, 1905 [= Tachyglossus aculeatus multiaculeata ( Rothschild, 1905) ].
Echidna hobartensis Kowarzik, 1909:215. Replacement name for Echidna setosa Geoffroy Saint-Hilaire, 1803.
CONTEXT AND CONTENT. Context as for genus. It is recognized that subspecies designations are unclear and revision is needed (Wilson and Reeder 2005; Jackson and Groves 2015); however, genetic data are not yet completed. Morphological differences that currently characterize the five subspecies are degree of hairiness, ratio between length of the second and third claw on the hind feet ( Thomas 1885), and thickness and length of the spines ( Griffiths 1978). The currently recognized subspecies (Wilson and Reeder 2005; Jackson and Groves 2015) are:
T. a. acanthion Collett, 1884:1. See above; ineptus Thomas, is a synonym.
T. a. aculeatus Shaw, 1792 :pl. 109. See above; australiensis (Lesson), australis (Lesson), corealis (Krefft), eracinius (Mudie) , hystrix (Home) , longiaculeata (Tiedemann), myrmecophagus (Goldfuss) , novaehollandiae (Lacépède), orientalis (Krefft), sydneiensis (Kowarzik), typica (Thomas) are synonyms.
T. a. lawesii Ramsay, 1877:32 . See above.
T. a. multiaculeatus Rothschild, 1905:306. See above.
T. a. setosus Geoffroy Saint-Hilaire, 1803:126. See above; breviaculeata (Tiedemann), hobartensis (Kowarzik) and longirostris (Perry) are synonyms.
NOMENCLATURAL NOTES. The holotype of Echidna setosa Geoffroy was based on the animal shot at Adventure Bay, Van Diemen’s Land (now called Tasmania) in 1790 by Lieutenant Guthrie ( Home 1802; Griffiths 1978). It appears E [chidna]. breviaculeata Tiedemann was based on the same specimen, as he notes the animal was found at Van Diemen’s land and was illustrated by Home (1802). Iredale and Troughton (1934) consider Platypus Longirostra Perry a synonym of setosus and state the source as “ Tasmania.”
After examination of Tachyglossus aculeatus from Western Australia, Griffiths (1978:62) concluded it could not be distinguished from T. aculeatus in the arid part of Queensland, New South Wales, and South Australia. He suggested the subspecies T. a. ineptus be dropped and that animals from arid and semiarid regions be referred to as T. a. acanthion.
Griffiths (1978:60) believes that Rothschild (1905) received his Tachyglossus specimens from Kangaroo Island. Because they are geographically isolated and very distinctive from all other Tachyglossus , he proposed the subspecies name not be applied to mainland animals but be restricted to Kangaroo Island Tachyglossus .
French naturalist, Baron Georges Cuvier applied the name “Echidna” to the “native porcupine” in 1797 ( Liverani 1987). The name has two possible derivations, one named after Ekhidna , a Greek goddess, who was one-half reptile and one-half mammal. Many of her offspring, including the Sphinx, bore a mixture of characteristics, like Tachyglossus itself. The other source is Echinos, which means “spiny,” the Greek word for hedgehog. Echidna as a genus name had been assigned earlier to moray eels (family Muraenidae ) in 1788 ( Fisher 1788). In 1811, the genus name Tachyglossus , meaning “swift tongue,” was applied (Iredale and Troughton 1934). Echidna remains the common English name, but in some parts of Australia it is referred to as “porky,” “porcupine,” “hedgehog,” “spiny-anteater,” and even “igniter.” Many zoological gardens in the Northern Hemisphere erroneously refer to the short-beaked echidna as short-nosed.
DIAGNOSIS
The two genera in the family Tachyglossidae are the monotypic Tachyglossus ( Mahoney 1988) and Zaglossus , represented by three extant species (Flannery and Groves 1998). The geographic range of Tachyglossus and Zaglossus overlaps only in Papua New Guinea, where the genera can be differentiated by size and other morphological features. Zaglossus obtains a length of up to 1 m and a body mass of 16 kg, although average size is about 10 kg ( Griffiths 1978, 1989). By contrast, most Tachyglossus weigh less than 5 kg and are less than 500 mm in length. Z. bruijni , the western long-beaked echnida, has a rostrum nearly twice the length of Tachyglossus , 105 and 53 mm, respectively ( Griffiths et al. 1991). Furthermore, the Zaglossus tongue is grooved on the dorsal anterior one-third and has three rows of backward-directed keratinous spines ( Griffiths 1978), features absent from Tachyglossus .
GENERAL CHARACTERS
The compact body of Tachyglossus aculeatus is dorsoventrally compressed with a domed back and flat or slightly concave ventral surface ( Fig. 1 View Fig ). The average length of an adult T. aculeatus is 400–450 mm ( Griffiths 1978). The back and sides are covered with spines of varying sizes. Spines along the dorsal midline converge, forming a crisscross pattern along the back and two semicircular rosettes of spines cover the short, bald tail nub ( Fig. 2 View Fig ). Interspersed among the spines is fine to thick hair. Pelage length and density differ among subspecies. Pelage color of the two island subspecies, T. a. multiaculeatus and T. a. setosus, varies from light straw-colored to dark brown. Mainland subspecies are generally uniformly dark in color. Albinism occurs in all subspecies. The legs and venter of T. aculeatus are free from spines but covered with hairs. Both front and back limbs are short, stout, and pentadactylous. The front digits bear strong spatulate claws, the three in the middle being longer than the others. The hind limbs are rotated backwards.
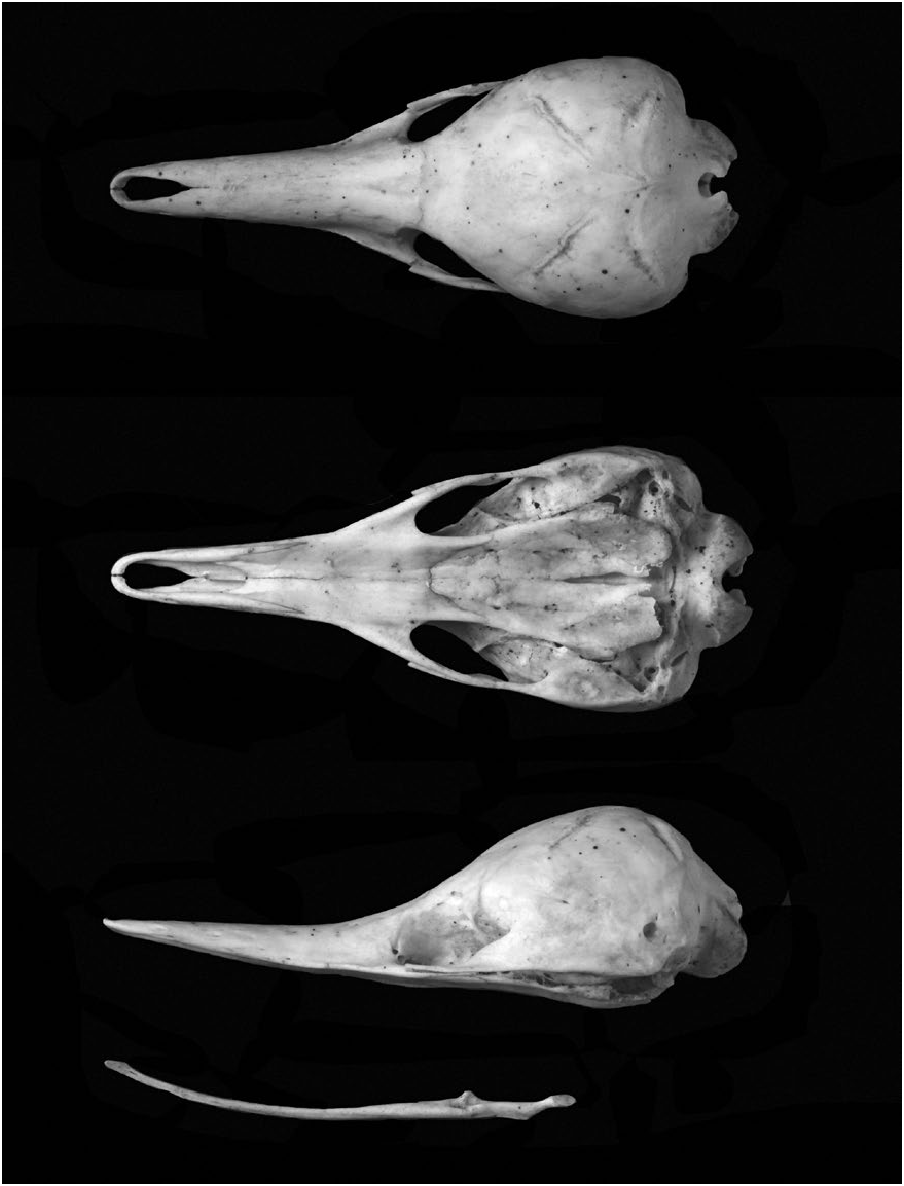
Due to the spiny coat, T. aculeatus lacks a defined neck. The anterior portion of the skull is elongated into a beak-like structure ( Fig. 3 View Fig ), which is covered with a soft, hairless skin. The small (ca. 9 mm diameter— Griffiths 1989) forwardly directed eyes are located almost at the base of the snout ( Fig. 4 View Fig ). The nares are located on the dorsal apex of the snout and the tiny mouth opening (ca. 5 mm) is on the underside. The mouth only opens the width of the tongue, which can protrude up to 180 mm beyond the end of the beak ( Griffiths 1989). The ears usually lack external pinnae and appear as vertical slits on the side of the head. Small spines and hairs often obscure these openings from view.
Calculating a mean body mass for T. aculeatus is not practical because the annual mean minimum and mean maximum body mass of sexually active animals varies from year to year. Males and females show seasonal fluctuations of 37% and 25%, respectively (Rismiller and McKelvey 2000; Nicol and Morrow 2012). Minimum and maximum body mass for sexually active T. a. multiaculeatus and T. a. setosus males and females is similar, 2.8–4.4 kg and 2.5–5.0 kg, respectively (Rismiller and McKelvey 2000; Nicol and Morrow 2012). The heaviest documented T. aculeatus is a mature female from the Snowy Mountains with a body mass of 6.95 kg in March and 5.4 kg in December of the same year ( Grigg et al. 1992).
Tachyglossus aculeatus shows no consistent sexual dimorphism and sex of an individual generally cannot be visually determined because there are no secondary sex characteristics. However, during the breeding season some T. aculeatus males have a pronounced penis bulge ( Morrow et al. 2009).
Monotreme means “one hole,” referring to the single orifice, the cloaca, for the passage of feces, urine, and reproductive products. There is no way to determine the age of adult animals, but juveniles up to 4 years can be identified by the presence of a sheath-covered spur on the inside ankle of both hind feet ( Rismiller 1999; Rismiller and McKelvey 2000). After the sheath is lost, the spurs change in form or are totally lost.
The pectoral (shoulder) girdle of T. aculeatus has retained a therapsid pattern reminiscent of a crocodile or lizard, consisting of two scapulas, two coracoids, and two clavicles along with two bones found in no other mammal, one T-shaped interclavicle and two epicoracoids ( Griffiths 1968). The vertebral formula of T. aculeatus is reminiscent of the Marsupialia, with 7 C, 15 T, 3 L, 4 S (two sacral and two sacrococcygeal), total 29 ( Gregory 1947). The pelvic girdle has the three usual mammalian bones, dorsal trihedral ilium, ventro-posterior ischium, and ventro-anterior pubis; however, at the anterior margin of each pubis is an epipubic or marsupial bone that is articulated and extends the entire length of the pubis ( Griffiths 1968).
DISTRIBUTION
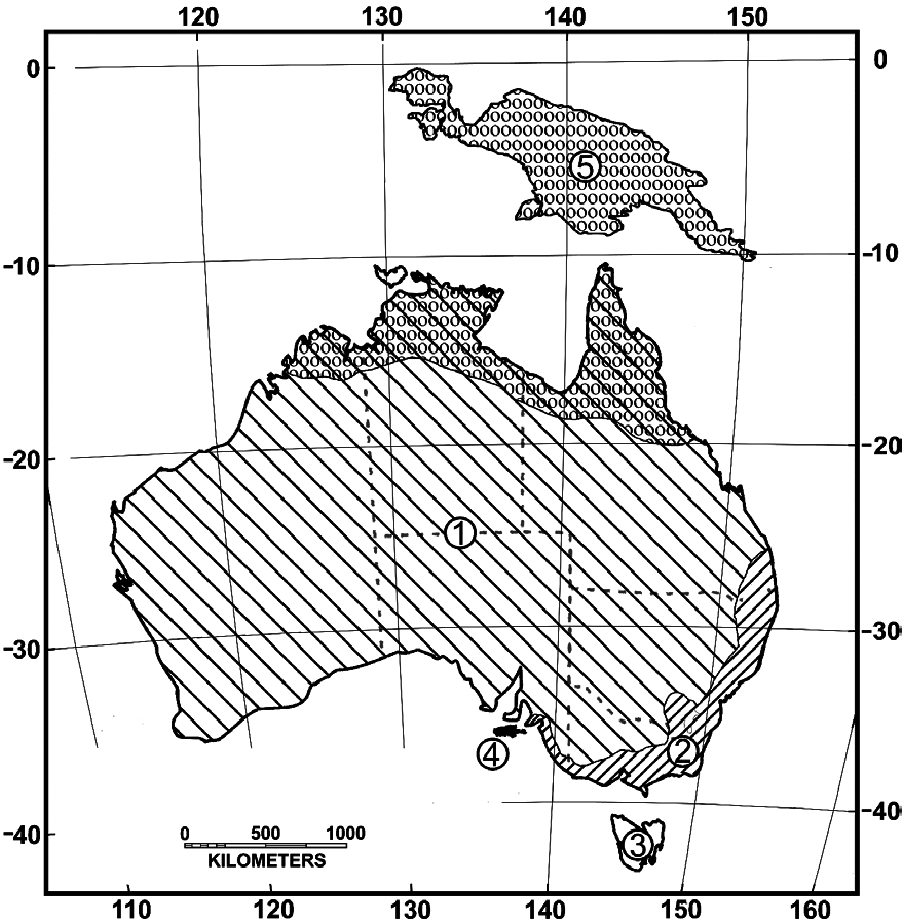
Tachyglossus aculeatus is Australia’s most common native mammal, by distribution ( Archer 1982). Its geographic range extends from Papua New Guinea (1°S) to the southernmost part of Tasmania (43°S) and across the Australian continent, from 112°E to 154°E ( Fig. 5 View Fig ). Elevation range is from sea level to 1,800 m at Mount Kosciuszko National Park, New South Wales ( Grigg et al. 1989). T. aculeatus inhabits the Central Highlands of Papua New Guinea up to 1,600 m ( Augee et al. 2006).
Tachyglossus aculeatus multiaculeatus is found solely on Kangaroo Island and T. a. setosus is confined to Tasmania and the Bass Strait islands. T. a. lawesii was first described from Papua New Guinea; however, Griffiths (1978) suggests it also occurs in the northern tropical areas of Australia. T. a. acanthion occurs in Western Australia, Northern Territory, and throughout the arid regions of Queensland and South Australia. The range of T. a. aculeatus includes southern Queensland, eastern New South Wales, and Victoria through to the Fleurieu Peninsula in South Australia ( Griffiths 1978; Fig. 5 View Fig ). A zone of hybridization between T. a. acanthion and T. a. aculeatus in a roughly rectangular area with Port Macquarie, New South Wales, Forbes, New South Wales, Tara, Queensland, and Brisbane, Queensland at the corners has been speculated ( Griffiths 1978).
FOSSIL RECORD
Due to the scarcity of monotreme fossils, especially those of Tachyglossidae , divergence times and relationships within Monotremata are still equivocal ( Musser 2013). The few recorded Tachyglossus aculeatus fossils are from various parts of Australia. The earliest known fossil tachyglossid is Zaglossus robusta (= Megalibgwilia ), described from bones found in the Canadian alluvial gold mine at Gulgong, New South Wales ( Dun 1895; Murray 1978). These fossils date to the mid-Miocene ( Woodburne et al. 1985), or about 12–15 million years ago. Remains of Tachyglossus from Pleistocene sediment (<100,000 years old) have been found in Mammoth Cave, Western Australia ( Merrilees 1979), and the Naracoorte Caves of South Australia ( Pledge 1980; Wells and Pledge 1983). Tachyglossus fossils from Lake Tandou, New South Wales are believed to represent the refuse from Aboriginal meals dating from late in the Quaternary ( Merrilees 1973).
FORM AND FUNCTION
Form. —The skull of Tachyglossus aculeatus is smooth with elongate maxilla and premaxilla forming the upper jaw. The lower jaw is formed by the right and left dentaries, which are long and reduced to splinters. Rotation of the dentaries about their own axes causes the mouth to open and close ( Murray 1981). Although teeth are absent in Tachyglossus , prey are masticated by the grinding action between the posterior horny pad of the tongue and the keratinous rows of backward directed spines on the palate ( Griffiths 1989).
The surface of the long vermiform tongue is sticky due to mucus secreted by paired sublingual salivary glands ( Lew et al. 1975). The rapid protrusion and retraction of the tongue, about 100 actions/min (Doran and Baggett 1970), is achieved by contraction of uniquely arranged circular muscles and three pairs of longitudinal muscles ( Griffiths 1968, 1978).
The stomach of T. aculeatus consists of a rough horny skin made of cornified stratified epithelium and without secretory glands ( Griffiths 1965a). Peristaltic movements of the stomach help grind dirt and food, breaking down hard invertebrate exoskeletons. Digestion takes place primarily in the 3.4-m long small intestine ( Home 1802; Bridgewater et al. 1962). Movement through the bowel is slow, up to 2 days ( Griffiths 1965a), allowing thorough digestion of soft invertebrate parts and reabsorption of water. Dirt and undigested exoskeletal material are eliminated through the cloaca. The well-formed scat has a characteristic cylindrical shape with blunt ends. Renal anatomy of T. aculeatus is typically mammalian except for the presence of a dwarf nephron, the Zwerg-kanaelchen of Zarnik ( Zarnik 1910), which is found in the kidney of many reptiles. Ureters from the kidneys discharge into an unpaired urogenital sinus, opposite the neck of the bladder. Contraction of the bladder causes the urine to pass into the sinus and out of the body through the cloaca. This system applies to both males and females ( Zarnik 1910).
The central nervous system of T. aculeatus is considered remarkable because of the short length of the spinal cord, which terminates at the seventh thoracic vertebra compared to between the first and second lumbar vertebrae in humans, and the large, unusual brain ( Hassiotis et al. 2004; Ashwell 2013a). The position and relationship of visual, auditory, and sensory areas are unlike those in Eutherian mammals ( Ashwell 2013b). Nearly onehalf of the brain sensory area is allocated to tactile organs such as the snout and tongue ( Abbie 1934). The brain shows a high degree of cortical folding or gyrification, which is considered the hallmark of more neurologically advanced mammals ( Hassiotis et al. 2004; Ashwell 2013b). The prefrontal cortex, associated with complex cognition, retrieval of long-term memory, and decision-making, accounts for 50% of the isocortex in T. aculeatus but only 29% in humans (Pirlot and Nelson 1978; Ashwell 2013b). When T. aculeatus was tested using maze running and positional, visual, and tactile experiments, it learned quickly, improved its performance with practice, and showed evidence of storing, classifying, and integrating information (Buchmann and Rhodes 1978). In more advanced cognitive trails, one captive T. a. aculeatus demonstrated abilities to categorize stimuli on a same and different concept and transfer that discrimination to a novel concept (Russell and Burke 2016).
The spines of T. aculeatus are modified hairs (Spencer and Sweet 1899; Hausman 1920). There is no annual moult, but spines are randomly shed and replaced. The base of each spine is surrounded by a bundle of muscles and its root is anchored in the panniculus carnosus, a large muscle mass that extends around the trunk ( Griffiths 1978). This arrangement allows control of individual spines when the animal is climbing, foraging, preening, or avoiding a threat. The panniculus carnosus, thicker on the dorsal and thinner on the ventral side, is responsible for the ability of Tachyglossus to roll itself into a ball ( Griffiths 1978). It is impossible to separate the true skin (epidermis and dermis) plus spines and hair from the panniculus carnosus. This massive organ accounts for one-third to one-half of body mass (P. D. Rismiller, in litt.). Unlike other mammals, the skin of T. aculeatus contains very few sweat glands ( Pinkus 1906).
Tachyglossus aculeatus has no permanent pouch but both sexes can contract abdominal muscles, giving the appearance of a shallow pouch. Females develop a distinct pseudopouch only after mating, when the mammae swell and form thick tumescent lips ( Griffiths 1968). T. aculeatus has no nipples, but two small (ca. 5 mm diameter) hairy patches, the milk areolae, are located at the anterolateral margins of the pouch area. Each milk patch consists of 100–150 separate lobules, opening into an invagination of the skin with a hair follicle ( Gegenbaur 1886). These specialized hairs are associated only with the ducts of the mammary glands, where the young suckles ( Bresslau 1920).
Tachyglossus aculeatus has remarkable strength due to its unusual anatomy. Well-developed muscles from the short, stout limbs attach to the solid pectoral girdle and form a mechanical arrangement called a third-order lever ( Augee et al. 2006). The ratio of large muscle mass to bone length accounts for its effective power. The strong claws on the forepaws are effective digging tools for foraging. The beak also has the mechanical advantages of a lever. Shaped like a double wedge, the effort exerted by the beak equals six times its full length ( Augee et al. 2006). T. aculeatus uses its beak for splitting firm soil, flipping rocks, or rupturing the bodies of invertebrate prey that are too large to ingest whole ( Rismiller 1999; Augee et al. 2006).
Due to the outward rotation of the tibia and fibula and the horizontal femur, the hind feet and claws of T. aculeatus are directed backward. The first claw on the outside of the hind foot is the shortest, the second is usually the longest, and depending on subspecies, the third is either shorter, of equal length or longer than the second. Claws on the fourth and fifth toes are notably short. Most subadult T. aculeatus have a sheath-covered spur on the inside ankle of both hind feet ( Griffiths 1989; Rismiller 1999). Spurs in adults are hollow, perforated, and connected via a duct to the crural gland located just below the knee. Unlike the spur in male Ornithorhynchus , which conducts venom ( Grant 1995), the structure in Tachyglossus is not venomous ( Krause 2010). In both Ornithorhynchus and Tachyglossus , the crural glands exhibit seasonal cycles, being most active during the breeding season ( Wong et al. 2013; Morrow et al. 2016). Similar seasonal cycles are found in plasma testosterone ( Nicol et al. 2005) and testes size ( Morrow et al. 2016).
Owing to the structure and orientation of the hind limbs, the gait of T. aculeatus can be described as rolling. A cineradiographic record of movements shows the abduction of the humerus, that is the excursion arc from the parasagittal plane, to be about 90°. This arc of movement is far greater than in cursorial mammals. The humerus shows no evidence of a pattern of elevation and depression that could be related to the discrete phases of movements seen in other noncursorial mammals ( Jenkins 1970). Due to their similar bone structure, locomotion in Zaglossus and Tachyglossus is thought to be similar despite significant differences in size and mass ( Pridmore 1985).
Function. —Despite small external eyes, Tachyglossus aculeatus has good visual learning abilities ( Gates 1973; Griffiths 1978). The number of fibers in the optic nerve (ca. 15,000) indicates a reasonable degree of visual discrimination ( Gates 1973, 1978). The arrangement of the ear ossicles implies that auditory information may be received via mechanoreceptors in the beak ( Iggo et al. 1985) as well as via the large cartilaginous ear trumpets (Aitken and Johnstone 1972).
Organization of the olfactory system in T. aculeatus follows the usual mammalian pattern, but the area of olfactory epithelium, which is carried on vertical bony septa called ethmoturbinals, is large ( Ashwell 2013d). Tachyglossus has 13 times more olfactory nerve fibers than Ornithorhynchus . The trigeminal nerve, from the skin of the beak, ends in the thalamus, linking the tactile sensibility of the beak with smell ( Abbie 1934; Griffiths 1978; Augee et al. 2006; Ashwell 2013b).
The circulatory system of T. aculeatus is similar to that of other mammals, consisting of two completely separate circulations with the systemic aorta derived from the left fourth arch ( Hochstetter 1896). T. aculeatus has a diaphragm and asymmetric lungs, with the right side making up about 65% of lung volume ( Perry et al. 2000). T. aculeatus tolerates high levels (up to 12%) of CO 2 ( Augee et al. 1971), making it physiologically well-suited for burrowing and remaining underground for extended periods.
Tachyglossus aculeatus does not sweat or pant to help control body temperature ( Augee 1976). However, production of nasal mucus at high temperatures cools the nasal blood sinus and enhances evaporative cooling in T. a. acanthion ( Barker et al. 2016). Active body temperature of T. aculeatus is 31–33°C ( Wardlaw 1915; Dawson et al. 1979). The early speculations that T. aculeatus uses torpor ( Miklouho-Maclay 1883) were confirmed when those living above the snow line in the Australian Alps of New South Wales and in Tasmania exhibited patterns of body temperature similar to typical mammalian hibernators ( Grigg et al. 1989; Nicol and Andersen 2000). During hibernation of up to 7 months, body temperatures drop to 0.5°C above ground temperature with periodic arousals to euthermia. T. a. setosus in hibernation can lower its respiration rate to 1 breath/min and has periods of apnea lasting up to 2 h ( Nicol et al. 1992; Nicol and Andersen 2003). Use of torpor by T. aculeatus living in more temperate climates is very individualistic, much shorter (maximum 10 days—Rismiller and McKelvey 1996), and not seasonally restricted ( Rismiller 1999). Some individuals use torpor during the hot, dry periods of summer, as well as at other times of the year. Torpor has also been implicated as a significant survival strategy for T. aculeatus ( Nowack et al. 2016) .
Metabolic rate, heart rate, and respiration rate vary from 0.03 ml O 2 /g/h, 7 beats/min, and 1 breath/3 min in a torpid T. aculeatus with a body temperature of 5°C to 0.5 ml O 2 /g/h, 70 beats/min, and 6 breaths/min in an active 33°C animal (Augee and Ealey 1968; Nicol et al. 1992; Nicol and Andersen 2003). Field energetic studies show both lactating and nonlactating T. aculeatus have similar low metabolic rates and water influx rates ( Green et al. 1992a, 1992b; Schmid et al. 2003). These surprising physiological responses are consistent with other energysaving strategies used by T. aculeatus , such as daily or longer bouts of torpor.
The efficient renal system of T. aculeatus is able to produce hypertonic urine with concentrations as great as 2,300 mOsm (Bentley and Schmidt-Nielsen 1967). The ability to decrease urine volume and conserve water is crucial to survival in arid environments ( Bentley 2002).
ONTOGENY AND REPRODUCTION
Ontogeny. — Tachyglossus aculeatus is oviparous ( Caldwell 1884, 1887; Haacke 1884). The egg is telolecithal with discoidal meroblastic cleavage like that of reptiles ( Caldwell 1887). The first cleavage divides the germinal disc into disproportionate areas. The four-cell stage consists of two large and two smaller blastomeres lying on top of the yolk. In utero, blastomere differentiation is similar to that of reptiles ( Caldwell 1887). At the time of egg-laying the embryo has 19 pairs of somites (Hill and Gatenby 1926). Normal gestation is 20–24 days, depending on subspecies (Rismiller and McKelvey 2000; Nicol and Morrow 2012).
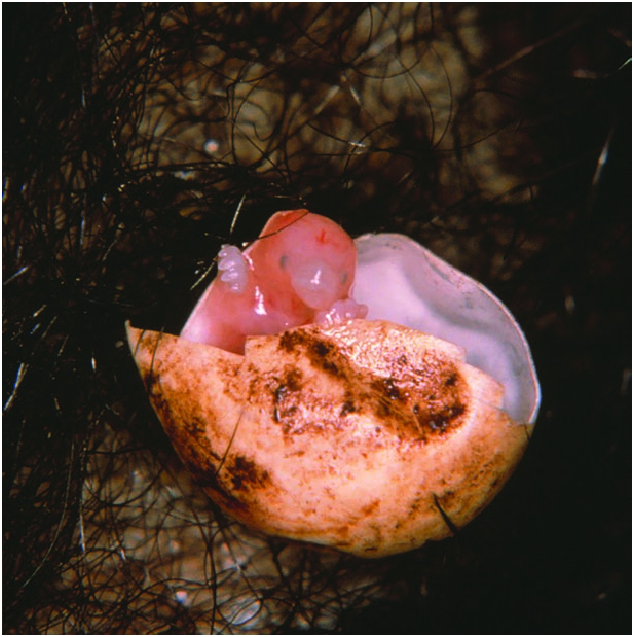
Incubation in the pouch lasts about 10.5 days ( Griffiths et al. 1969). There are 14 growth stages of development, termed 40–53, during incubation ( Semon 1894b, 1894c). More recently, the Standard Event System (SES) has been used to describe the sequence of embryonic development in T. aculeatus (Werneburg and Sánchez-Villagra 2011) . At stage 40 the embryo has about 39 somites, lobate forelimbs, buds for hind limbs, a tail, and a total length of 7 mm. An egg tooth appears at the anterior end of the snout at stage 43. Before hatching (stage 45) the egg tooth acquires a sharp point which is overlain with enamel ( Owen 1865; Seydel 1899). The egg tooth, used to rupture the egg shell, is lost within 48 h after hatching (Rismiller and McKelvey 2000). Fluid from the egg spills into the pouch and the young, known as a puggle, grasps the hairs on the mother’s belly and pulls itself out ( Fig. 6 View Fig ).
The hatchling, with a mean body mass of 0.303 g ± 0.011 SE (range 0.231 –0.371, n = 11—Rismiller and McKelvey 2003), fills only about one-third the volume of the egg. Five puggles under the age of 24 h averaged 22.2–24.3 mm from snout to tip of tail (Rismiller and McKelvey 2000). At hatching, the body is translucent, with colored bands indicating locations of vital organs, and within hours of hatching, ingested milk is visible through the body walls. It is possible that newly hatched T. aculeatus respires through its skin, as has been documented for 380 mg newborn tammar wallaby Macropus eugenii ( MacFarlane et al. 2002) .
The presence of well-developed sense cells suggests olfaction plays a role in finding the milk patch ( Griffiths et al. 1969; Griffiths 1978). T. aculeatus has no teats to dispense milk, but the histology of the milk patch resembles that of the human nipple ( Griffiths 1965b). Milk passes to the exterior and the young suckle at the base of specialized mammary hairs ( Griffiths 1978). The composition of milk changes during the growth of the young, being dilute initially, but becoming more concentrated. Early lactation milk is about 12% solids, of which 1.25% is fat and 7.85% protein; later, mature milk has 48.9% solids, of which 31% is fat and 12.4% protein ( Griffiths et al. 1984). Unlike milk of eutherians, T. aculeatus milk contains little free lactose. The principal carbohydrates in milk of T. a. multiaculatus and T. a. aculeatus are fucosyllactose and sialyllactose (Messer and Kerry 1973). Additional oligosaccharides are present in the milk of T. a. setosus ( Oftedal et al. 2014). Milk transcriptome data show gene expressions similar to other mammals as well as a number of monotreme-specific components, including the monotreme lactation protein ( Enjapoori et al. 2014).
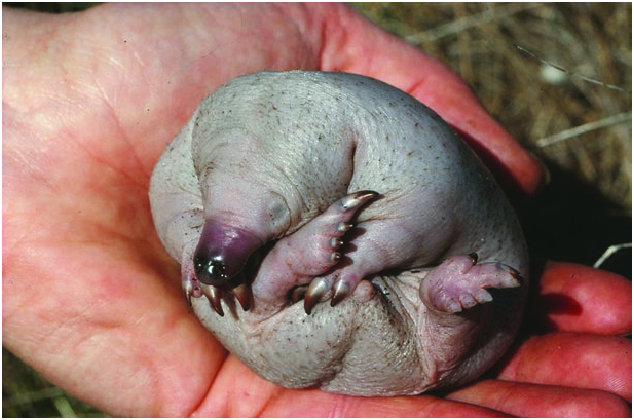
Patterns of maternal care vary among some subspecies. The young of T. a. multiaculeatus is carried in the mother’s pouch from hatching until about 50 days old ( Fig. 7 View Fig ), during which she is out actively foraging. During this time body mass of the suckling increases from about 300 mg to 180–250 g and skin color changes from pink to gray as hair and spines start to grow ( Griffiths 1978; Rismiller 1999; Rismiller and McKelvey 2000). At this stage the female places the young in a self-dug nursery burrow, back-filling the entrance each time she leaves to keep the young cool and to protect it from predators. For the next 5 months the female returns once every 5–6 days to suckle the young, until weaning at 204–210 days ( Griffiths 1978; Griffiths et al. 1988; Rismiller and McKelvey 2000). Body mass at weaning is 800–1,500 g, being heavier in larger mothers (Rismiller and McKelvey 2003). One field observation for T. a. acanthion documented a young that was weaned between 195–200 days at 1.7 kg ( Abensperg-Traun 1990).
In contrast, T. a. setosus females remain in a plugged burrow during egg-laying, incubation, and hatching, only emerging after 37–51 days to feed for the first time without the young in the pouch. During confinement the female loses a mean of 1.2 ± 0.005 g /day while the young increases body mass from <400 mg to 200 g (Morrow and Nicol 2013). Later, females return to the burrow at varying intervals to suckle the young. Two young weaned at 139 and 152 days had body masses of 1.5 and 2.2 kg, respectively ( Morrow et al. 2009; Morrow and Nicol 2013). The relatively short lactation period and rapid growth of T. a. setotus young may be a geographic adaptation for survival of a subspecies at the southernmost extent of its range (Morrow and Nicol 2013). A sexually mature female can reproduce in consecutive years (Rismiller and McKelvey 2000; Morrow and Nicol 2013), but most females have one young per breeding season every 2–3 years. One T. a. aculetus (Beard and Grigg 2000) and one T. a. setosus (Harris and Nicol 2014) bred a second time in the same season after losing a young.
Reproduction. —Courtship and breeding of Tachyglossus aculeatus usually occur between June and September throughout its range ( Griffiths 1978; Rismiller and Seymour 1991; Nicol and Morrow 2012). In some years and in different parts of Australia, breeding may be advanced or delayed by several weeks ( Beard et al. 1992; Rismiller and McKelvey 2000; Nicol and Morrow 2012). It is still unclear what cues or physiological factors induce ovulation in T. aculeatus but the stimulus simply may be the presence of a male with a female over multiple days ( Morrow et al. 2016).
As seasonal breeders, testes size of T. aculeatus varies from 1–3 g /kg body weight in the nonbreeding season, up to 15 g /kg body mass at peak breeding ( Griffiths 1978; Augee et al. 2006; Morrow et al. 2016). The paired Cowper’s glands are welldeveloped ( Home 1802), but adult males have no prostate gland or seminal vesicles, or scrotum for that matter; the paired ovoid testes are posterior to the kidneys. Seminiferous tubules at the anterior end of the testes join seven ductuli efferentes passing to an epididymis. This differentiates into a large caput and small cauda connected by a vas deferens, which conveys the sperm to the penis ( Jones et al. 2004). The penis, used solely for the passage of spermatozoa, is about 70 mm long and has a bifid glans with two openings on each glans ( Griffiths 1978). T. aculeatus has an unusual form of one-sided ejaculation that produces highly motile sperm bundles of up to 100 spermatozoa (Djakiew and Jones 1981; Johnston et al. 2007). Except during copulation when the penis is engorged and forced out through the cloacal sphincter, it lies internally within a preputial sac that is ventral to the cloaca. Whereas only single copulations with intromissions of 30–180 min are recorded for T. a. multiaculeatus ( Rismiller 1992; Rismiller and McKelvey 2000), T. a. setosus may have multiple copulations of varying duration ( Morrow et al. 2009).
The paired ovaries are attached to the anteroventral faces of the kidneys by a fold of peritoneum. Unlike Ornithorhynchus , where only the left ovary and oviduct function, both ovaries in T. aculeatus are functional ( Griffiths 1978). The anterior portions of the thin-walled infundibular funnels which encase the ovaries make up the fallopian tubes; the posterior ends are differentiated to form the uteri, which open separately into the urogenital sinus. At the height of the breeding season, each ovary has clusters of ripe follicles up to 5 mm in diameter ( Hughes 1984). After the first maturation spindle of meiosis appears, the ovum is shed into the infundibulum and the egg passes to the fallopian tube, where fertilization occurs (C. J. Hill 1933; Flynn and Hill 1939). The fertilized egg is first covered with a mucoid coat ( Griffiths 1978), followed by two layers of egg shell ( Griffiths 1989). At the final stage of intrauterine development, when the embryo has 19 pairs of somites, the egg is about 15 cm in diameter ( Luckett 1977; Griffiths 1978), and the egg shell has its definitive form consisting of three layers (J. P. Hill 1933).
The normal period between mating and egg-laying (gestation) in T. a. setosus is 20–24 days, but varies between 20–80 days depending on the length of postfertilization hibernation (Nicol and Morrow 2012; Morrow and Nicol 2013). Gestation determined for a population of T. a. multiaculeatus monitored over 8 years was 23 days ± 12 h SD (Rismiller and McKelvey 2000). The egg is laid directly into the pouch by the female curling her body and extruding the cloaca over the pouch opening (Rismiller and McKelvey 2000).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Tachyglossus aculeatus ( Shaw, 1792 )
| Rismiller, Peggy D & Grutzner, Frank 2019 |
Tachyglossus aculeatus ineptus
| THOMAS, O. 1906: 2 |
hystrix
| ROTHSCHILD, W. 1905: 306 |
Tachyglossus lawesii
| RAMSAY, E. P. 1877: 32 |
Ornithorhynchus eracinius
| MUDIE, R. 1829: 180 |
Tachyglossus
| ILLIGER, J. K. W. 1811: 114 |
Acanthonotus myrmecophagus
| GOLDFUSS, G. A. 1809: 309 |
Ornithorhynchus Hystrix
| HOME, E. 1802: 348 |