Caliroa cerasi ( Linné, 1758 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4768.3.1 |
|
publication LSID |
lsid:zoobank.org:pub:C8036F69-F881-4727-96E7-C78AA6C7F920 |
|
DOI |
https://doi.org/10.5281/zenodo.3794883 |
|
persistent identifier |
https://treatment.plazi.org/id/03B387A9-FFD0-FF82-1DC6-213236F1FCD6 |
|
treatment provided by |
Plazi |
|
scientific name |
Caliroa cerasi ( Linné, 1758 ) |
| status |
|
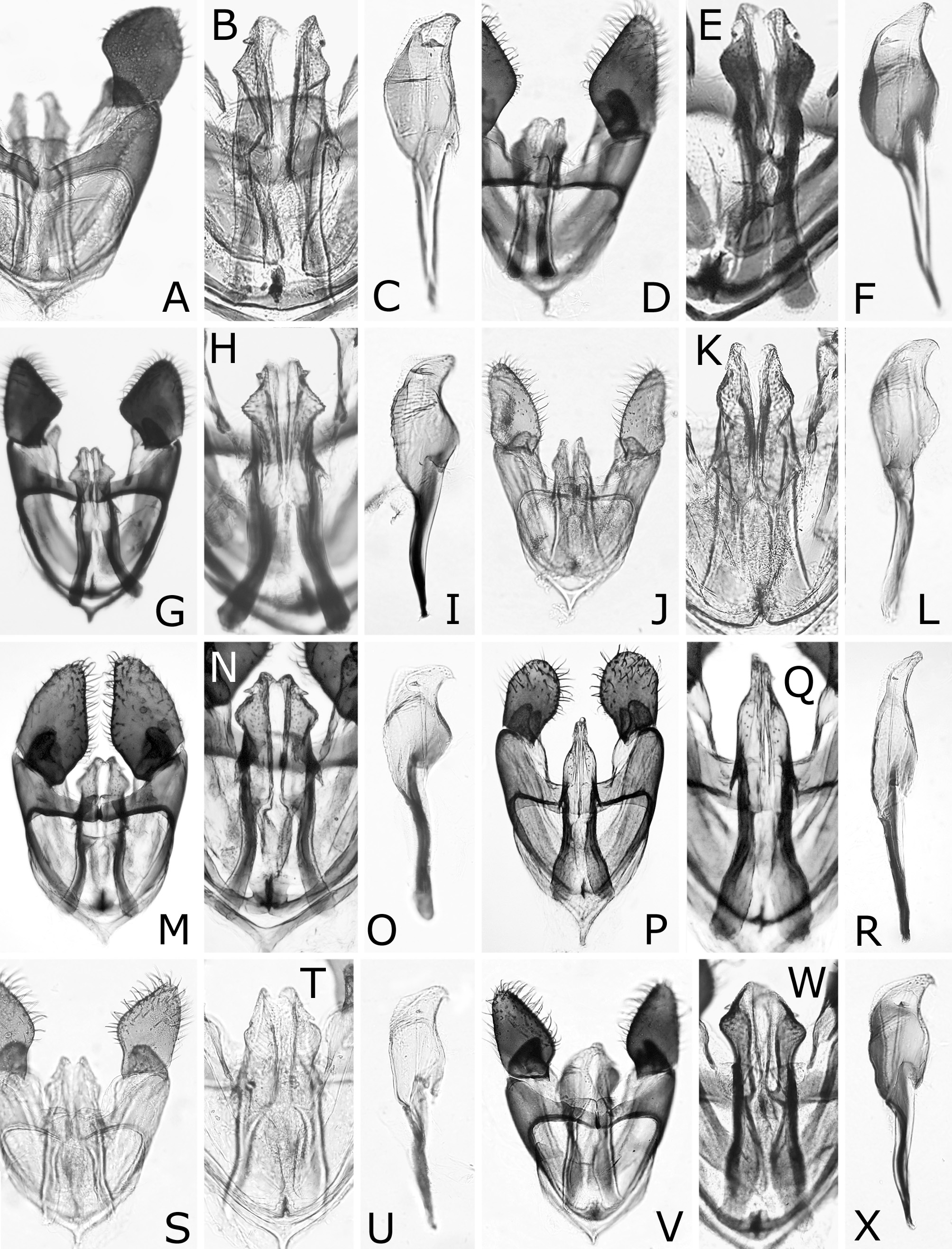
( Figs 1A, B View FIGURE 1 , 3 View FIGURE 3 B–D, N, X, 4A–C, 5A, N, 6A–D, 11A–C)
Tenthredo Cerasi [sic] Linné, 1758: 557.
Caliroa cerasi: Benson, 1952: 96 ; Oishi, 1961: 31; Okutani, 1965: 29; Okutani, 1967: 95; Smith, 1971: 14; Carl, 1972: 58; Chevin, 1974: 161; Abe & Togashi, 1989: 547; Wei, 1997: 54; Lacourt, 1999: 54; Lacourt, 2002: 128; Wei et al., 2006: 521; Lelej & Taeger, 2007: 954; Taeger et al., 2010: 365; Koch & Smith, 2011: 448; Lelej, 2012: 78.
Tenthredo limacina Retzius, 1783: 73 View in CoL .
Eriocampoides limacina: Uchiike, 1926: 4 ; Toyoshima, 1950: 104.
Eriocampoides (Caliroa) limacina: Tkahashi, 1930b: 585 .
Caliroa limacina: Benson, 1950: 83 , 113.
For more synonymy, see Smith (1971), Lacourt (1999) and Taeger et al. (2010).
Redescription: female and male. Length 4.5–5.5 mm in female, 4.0 mm in male. Black, shiny with colorless reflection ( Fig. 1A, B View FIGURE 1 ). Labrum dark brown to black. Mandible black, apically reddish brown. Palpi dark brown to black. Legs black; foreleg yellow to brown from apex of femur to tarsus; middle leg brown to dark brown from apex of femur to tarsus; hind leg with apex of femur narrowly brown and tibia and tarsus often dark brown; spurs and claws yellow to brown. Wings mostly colorless transparent; forewing slightly blackish below stigma and on cells C and Sc; veins and stigma black.
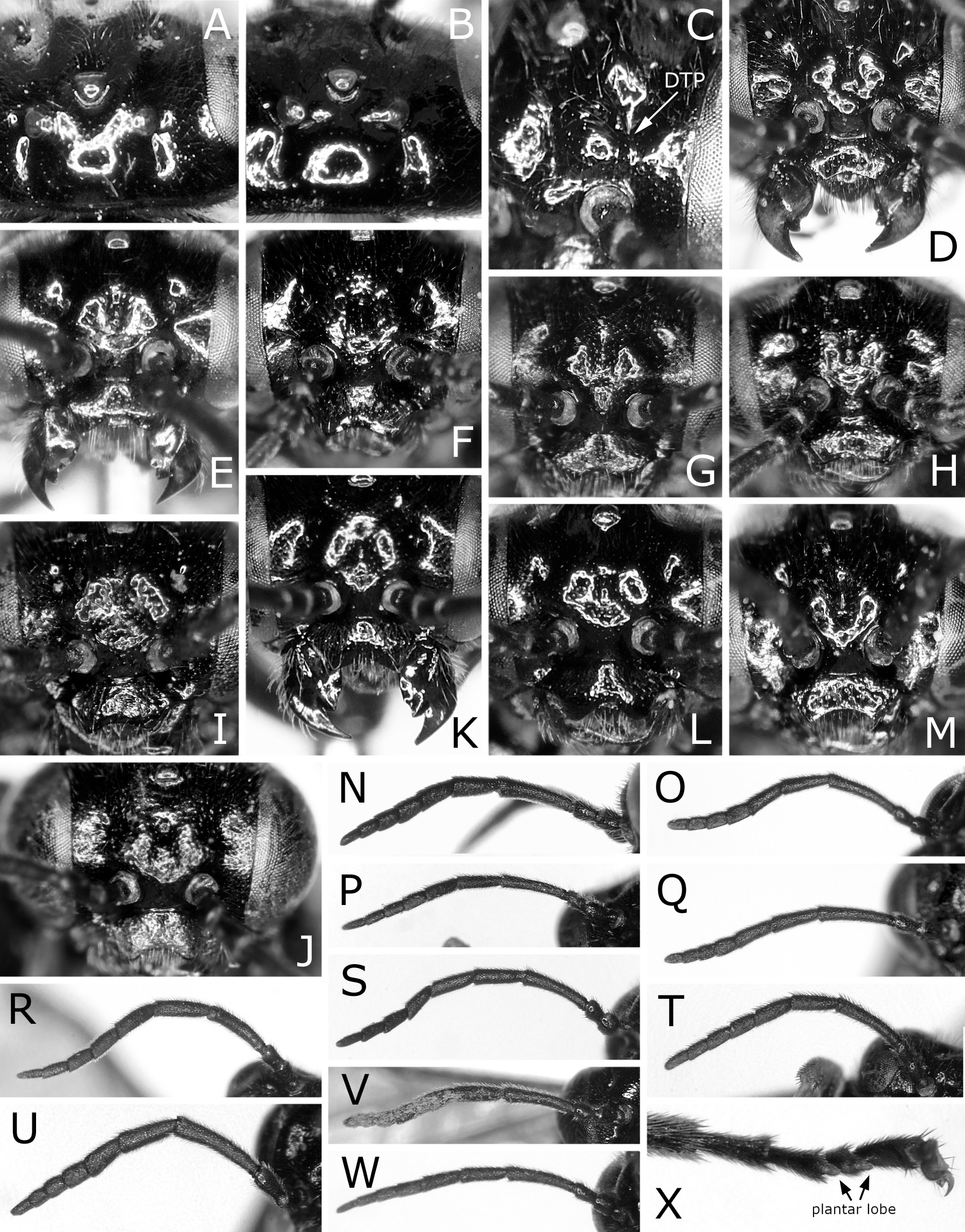
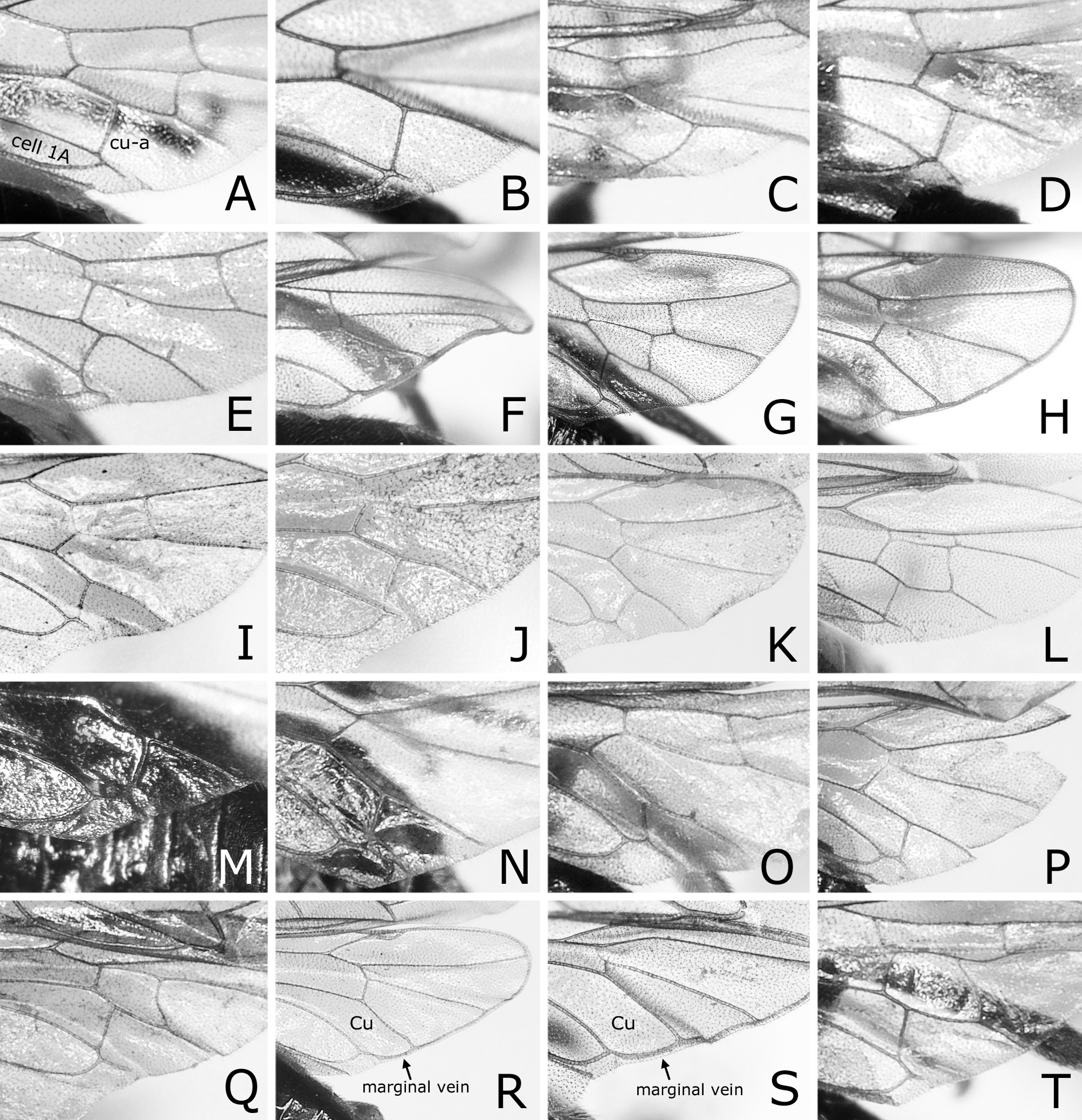
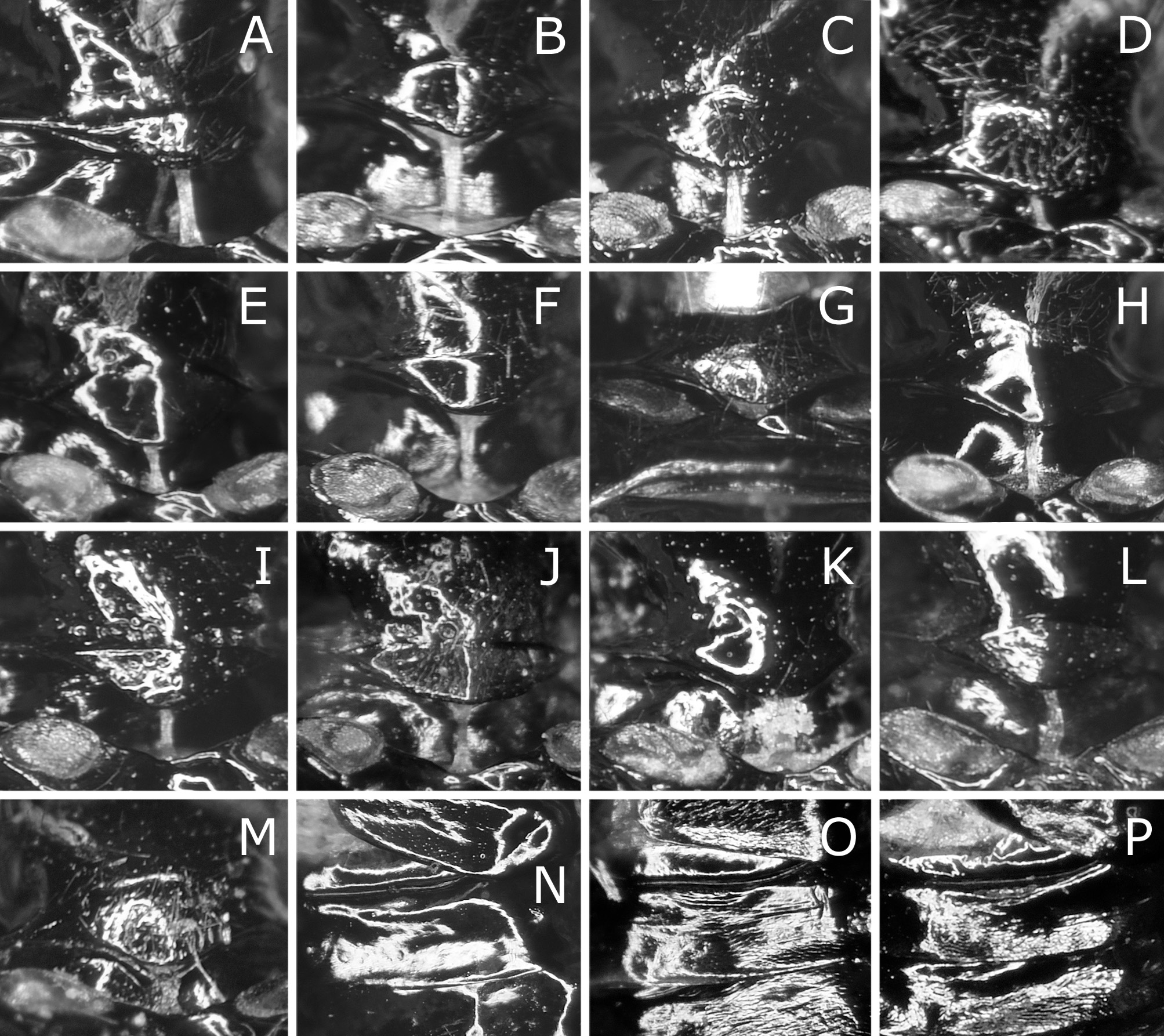
Postocellar area 1.3–1.5 × as wide as length behind lateral ocellus, with anterior groove laterally ( Fig. 3B View FIGURE 3 ). Clypeus roundly or angularly emarginate on ventral margin ( Fig. 3D View FIGURE 3 ); depth of emargination 0.3–0.4 × median length of clypeus. Malar space linear, about as wide as or slightly wider than facet of eye, without setae. First flagellomere 0.9–1.0 × as long as second and third flagellomeres combined ( Fig. 3N View FIGURE 3 ); apical four flagellomeres combined 1.0–1.2 × as long as first flagellomere. Forewing with joint of vein Rs and crossvein 2r-rs located at apical 0.00–0.19 of anterior margin of cell 1Rs2 ( Fig. 1A View FIGURE 1 ), rarely very slightly apical to joint of vein Rs with crossvein 3r-m; basal corner of cell 1M right-angled or slightly obtuse, rarely slightly acute. Hind wing of female with joint of vein 1A and crossvein cu-a located at or slightly basal to apex of cell 1A ( Fig. 4A View FIGURE 4 ), rarely very slightly apical to apex of cell 1A ( Fig. 4B View FIGURE 4 ); crossveins 2r-m and m-cu present ( Fig. 4A View FIGURE 4 ), or either of them or both absent ( Fig. 4B View FIGURE 4 ). Hind wing of male ( Fig. 4C View FIGURE 4 ) with crossvein cu-a posteriorly curved medially and fused with apex of cell 1A; vein 1A not protruding beyond apex of cell 1A; apex of cell 1A widely separated from wing margin; crossvein 2r-m absent; crossvein m-cu absent or present; marginal vein absent.
Punctures mostly minute or inconspicuous. Head and thorax mostly smooth. Mesoscutellum with inconspicuous minute punctures only. Mesoscutellar appendage mostly setose ( Fig. 5A View FIGURE 5 ). Dorsum of abdomen not microsculptured ( Fig. 5N View FIGURE 5 ), sometimes faintly reticulately microsculptured on first tergum.
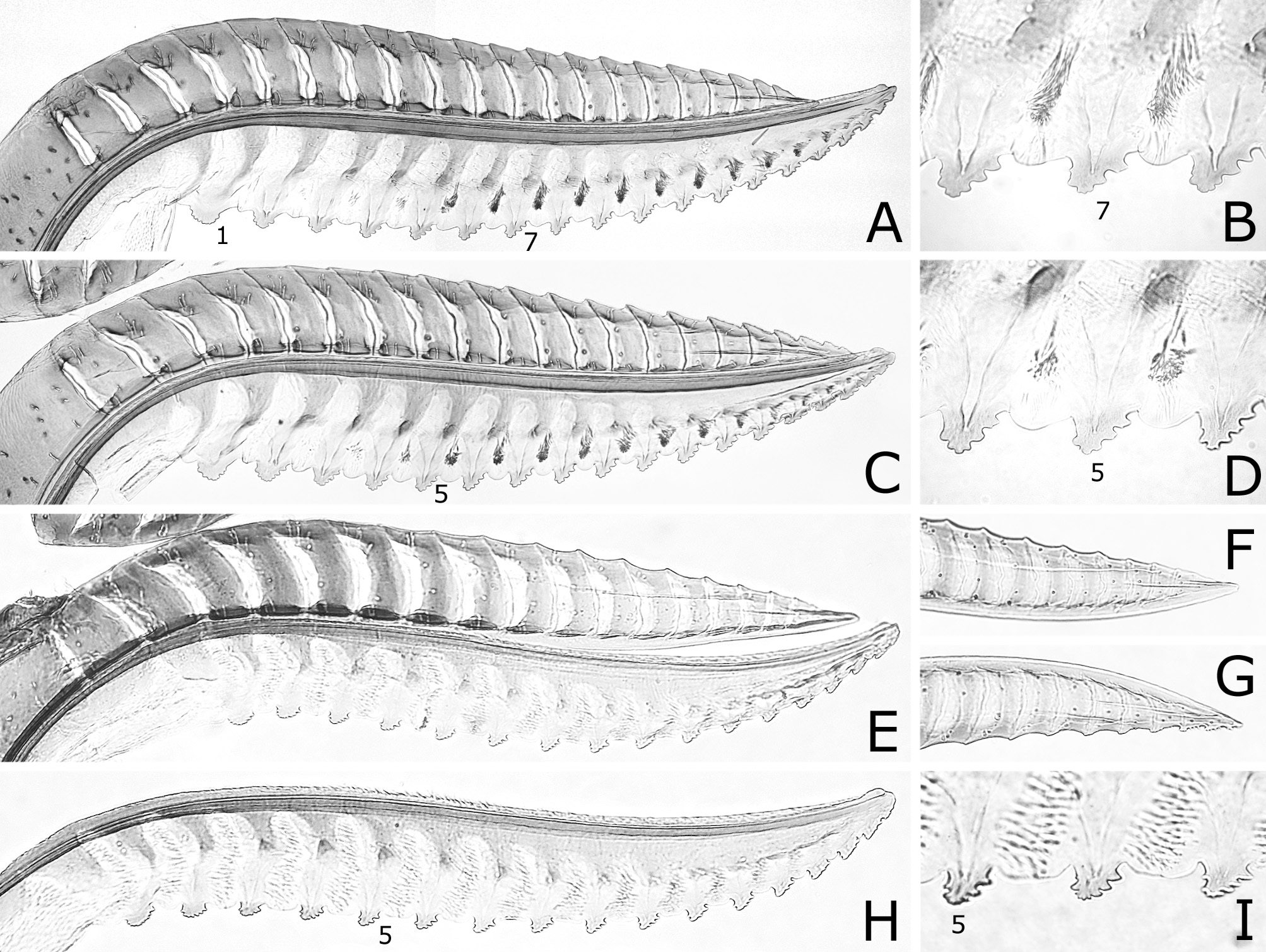
Lance ( Fig. 6A, C View FIGURE 6 ) with dorsal margin slightly serrate on apical half. Lancet ( Fig. 6 View FIGURE 6 A–D) with 18–19 serrulae; ctenidia small, distinctly darkened, widely separated from ventral margin of lancet; middle serrulae each with one to three anterior teeth and three to five posterior teeth; areas between middle serrulae convex, about as wide as or narrower than adjacent serrula.
Male genitalia ( Fig. 11 View FIGURE 11 A–C) in ventral view with gonostipes weakly concave on posteromedial margin and harpe widest at middle, gently rounded on lateral margin and distinctly convex on medial margin. Penisvalve with hooked apex.
Material examined. JAPAN—HOKKAIDO: 1♀, Nayoro , 26. VI. 2009, H. Hara ; 1♀, Urahoro , 20. VI. 2001, H. Hara ; 1♀, Shikaoi, Yamada-onsen , 25. VI. 2005, H. Hara & A. Shinohara ; 4♀, Tokachi, Shimizu , Shimizu, coll. larvae on Cerasus avium 6. VII. 2011, mat. 7–9. VII., em. 25–26. VII. 2011, H. Hara ; 1♀, same locality, 17. VIII. 2012, H. Hara ; 1♀, Hidaka, Nissho-toge , 7. VI. 2018, H. Hara ; 2♀, Sapporo , 14. VI. 1978, host: Cerasus sargentii ; 5♀, Sapporo, Ainosato , on Chaenomeles japonica (oviposition observed), 8. VI. 2009, H. Hara ; 1♀, Sapporo , Hitsu- jigaoka, 2–4. VII. 2003, K. Konishi. —HONSHU: Fukushima Pref .: 1♀, “ Japan T. Uchida. ”, “[Fukushima Uchiike (in Japanese)]” [probably material of Uchiike, 1926] ; 2♀, Hobara , V. 1933, Takeuchi, Oishi [probably material of Oishi, 1961].— FINLAND : 2♀ 1♂, Karislojo , R. Forsius. — GERMANY : 2♀, “ Dahlem , Garten, Dr. Horn ” ; 1♀, Darmstadt , Meyer. — USA : 1♀, Washington State, Wenatchee , 21. V. 1914, “on pear”, E. J. Newkomer .
Distribution. Japan: Hokkaido ( Toyoshima, 1950), Honshu ( Uchiike, 1926). Temperate Eurasia, North America, South America, Africa, Australia, Tasmania and New Zealand ( Smith, 1971).
Benson (1950, pp. 83, 113, as limacina ) stated that this species is of European origin and it was introduced to almost all parts of the world where pears, cherries and plums are grown. Koch & Smith (2011) stated that it is a widespread species in the Palearctic Region, and has been introduced into North America, South America, South Africa, Australia and New Zealand. In Japan, this sawfly is regarded as an alien species (e.g. Goka, 2019). It was recorded from Japan by Uchiike (1926) for the first time. He found the severe infestation of the sawfly in 1924 at orchards in Fukushima Prefecture, Honshu, where the saplings of cherry and plum introduced from North America were planted (see also Oishi, 1961; Uchiike and Oishi are the same person). Therefore, this sawfly was probably introduced into Japan from North America with the saplings of cherry or plum.
In Japan, C. cerasi is now common in Hokkaido but appears to be very rare in Honshu. Except for the old specimens collected in early 20th century by Uchiike (= Oishi) in Fukushima Prefecture, northeastern Honshu, we have never seen any specimens from Honshu. Murase (2010) recorded C. cerasi from Osaka Prefecture, western Honshu, but that species is not C. cerasi but probably C. matsumotonis , judging from the photograph of the adult.
Bionomics. Host plants observed in Japan are only Rosaceae : Amygdalus persica ( Oishi, 1961) , Cerasus avium ( Uchiike, 1926) , C. sargentii (new record), C. × yedoensis ( Okutani, 1967), Chaenomeles japonica (new record), Cydonia oblonga ( Oishi, 1961) , Pyrus communis ( Oishi, 1961) , P. pyrifolia ( Oishi, 1961) .
Outside Japan, the recorded hosts are various: Rosaceae ( Amelanchier , Amygdalus , Cerasus , Chaenomeles , Cotoneaster , Crataegus , Cydonia , Malus , Mespilus , Padus , Prunus , Pyrus , Rosa , Rubus , Sorbus ), Betulaceae (Betula) , Fagaceae (Quercus) , Juglandaceae (Juglans) and Salicaceae (Salix) ( Benson, 1952; Smith, 1971; Carl, 1972; Raffa & Lintereur, 1988; Taeger et al., 1998). However, Carl (1972, p. 73) stated that “No host plants other than Rosaceae were confirmed during the present study. ... Larvae collected from cherry were subjected to feeding tests and did feed on Sorbus , Rubus , Rosa and Cotoneaster , but not on Salix , Quercus and Juglans . ... the present evidence suggests that the main host spectrum of C. cerasi is in one taxonomic group, the Rosaceae , and reports of its occurrence on other, taxonomically unrelated plants require verification”. According to Schönrogge (1991), C. cerasi is an oligophagous species that is largely confined to tree and shrubby Rosaceae .
This sawfly is known as a pest of rosaceous fruit trees ( Benson, 1952). In Japan, this sawfly has two or three generations a year ( Takahashi, 1930b; Toyoshima, 1950; Hayashi et al., 1985). In Honshu, adults occur from late May to middle June, from middle July to late July and from late August to early September and larvae from early June to middle July, from late July to late August and from early September to late October ( Takahashi, 1930b). In Hokkaido, adults occur from June to August and larvae from late June to early September ( Hayashi et al., 1985). For detailed information on the life history, see Carl (1972).
Remarks. Previous authors wrote that C. cerasi has a first flagellomere about as long as or not shorter than second and third flagellomeres together ( Benson, 1952; Okutani, 1965; Smith, 1971; Chevin, 1974; Viitasaari, 1981; Wei, 1997; Zhelochovtsev & Zinovjev, 1988; Lacourt, 2002) and the anal cell of a hind wing is sessile [= the joint of a vein 1A and a crossvein cu-a is located at or basal to the apex of a cell 1A] ( Smith, 1971; Wei, 1997). However, in our material, a first flagellomere is sometimes 0.9 × as long as second and third flagellomeres together, and the anal cell of a hind wing is rarely very shortly petiolate [= the joint of a vein 1A and a crossvein cu-a is located slightly apical to the apex of a cell 1A] ( Fig. 4B View FIGURE 4 ).
Caliroa cerasi will be distinguishable from other congeners by the following features: black body without colored reflection; hind leg dark brown to black, except for narrow apex of femur slightly pale; wings colorless transparent, slightly blackish below stigma and on cells C and Sc of forewing ( Fig. 1A, B View FIGURE 1 ); postocellar area with anterior margin grooved laterally ( Fig. 3B View FIGURE 3 ); apical four flagellomeres combined 1.0–1.2 × as long as first flagellomere ( Fig. 3N View FIGURE 3 ); forewing with joint of vein Rs and crossvein 2r-rs usually located at or close to joint of vein Rs and crossvein 3r-m ( Fig. 1A View FIGURE 1 ); hind wing of female with joint of vein 1A and crossvein cu-a located basal to, at or very slightly apical to apex of cell 1A ( Fig. 4A, B View FIGURE 4 ); hind wing of male without marginal vein ( Fig. 4C View FIGURE 4 ); lancet with ctenidia distinctly darkened and widely separated from ventral margin of lancet ( Fig. 6 View FIGURE 6 A–D).
| VI |
Mykotektet, National Veterinary Institute |
| T |
Tavera, Department of Geology and Geophysics |
| V |
Royal British Columbia Museum - Herbarium |
| R |
Departamento de Geologia, Universidad de Chile |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Caliroa cerasi ( Linné, 1758 )
| Hara, Hideho & Ibuki, Shinichi 2020 |
Caliroa cerasi: Benson, 1952: 96
| Lelej, A. S. 2012: 78 |
| Koch, F. & Smith, D. R. 2011: 448 |
| Taeger, A. & Blank, S. M. & Liston, A. D. 2010: 365 |
| Lelej, A. S. & Taeger, A. 2007: 954 |
| Wei, M. & Nie, H. & Taeger, A. 2006: 521 |
| Lacourt, J. 2002: 128 |
| Lacourt, J. 1999: 54 |
| Abe, M. & Togashi, I. 1989: 547 |
| Chevin, H. 1974: 161 |
| Carl, K. P. 1972: 58 |
| Smith, D. R. 1971: 14 |
| Okutani, T. 1967: 95 |
| Okutani, T. 1965: 29 |
| Oishi, T. 1961: 31 |
| Benson, R. B. 1952: 96 |
Caliroa limacina: Benson, 1950: 83
| Benson, R. B. 1950: 83 |
Eriocampoides limacina:
| Toyoshima, A. 1950: 104 |
| Uchiike, T. 1926: 4 |
Tenthredo limacina
| Retzius, A. J. 1783: 73 |
Tenthredo
| Linne, C. 1758: 557 |