Drusus chelchamaensis Ibrahimi & Mohammadi, 2024
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5406.3.4 |
|
publication LSID |
lsid:zoobank.org:pub:6D969607-6BC3-467A-9D91-C08B6D6F9441 |
|
DOI |
https://doi.org/10.5281/zenodo.10628080 |
|
persistent identifier |
https://treatment.plazi.org/id/377C0471-FF8A-345D-FF33-FB40E3ADF816 |
|
treatment provided by |
Plazi |
|
scientific name |
Drusus chelchamaensis Ibrahimi & Mohammadi |
| status |
sp. nov. |
Drusus chelchamaensis Ibrahimi & Mohammadi sp. nov.
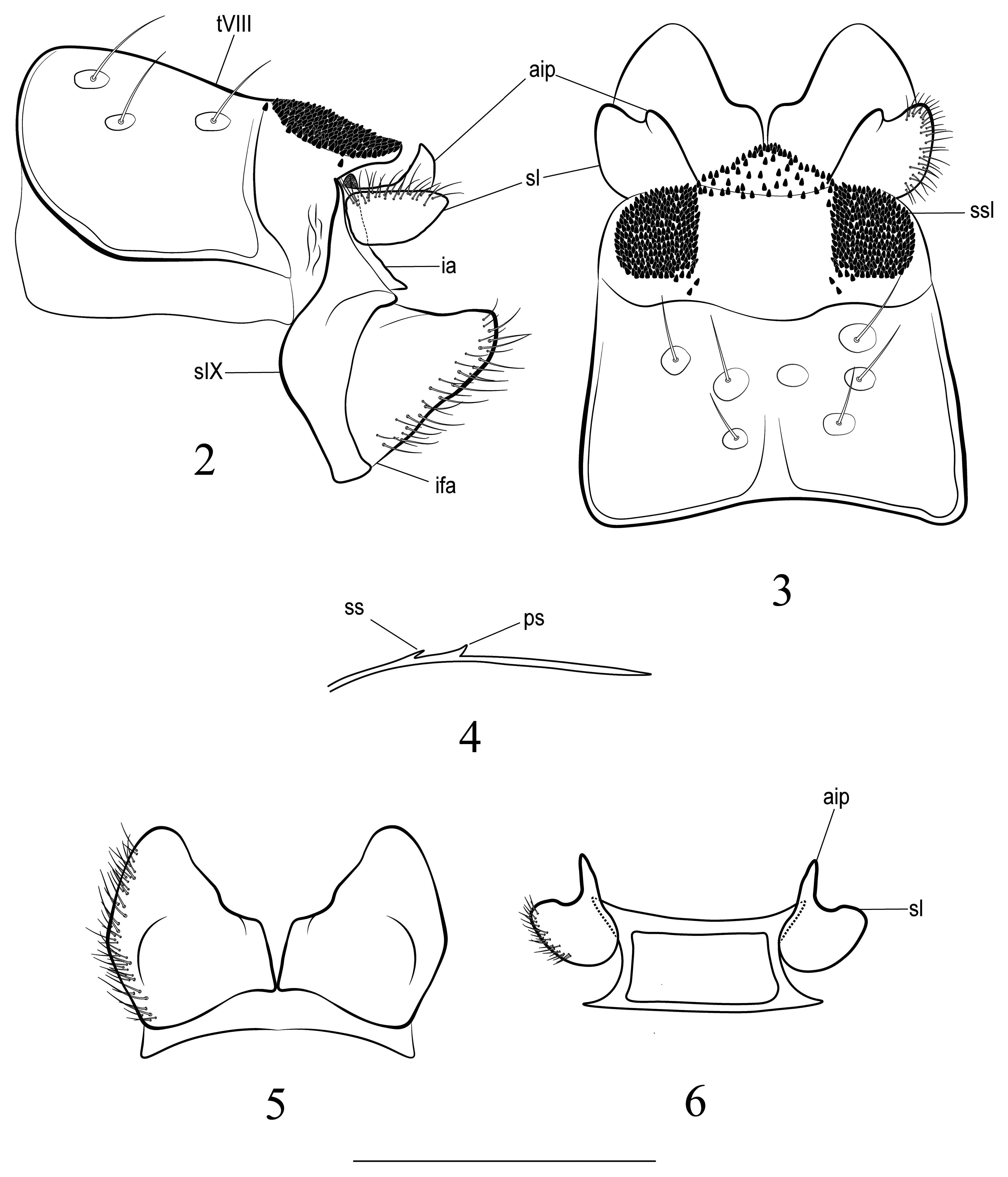
( Figures 2–5 View FIGURES 2–6 )
Type material. Holotype (male): Iran, Kurdistan Province, Zagros Mountain ranges , Chel Chama Mountain , Kani Chaow Rash spring (meaning Black Eye Spring ), 35.8387˚N, 46.5224˚E, 2816 m above sea level, 24.vii.2021, leg. H. Ibrahimi, H. Mohammadi, E. Karimian, and E. Ghaderi. Paratypes (2 males): with the same label data as the holotype .
Distribution. Iran, Kurdistan province, Chel Chama (Chehel Cheshmeh).
Diagnosis. The male of the new species is most similar to that of Drusus bayburtii Çakin 1983 (known from many places in Antalia, Turkey), D. ketes Oláh & Mey 2017 (in Oláh et al. 2017) (known from the Hamedan Province of Iran), and D. kazanciae Çakin 1983 (known from the Hakkari Province, Turkey) and differs mainly in exhibiting (1) elongate, ovate, setose lobes of superior appendages each with a triangular, blunt apex in lateral view; (2) robust, round setose lobes of superior appendages in caudal view; (3) low, robust asetose inner processes of the superior appendages in lateral view; (4) high, tapering asetose inner processes in caudal view; (5) a very long terminal part of each paramere in lateral view, 2 to 3 times longer than in D. bayburtii and 1.2 to 1.3 times longer than in D. ketes , less inflated than in both species; (6) a robust and very high principal spine of each paramere and a considerably high, single secondary spine; 7) segment IX with narrow acuminate dorsal portion and longer ventral portion, laterally even longer and convex in lateral view; (8) the spinate area of tergum VIII consisting of two nearly semicircular lobes with straight anterior bases and mesal margins, connected posteriorly by a band of scarce spines; (9) moderately long inferior appendages each with a median ventromesal incision in ventral view.
The D. bayburtii male has (1) subquadratic setose lobes of the superior appendages, each with an arcuate apex in lateral view; (2) slender setose lobes of the superior appendages each with a tapering apex in caudal view; (3) high, slender asetose inner processes of the superior appendages in lateral view; (4) high asetose inner processes with pointed apices in caudal view; (5) a short and inflated terminal part of each paramere; (6) a delicate principle spine on each paramere and 2 or 3 secondary spines; (7) segment IX with an acuminate dorsal portion, ventrally and laterally almost equally long in lateral view; (8) the spinate area of tergum VIII consisting of two oval lobes, with round anterior bases and connected posteriorly by a band of scarce spines; and (9) moderately long inferior appendages each with a median ventromesal incision in ventral view.
The D. ketes male has (1) subquadratic setose lobes of the superior appendages, each with a longer and round dorsal half in lateral view; (2) robust, round setose lobes of superior appendages in caudal view; (3) high, slender asetose inner processes of the superior appendages in lateral view; (4) low, robust asetose inner processes in caudal view; (5) a medium-long terminal part of each paramere; (6) a long principal spine on each paramere and a small single secondary spine; (7) segment IX with acuminate dorsal portion, ventrally and laterally almost equally long in lateral view; (8) the spinate area of tergum VIII composed by two circular lobes with round anterior bases, connected posteriorly by a band of scarce spines; (9) moderately long inferior appendages each with a straight ventromesal margin in ventral view.
The D. kazanciae male has (1) low, elongate, subrectangular setose lobes of the superior appendages, each with a sharply angled triangular apex in lateral view; (2) slender setose lobes of the superior appendages with tapering apices in caudal view; (3) high and long asetose inner processes of the superior appendages in lateral view; (4) low asetose inner processes, each with a very long base and spine-like apex in caudal view; (5) segment IX dorsally and ventrally tapering, almost equally long throughout the remaining height in lateral view; (6) the spinate area of tergum VIII composed by two semicircular lobes with rounded anterior bases, connected posteriorly by a band of scarce spines, more dense at the posterior apex; (7) long inferior appendages each with concave ventral and dorsal margins.
Description. General appearance. Habitus brownish yellow, sclerites and tergites light brown with few darker patches in tergites; cephalic and thoracic setal areas pale; cephalic, thoracic, and abdominal setation blonde; legs brown to fawn, with darker areas; wings yellowish-brown, translucent, with thin blonde setae. Male maxillary palps each 3-segmented. Length of each forewing 12.2 mm (holotype), 12.0– 12.5 mm (n = 3). Spur formula 1,3,3.
Male genitalia. Tergite VIII light brown ( Fig. 3 View FIGURES 2–6 ), with darker patches laterally below spinate areas in lateral view, pair of posterior spinate areas nearly semicircular in dorsal view, slightly incised anteromesally, anterior and mesal edges nearly straight, lateral-distal edge rounded and tapering posteromesally, with lighter areas around alveoli; lobes connected posteriorly by triangular and apically blunt band of scattered spines protruding broadly beyond semicircular lobes. Segment IX ( Fig. 2 View FIGURES 2–6 ) with narrow acuminate dorsal portion and longer ventral portion, laterally even longer and irregularly convex anteriorly, acute posteriorly in lateral view. Superior appendages bipartite ( Figs 2, 3, 6 View FIGURES 2–6 ), each consisting of setose lateral lobe and asetose dorsomesal process; setose lobe elongate, ovate, tapering to blunt apex, covered with fine setae of medium length dorsolaterally, ventral margin convex and dorsal one mostly straight in lateral view; asetose inner process short and broad with acuminate apex, originating from base of setose lobe in lateral view; setose lobe broadly connected with the asetose inner process, diverging only apically in dorsal view. Intermediate appendages ( Fig. 2 View FIGURES 2–6 ) much shorter than superior appendages, hidden beneath tergum IX in dorsal view and mostly hidden by superior appendages and segment IX in lateral view, apicodorsally rounded and black, caudoventrally triangular in lateral view. Inferior appendages ( Figs 2, 5 View FIGURES 2–6 ) subtriangular in lateral and ventral views, each twice long as longest part of segment IX in lateral aspect, with wide base and tapering apex, dorsal margin slightly concave, ventral margin slightly concave in lateral view; attached only basally, outer margin convex and slightly angled at midlength, inner margin straight basally, with median ventromesal incision, and sinuate distally in ventral view. Phallic apparatus consisting of simple long aedeagus and pair of parameres; parameres ( Fig. 4 View FIGURES 2–6 ) slender, each with principal spine long and triangular, secondary spine single, slightly shorter, and triangular, terminal part long and only slightly inflated before acute apex.
Female, larva, pupa. Unknown.
Etymology. Species epithet chelchamaensis , coined from ‘Chel Chama,’ referring to the name of the mountain where the type locality is located and, in the Kurdish language, meaning ‘forty springs.’
Habitat. Males of Drusus chelchamaensis sp. nov. were found around the spring area of an open spring and a few meters downstream. The spring and the stream were completely surrounded by dense riparian vegetation for more than 30 m, without signs of human activities. The substrate consisted of gravel, cobble, pebbles, and large stones; stream width was 0.5 to 0.9 meters, stream depth was 30–40 cm. During August, when the specimens of the new species were sampled, most of the stream, except the spring source, was covered by vegetation and with flowing water visible only at a few segments. The following physico-chemical parameters were observed at the sampling site on 24.vii.2021: water temperature 2°C, electrical conductivity 57 µS/cm, and dissolved oxygen 8.59 mg /l.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |