Tyrannosaurus rex, Osborn, 1905
|
publication ID |
https://doi.org/10.1038/srep23099 |
|
DOI |
https://doi.org/10.5281/zenodo.5205205 |
|
persistent identifier |
https://treatment.plazi.org/id/03D087F0-FF83-FFEC-FE41-FCF3461FFB35 |
|
treatment provided by |
Jeremy |
|
scientific name |
Tyrannosaurus rex |
| status |
|
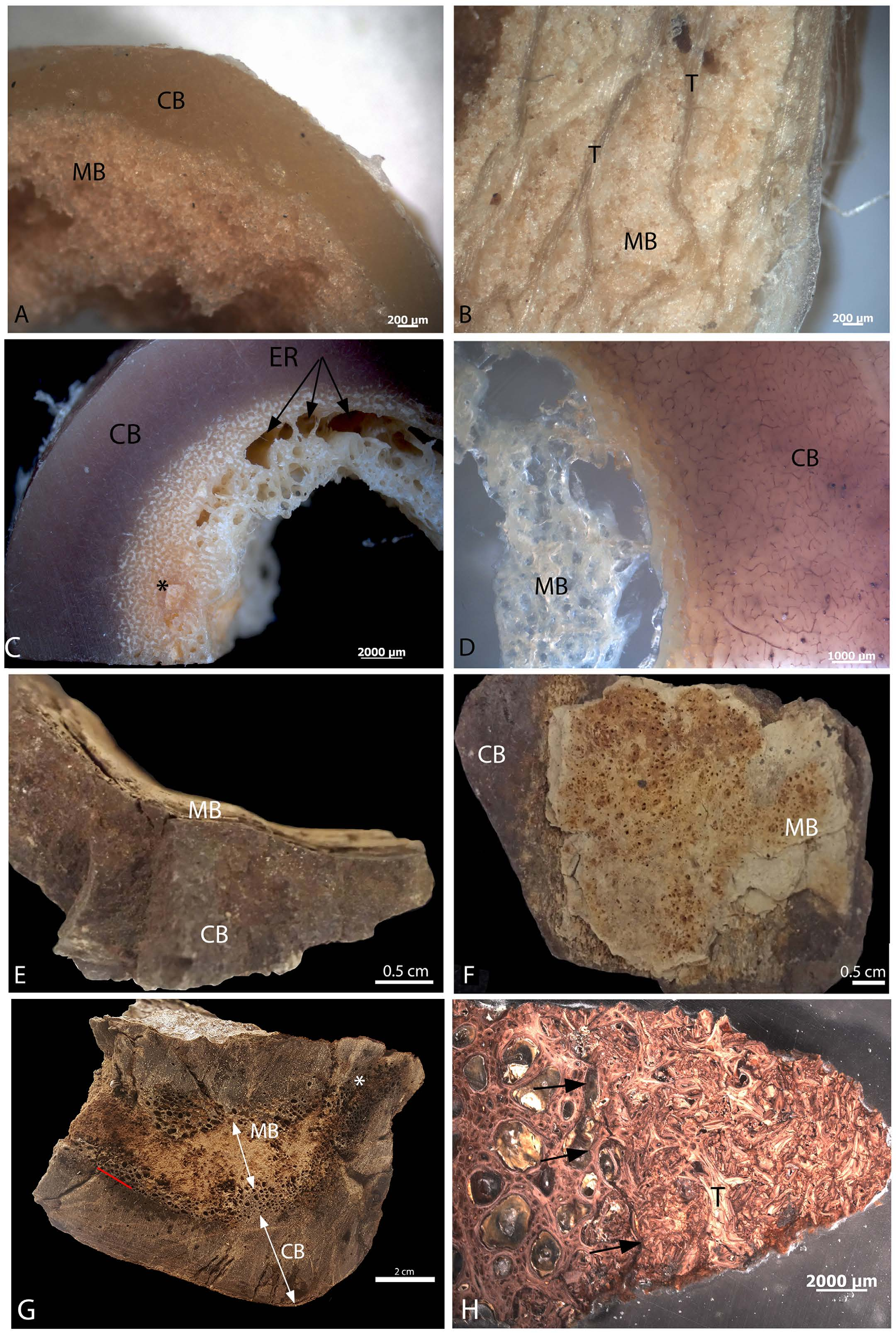
MB is clearly visible in both extant and extinct dinosaur bone ( Fig. 1 View Figure 1 ), and is morphologically and texturally distinct from overlying CB and/or TB in hand sample. MB is fibrous, vascular and randomly organized, non-lamellar woven bone, consistent with its rapid deposition (e.g. 40). In chicken ( Fig. 1A,B View Figure 1 ) MB is distinct in color and texture from overlying CB ( Fig. 1A View Figure 1 ) and TB ( Fig. 1B View Figure 1 ). MB in ostrich femur ( Fig. 1C,D View Figure 1 ) likewise shows visible differentiation in color and organization, but in hand sample, MB seems to grade from CB, becoming less dense and more spiculated as it extends toward the medullary cavity. Large erosion rooms (ER) are visible in the loosely organized MB ( Fig. 1C View Figure 1 ) and at the boundary between CB and forming MB, and white crystalline MB occasionally fills the erosion rooms ( Fig. 1C View Figure 1 , *). At higher magnification in ground section ( Fig. 1D View Figure 1 ), a distinct separation between ostrich CB and MB can be seen that is not obvious in hand sample. Similarly, MB is attached to, but distinct from overlying T. rex CB in texture, color, vascularity and organization ( Fig. 1E,F View Figure 1 ). The proximal femur shaft of MOR 1125 shows no expansion or distortion consistent with osteopetrosis in gross transverse section ( Fig. 1G View Figure 1 ), but cortical bone (CB) is clearly distinct from hypothesized MB (arrows), which completely fills the medullary cavity. A red line marks the distinction between CB and MB, separated by erosion rooms. These areas of porous bone can be seen to extend from the medullary cavity almost to the periosteal surface in some regions ( Fig. 1G View Figure 1 ,*). A petrographic ground section is shown in Fig. 1H View Figure 1 . Black arrows show a distinct line of separation between CB with large erosion rooms and adjacent MB, which is randomly oriented and appears fragmented. An apparent trabecular fragment (T) is seen interspersed with the MB ( Fig. 1H View Figure 1 ). Figure S1 View Figure 1 shows an expanded view of this region, with more of the inner cortical bone visible. In this microscopic section, the contrast between dense CB with secondary osteons, the region of intense resorption, and the origin of non-lamellar MB is easily visualized.
Ground sections of T. rex and ostrich CB is birefringent and anisotropic when observed using polarized light ( Fig. S2A,C View Figure 2 ), demonstrating the lamellar nature of this bone type. MB from ostrich and T. rex ( Fig. S2B,D View Figure 2 ) lacks birefringence, supporting the randomly oriented, non-lamellar nature of MB tissues, consistent with rapidly deposited woven bone.
To confirm density and organizational differences between MB and CB in this specimen of Tyrannosaurus rex , we employed high-resolution computed tomography (CT) (see Materials and Methods). MB in MOR 1125 is structurally unique and shows substantial density disparity with overlying cortical bone both in volumetric renderings ( Fig. 2A–D View Figure 2 ) and in two-dimensional transverse section ( Fig. 2E–G View Figure 2 ), independently substantiating the diagnosis of this tissue.
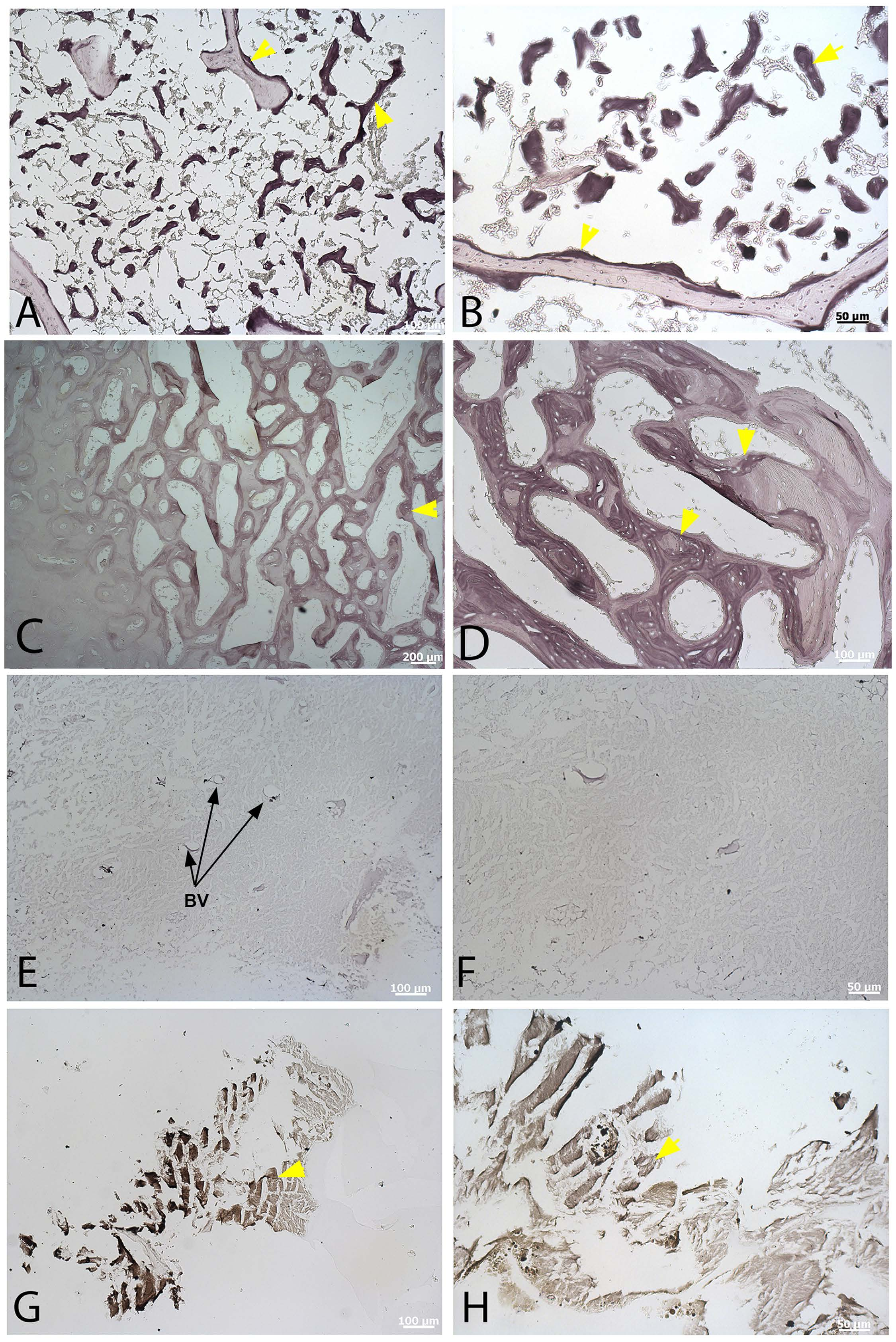
We used Alcian blue histochemical stain ( Fig. 3 View Figure 3 ) to capitalize on the chemical differences between CB and MB. This stain reacts with the acidic, sulfated glycosaminoglycan keratan sulfate 35, which is not found in cortical bone 36,37. Although Alcian blue lightly stains CB in both extant (chicken, ostrich) and extinct ( T. rex ) dinosaur bone, staining of MB is much more intense in all cases, allowing microstructural and compositional differentiation of bone types. Figure 3 A,B View Figure 3 shows low (A) and high (B) magnification images of demineralized sectioned bone from a hen in lay. Trabeculae (T) are lightly stained, supporting compositional similarity with cortical bone, but MB stains a dark blue (black arrows), and shows a pattern of MB deposition on existing trabeculae. MB arises from and lines the trabeculae and internal layers of cortical bone, but also forms as pockets within CB and TB ( Fig. 3A View Figure 3 , yellow arrows). This may be due to centripetal deposition and infilling of osteons or vessel channels with forming MB. Spicules of forming MB are occasionally penetrated by ovate “holes” that may represent vascular channels surrounded by MB matrix (red arrowheads). At higher magnifications, CB and MB are separated by large openings which may be vascular structures or, alternatively, erosion rooms that existed prior to MB deposition ( Fig. 3B View Figure 3 , black arrow).
Similarly, ostrich MB bone ( Fig. 3C View Figure 3 ) reacts more intensely to the stain, supporting its distinct chemical composition, but the pattern of deposition in this ratite differs from that of laying hen. Although CB is lightly stained, similar to the chicken ( Fig. 3C View Figure 3 , left), staining intensifies with increasing depth toward the medullary cavity (black arrows), reflecting compositional differences from CB, and allowing differentiation of MB by chemistry when it is not as obvious histologically. As in the chicken, MB can be seen to form “pockets” within pre-existing CB/TB (yellow arrows) and also is penetrated by empty ovate structures (red arrowheads), supporting the possibility that MB will deposit around vascular tissues as well as pre-existing bone. Alternatively, it may be that this represents MB from a previous lay cycle that was not fully resorbed. CB from T. rex ( Fig. 3D,E View Figure 3 ) was physically separated from underlying MB and analyzed separately (see Materials and Methods). The demineralized CB matrix is highly fibrous, and fibers show varying orientation. The matrix is only lightly stained, consistent with extant cortical bone samples, and neither the ovate structures or pockets of differentially stained regions were observed. In contrast, isolated fragments of demineralized T. rex MB ( Fig. 3F,G View Figure 3 ) are deeply stained relative to that seen in CB, and contain regions of even more intense staining (black arrows). The MB is deposited on, or retains, empty ovate “holes”, as seen in the other MB samples ( Fig. 3F View Figure 3 , red arrowheads). The significance of these is not known, but they may represent pre-existing vascular channels on which MB is deposited.
We exposed all bone types to high iron diamine (HID), a stain that reacts specifically to sulfated glycosaminoglycans 35. All are lightly stained, but MB in extant ( Fig. 4 View Figure 4 A–D, yellow arrowheads) and extinct dinosaur samples ( Fig. 4G,H View Figure 4 , yellow arrowheads) reacts much more intensely to this stain. Chicken CB and MB ( Fig. 4A,B View Figure 4 ) are differentiated by intensity of staining, which reveals spicules of new bone growth along surfaces of the more lightly stained CB. Ostrich ( Fig. 4C,D View Figure 4 ) shows a similar pattern. Histological distinction between CB and MB is not as clear as in chicken, with stain intensity increasing with depth of tissue into the medullary cavity; however, like Alcian blue, HID stain is capable of differentiating bone types and follows the same pattern as the Alcian blue stain. T. rex CB ( Fig. 4E,F View Figure 4 ) is minimally stained with HID, but within the matrix, blood vessels (BV, arrows) can be seen interspersed within the fibrous matrix. The much more intense reactivity of T. rex MB to this stain ( Fig. 4G,H View Figure 4 ) relative to CB independently supports the presence of original compounds in these dinosaur materials. Data collection parameters were identical between modern and fossil bone samples for all histochemical stains. Figure S3 View shows extant and dinosaur samples, demineralized and sectioned but unstained, as controls.
This pattern is repeated when Alcian blue is combined with HID stain, a common procedure to identify MB in modern birds 35. The dual stains ( Fig. S4 View ) differentiate MB and CB in both extant and non-avian dinosaur samples. Differential distribution of sulfated glycosaminoglycans in forming bone is clearly visualized in both chicken ( Fig. S4A,B View ) and ostrich ( Fig. S4C,D View ) by staining intensity; similarly, T. rex CB ( Fig. S4E,F View ) shows minimal reactivity, but isolated demineralized MB sections react intensely to the dual stains ( Fig. S4G,H View ).
Demineralized MB bone matrix also reacts to monoclonal antibodies specific to keratan sulfate ( Fig. 5 View Figure 5 ), allowing immunological differentiation of MB from CB or TB. Antibody-antigen complexes localize to globular structures within the MB matrix of Japanese quail 35, a pattern also seen in the present study for all samples tested. No binding is visualized when antibodies are exposed to chicken cortical bone ( Fig. 5A,B View Figure 5 ) but these antibodies bind MB, in a regular, globular pattern in the laying hen ( Fig. 5C,D View Figure 5 ). Similarly, ostrich cortical bone is negative for binding ( Fig. 5E,F View Figure 5 ), but MB shows a punctate pattern of antibody binding ( Fig. 5G,H View Figure 5 ) at identical data collection parameters. T. rex cortical bone is negative for antibody binding ( Fig. 5I,J View Figure 5 ), but a globular pattern of antibody binding is visualized in sections of demineralized dinosaur MB ( Fig. 5K,L View Figure 5 ).
Avian osteopetrosis is a viral-induced pathology which results in bone deposition on the endosteal and periosteal surfaces of affected long bones, and is usually accompanied by massive expansion in bone diameter. Because this bone is rapidly deposited and endosteally derived, it may superficially resemble MB in histological section. However, bone deposition is usually bilateral 39,41 and results in massive increases in bone diameter, most often accompanied by periosteal reaction and abnormality 41. None of these features were seen grossly in our T. rex samples ( Fig. 1G View Figure 1 ), but we applied these antibodies to sections of bone from a chicken in which DNA analyses confirmed the presence of an avian leukosis virus, the group of retroviruses that induces avian osteopetrosis 41. Positive binding would indicate molecular similarities between the matrix of MB and osteopetrotic bone. Figure 5M,N View Figure 5 is the cortical region of petrotic bone (Materials and Methods) and Fig. 5O,P View Figure 5 is the internal (medullary) regions of the same bone. These controls are negative for all samples. In other control experiments, we omitted primary antibodies, but kept all other steps identical to test conditions, to control for spurious or non-specific binding of secondary antibodies or fluorescent label to the tissues ( Fig. S5 View ).
Discussion and Conclusion
Medullary bone in extant birds is estrogen-dependent and linked to reproductive status and gender. Chemical differentiation of MB tissues in Tyrannosaurus rex implies that these factors can be extended deep into the theropod lineage. Homology can be inferred because of phylogenetic proximity, regional location within the skeleton, conserved histological features of endosteal derivation, high vascularity and isotropic arrangement of collagen fibers consistent with rapid deposition, as well as ephemeral nature (only 4 non-avian dinosaurs have been proposed to have this tissue), postcranial location, and now, molecular homologies. Identical tissues in multiple skeletal elements indicates a systemic process, consistent with MB deposition in extant birds, and the lack of periosteal reactive bone or abnormal bony enlargement in any elements negates an alternative hypothesis of avian osteopetrosis 39,42.
The fleeting nature of MB contributes to its rarity in the fossil record, but it may be possible, through careful study, to link MB unambiguously to other, less ephemeral traits, holding potential for rigorous examination of population structure, acquisition of reproductive novelties, and ontogenetic development in non-avian dinosaurs.
Original organic components are assumed to be completely destroyed during burial and fossilization processes over millions of years. However, we have shown that tissues 43 – 45, cells 46 and fragments of original molecules 44,46–51 can persist across geological time. Here we show the value of applying molecular methods to dinosaur bone to address important biological questions, using what is known of bone chemistry in homologous extant tissues.
Recently, MB was suggested to exist in pterosaurs, based upon purported histological similarity. Prondvai and Stein 52 claimed to have identified the tissue in mandibulae of both juvenile and adult specimens, and proposed a redefinition of MB to include endosteal tissues that did not play a role in reproduction. These authors then concluded that the presence of MB could not be used for gender identification. However, the assignment of this pterosaur tissue to MB is dubious, in part because it was described only in mandibular symphyses, and was not identified in any postcrania examined 52. In contrast, although MB occurs in some cranial material in a few birds, it is predominantly noted in the postcrania, specifically long bones (e.g. 3,9,10,19 and references therein). It has never been reported in mandibulae of living birds, suggesting the tissue in pterosaurs is not homologous to MB. Furthermore, although MB can be induced to form in male birds with the administration of estrogen, it does not occur naturally in either males or juveniles, as was reported for pterosaurs. Because MB in living animals is estrogen dependent 14,34 it cannot occur without the influence of these hormones. Thus the pterosaur tissues described by Prondvai and Stein 52 do not meet criteria for homology with MB in extant birds or non-avian dinosaurs as described.
Here, we demonstrate that the unique chemical composition of MB in birds is retained and can be identified in a non-avian theropod dinosaur, thereby supporting the homology of these tissues with MB in living birds. We show that it is possible to remove ambiguity associated with the assignment of MB in extinct taxa, using histochemical and immunological signatures, in cases where bone tissues retain original chemical components. The application of multiple and varied molecular techniques to fossils, as well as innovations in high resolution, nano-detection instrumentation will permit the exploration of sex-linked traits in theropod dinosaurs, offering a novel approach to investigate the paleobiology of birds and their extinct dinosaurian relatives.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |