Nanhaipotamon zhuhaiense, Huang, Chao, Huang, Jian Rong & Ng, Peter K. L., 2012
|
publication ID |
https://doi.org/10.5281/zenodo.213642 |
|
DOI |
https://doi.org/10.5281/zenodo.6180148 |
|
persistent identifier |
https://treatment.plazi.org/id/01307731-FF98-9932-B2B5-FE463FFEFB1C |
|
treatment provided by |
Plazi |
|
scientific name |
Nanhaipotamon zhuhaiense |
| status |
sp. nov. |
Nanhaipotamon zhuhaiense View in CoL , n. sp.
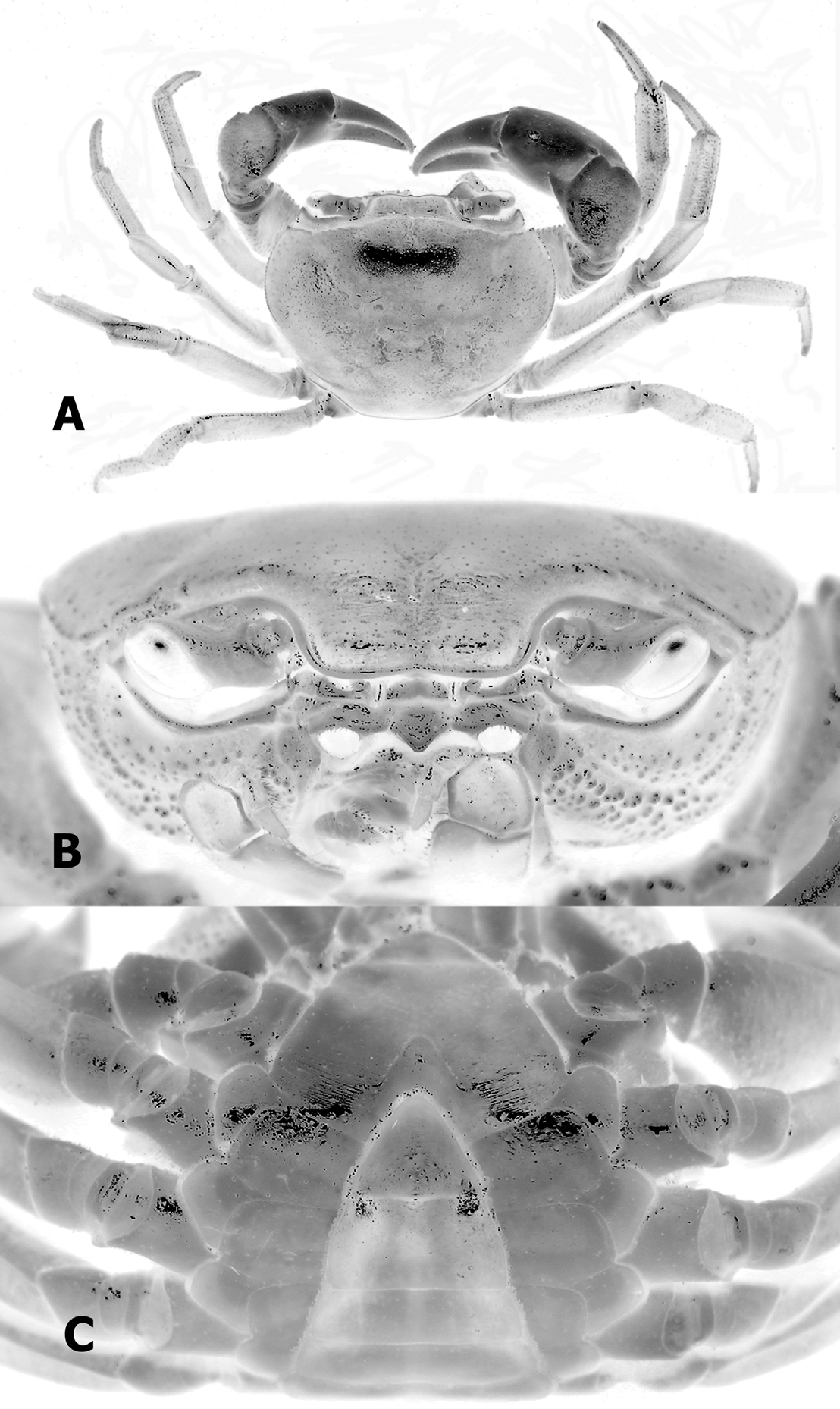
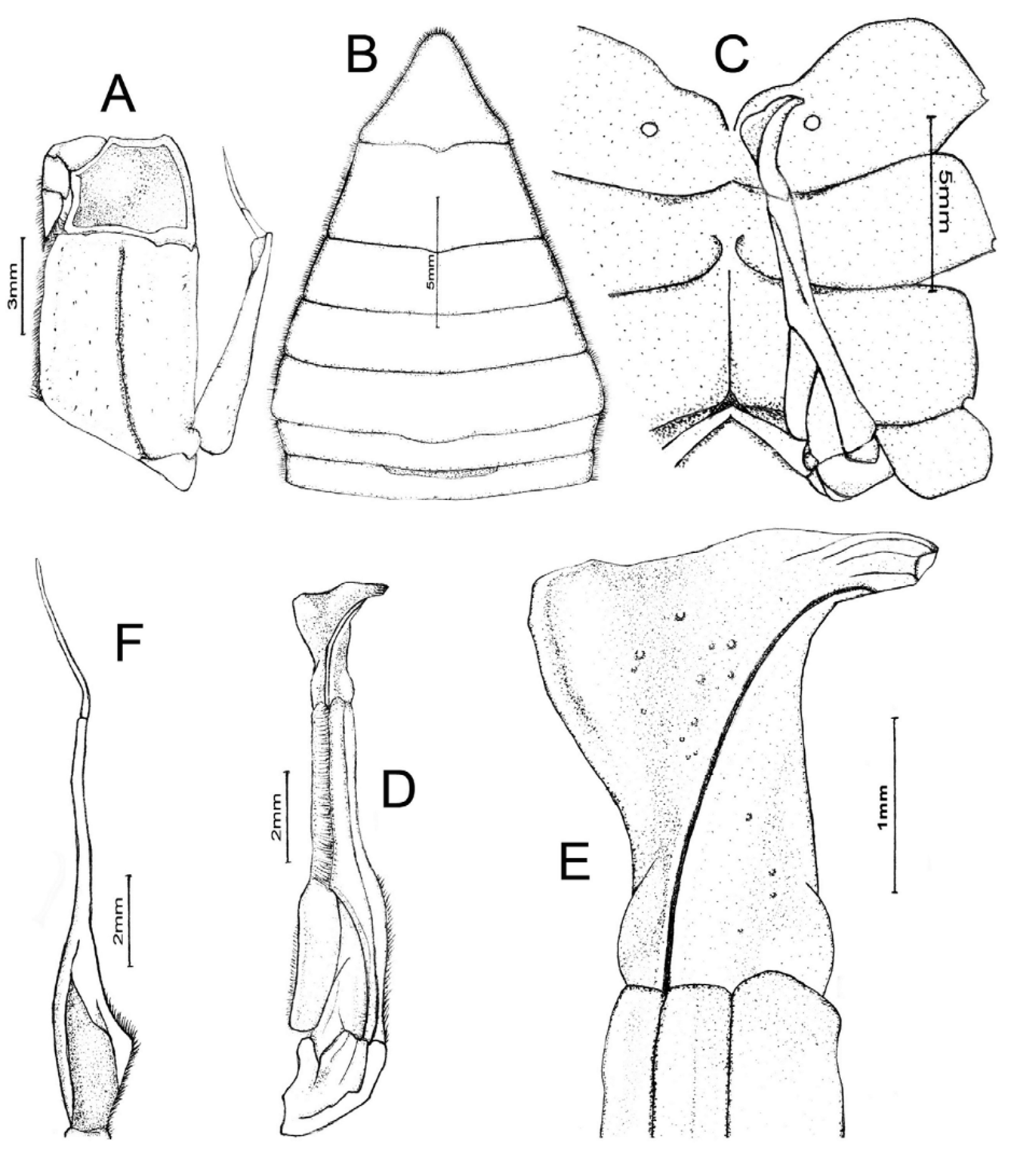
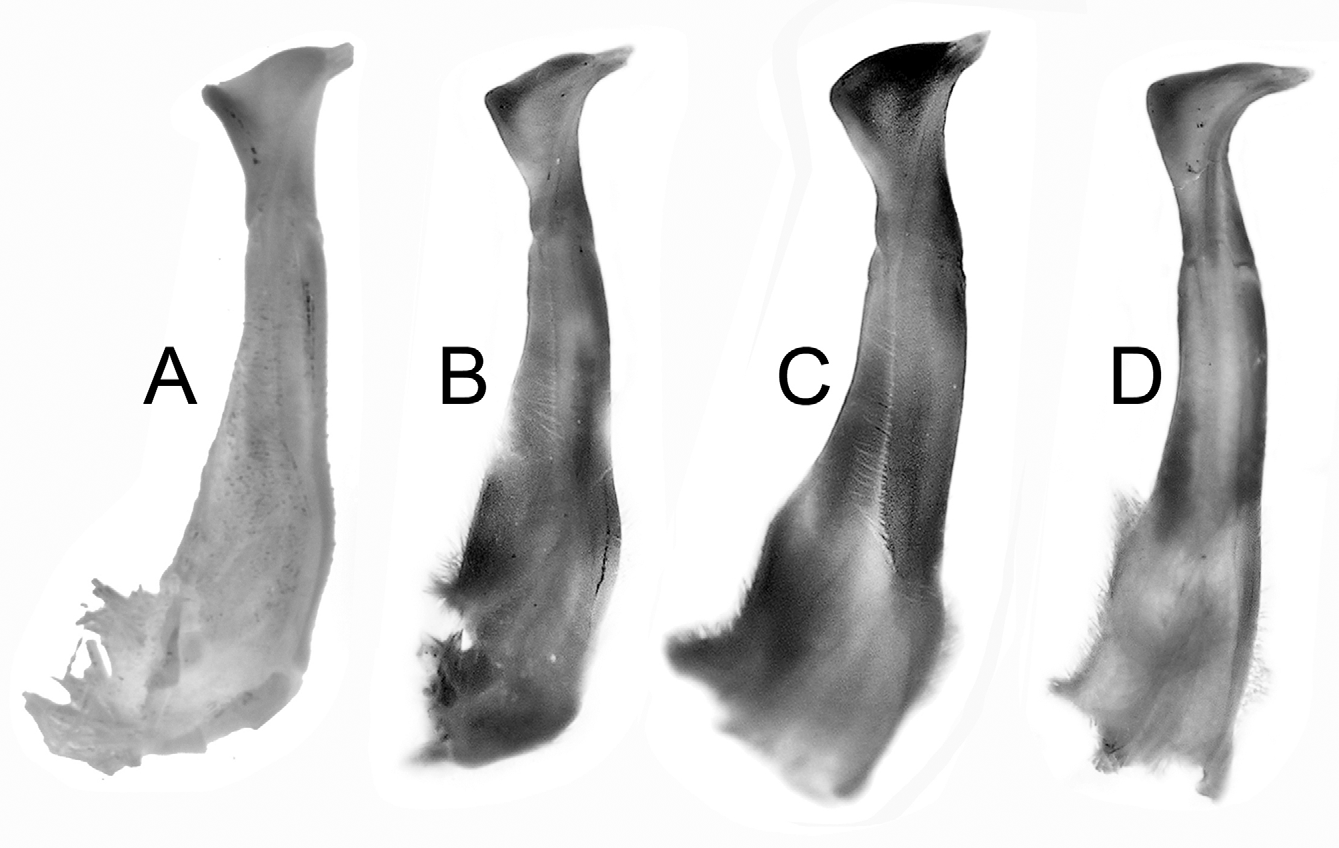
( Figs. 1 View FIGURE 1 B, 2, 3, 5D)
Material examined. Holotype: male (38.5 × 30.0 mm) ( SYSU 001001), Zhuhai, Guangdong, China, coll. C. Huang, May 2012. Paratypes: 1 male (27.4 × 22.5 mm) ( SYSU 001002), 1 male (30.5 × 25.2 mm) ( ZRC 2012.0792), same data as holotype.
Comparative material. Nanhaipotamon guangdongense Dai, 1997 : holotype male (33.2 × 26.4 mm) (AS-CB 05141), Guangdong Province, gift from Sun Yat-Sen Medical College, no date [photographs examined]; 1 male (36.2 × 28.4 mm) ( SYSU 001003), Zhuhai, Guangdong, China, coll. C. Huang, February 2011, 1 male (30.5 × 24.3 mm) ( SYSU 001004), 3 males (42.6 × 25.6 mm, 30.7 × 25.3 mm, 22.1 × 18.1 mm) ( ZRC 2012.0795), Zhuhai, Guangdong, China, coll. C. Huang, August 2012.
Diagnosis. Carapace surface generally smooth ( Fig. 2 View FIGURE 2 A); epigastric, postorbital cristae confluent, sharp ( Fig. 2 View FIGURE 2 A, B); external orbital angle acutely triangular with small but sharp epibranchial tooth ( Fig. 2 View FIGURE 2 A, B); anterolateral border cristate, lined with numerous granules Fig. 2 View FIGURE 2 A, B); sub-orbital, subhepatic, pterygostomian regions covered with rounded granules ( Fig. 2 View FIGURE 2 B); male sterno-abdominal cavity reaching to imaginary line joining median part of coxae of chelipeds ( Fig. 2 View FIGURE 2 C); male abdomen broadly triangular ( Figs. 2 View FIGURE 2 C, 3B); G1 slender, inner margin prominently concave, tip of terminal segment reaches beyond tubercle of abdominal locking structure in situ ( Fig. 3 View FIGURE 3 C), distal part distinctly elongated, bent laterally outwards at angle of about 90° to vertical ( Figs. 3 View FIGURE 3 D, E, 5D); G2 basal segment about 2.2 times length of distal segment ( Fig. 3 View FIGURE 3 F).
Description. Carapace surface generally smooth, convex transversely, longitudinally; regions behind front weakly rugose, pitted. Front deflexed; margin sinuous on dorsal view. Epigastric cristae low, separated by narrow gap. Epigastric, postorbital cristae confluent. Postorbital cristae sharp, confluent with epibranchial teeth. Branchial region swollen; cervical grooves shallow but visible, joining obscure H-shaped gastric groove. External orbital angle prominent, sharp, acutely triangular, outer margin gently convex to almost straight, longer than inner margin. Epibranchial tooth small but sharp, clearly demarcated from external orbital tooth by clear notch. Anterolateral border cristate, lined with numerous granules; posterolateral margin almost smooth or with very low oblique striae, sharply converging towards gently convex posterior carapace margin. Orbits large; margins sinuous, cristate. Suborbital, subhepatic, pterygostomian regions covered with rounded granules. Third maxilliped with merus about 1.1 times as broad as long, ischium about 1.5 times as long as broad; merus quadrate, with prominent median depression, covered with flattened granules; ischium subrectangular with distinct median sulcus; exopod reaching to proximal third of merus, flagellum about as long as width of merus. Posterior margin of epistome narrow; median triangular lobe large, lateral margins almost straight.
Chelipeds unequal, especially in males. Merus trigonal in cross-section; margins crenulated, granulated, otherwise unarmed. Carpus with sharp spine on inner distal angle, with spinule at base; inner dorsal surface with curved row of low granules. Larger chela about 1.3 times as long as high; fixed finger subequal to movable finger, with small gap when closed.
Ambulatory legs slender, surfaces smooth, unarmed; last leg with propodus about 2.4 times as long as broad, shorter than dactylus.
Thoracic sternum generally smooth or weakly pitted; sternites 1, 2 completely fused to form triangular structure with convex lateral margins; sternites 3, 4 fused without trace of median sutures; male sterno-abdominal cavity reaching to imaginary line joining median part of coxae of cheliped; median longitudinal groove between sternites 7, 8 deep.
Male abdomen broadly triangular; somites 3–6 progressively broader longitudinally, lateral margins almost straight to gently convex; somite 6 about 2.1 times as broad as long; telson about 1.2 times as broad as long, slightly concave proximally but otherwise gently convex, tip rounded.
G1 slender, inner (sternal) margin prominently concave, tip of terminal segment reaches beyond tubercle of abdominal locking structure in situ; subterminal segment about 3.1 times length of terminal segment; inner-distal angle of terminal segment angular but edge rounded, anterior margin sinuous; distal part distinctly elongated, bent laterally outwards at angle of about 90° to vertical. G2 basal segment about 2.2 times length of flagelliform distal segment.
Etymology. Nanhaipotamon zhuhaiense is named after the type locality, Zhuhai, in Guangdong Province, China.
Colour. Carapace and ambulatory legs are bluish-white, chelipeds generally white ( Fig. 1 View FIGURE 1 B).
Ecology. Like N. guangdongense , the new species inhabits mud burrows near the banks of densely vegetated creeks. The burrows can be as deep as one meter and always have some water at the bottom. Crabs reside in their burrows during the day and are relatively inactive, but are particularly active during hot, humid nights. They are omnivorous, eating dead leaves, earthworms, freshwater shrimps, other crabs, etc. Brooding females have been observed in February. Cannibalism has been observed in captive specimens and may also occur in the wild; large specimens are generally more aggressive.
Remarks. Nanhaipotamon zhuhaiense n. sp. can most easily be distinguished from N. guandongense by the live coloration of its carapace, the former being bluish-white ( Fig. 1 View FIGURE 1 B) with the latter generally brown ( Fig. 1 View FIGURE 1 A). Although the colour of N. guandongense varies slightly from yellowish to orangish-brown, it never attains the colours of N. zhuhaiense n. sp..
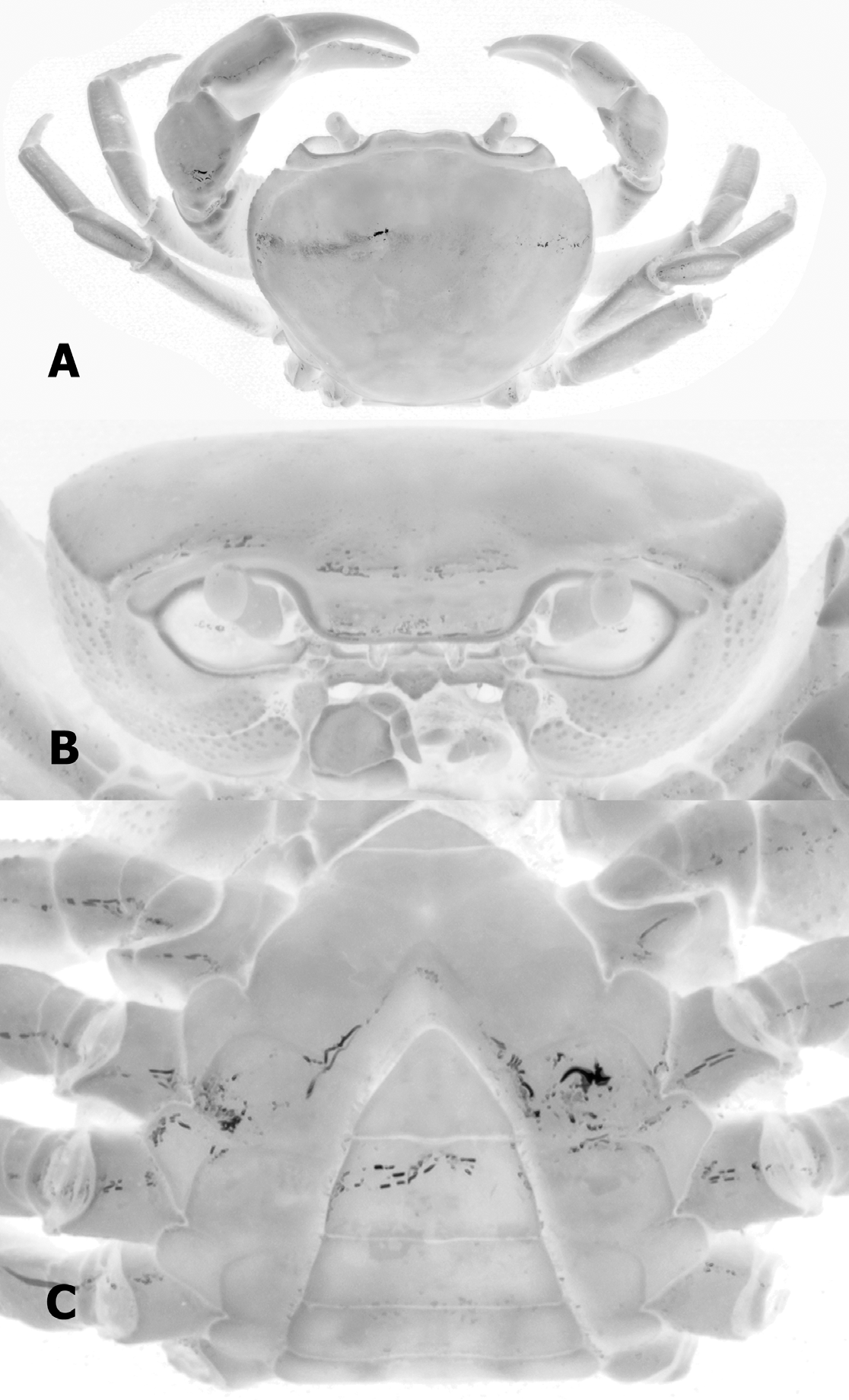
As with most Nanhaipotamon species, the carapace, chelipeds and ambulatory legs are extremely similar in form and we could discern no major differences between N. guandongense and N. zhuhaiense n. sp.. The male abdomen of N. guandongense is relatively wider, especially somite 6 ( Fig. 4 View FIGURE 4 C), with that of N. zhuhaiense , new species, relatively narrower ( Fig. 2 View FIGURE 2 C) (Table 1). The male abdomen of N. guandongense (see Dai 1997: Fig. 9-2) is not accurate as it shows a relatively narrower structure. The male abdomen of the holotype is actually proportionately wider ( Fig. 4 View FIGURE 4 C). The most significant difference is in the structure of the G1. While they are superficially similar, that of N. guandongense is more curved, with the inner distal margin distinctly concave ( Fig. 5 View FIGURE 5 A–C), while that of N. zhuhaiense n. sp., is relatively straight ( Fig 5 View FIGURE 5 D). The form of the terminal segment is also different; in N. zhuhaiense n. sp. the distal part is distinctively more elongated and is bent laterally at almost 90° to the vertical ( Figs. 3 View FIGURE 3 D, E, 5D). In N. guandongense , the distal part is shorter and bent obliquely outwards at an angle of about 45° to the vertical ( Fig. 5 View FIGURE 5 A–C). These differences are consistent and are beyond the variation observed or known for Nanhaipotamon species.
TABLE I. Morphological differences among Nanhaipotamon zhuhaiense n. sp. and N. guangdongense Nanhaipotamon zhuhaiense n. sp. has been found only in small numbers from just one site in Zhuhai, where they are sympatric with N. guangdongense . Their population does not appear to be large, and considering their very limited range, we had concerns about their conservation status and therefore restricted our collection of samples. Observations of several more adult and juvenile specimens of N. zhuhaiense n. sp. in the field and laboratory nevertheless confirm the validity of the colour differences observed with N. guangdongense . Although there are reports, based on the basis of their distinctive coloration, that it is present in other areas around of Zhuhai, we have not been able to find them. In all the other areas surveyed around Zhuhai, the only species present is N. guangdongense . This is only the second case of sympatric occurrence of two species in the genus, the other being N. hepingense Dai, 1997 , and N. pinghense Dai, 1997 . Shih et al. (2011: 437) indicated, however, that there is good evidence that these two species (both described on the basis of only one male each from the same location and collected on the same date) are synonymous. In the case of N. guangdongense and N. zhuhaiense n. sp. the most likely explanation is that the two species were originally allopatric and human activities (land development and moving of agricultural plants and animals) causing part of their ranges to overlap. More thorough surveys will need to be done to determine the precise and original distributions of both species.
At the one site where both N. guangdongense and N. zhuhaiense n. sp. are present, the former species is the dominant one. The burrows of these two species have even been found adjacent to each other.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
InfraOrder |
Brachyura |
|
Family |
|
|
Genus |