Ancylosis ciliatella ( Zeller, 1872 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4822.4.1 |
|
publication LSID |
lsid:zoobank.org:pub:4329F798-9591-4711-88E7-3B9EE1C8864D |
|
DOI |
https://doi.org/10.5281/zenodo.4458055 |
|
persistent identifier |
https://treatment.plazi.org/id/03CB87DC-FFE3-7E42-FF52-17BAFEE9F804 |
|
treatment provided by |
Plazi |
|
scientific name |
Ancylosis ciliatella ( Zeller, 1872 ) |
| status |
|
Ancylosis ciliatella ( Zeller, 1872) View in CoL spec. rev., stat. nov.
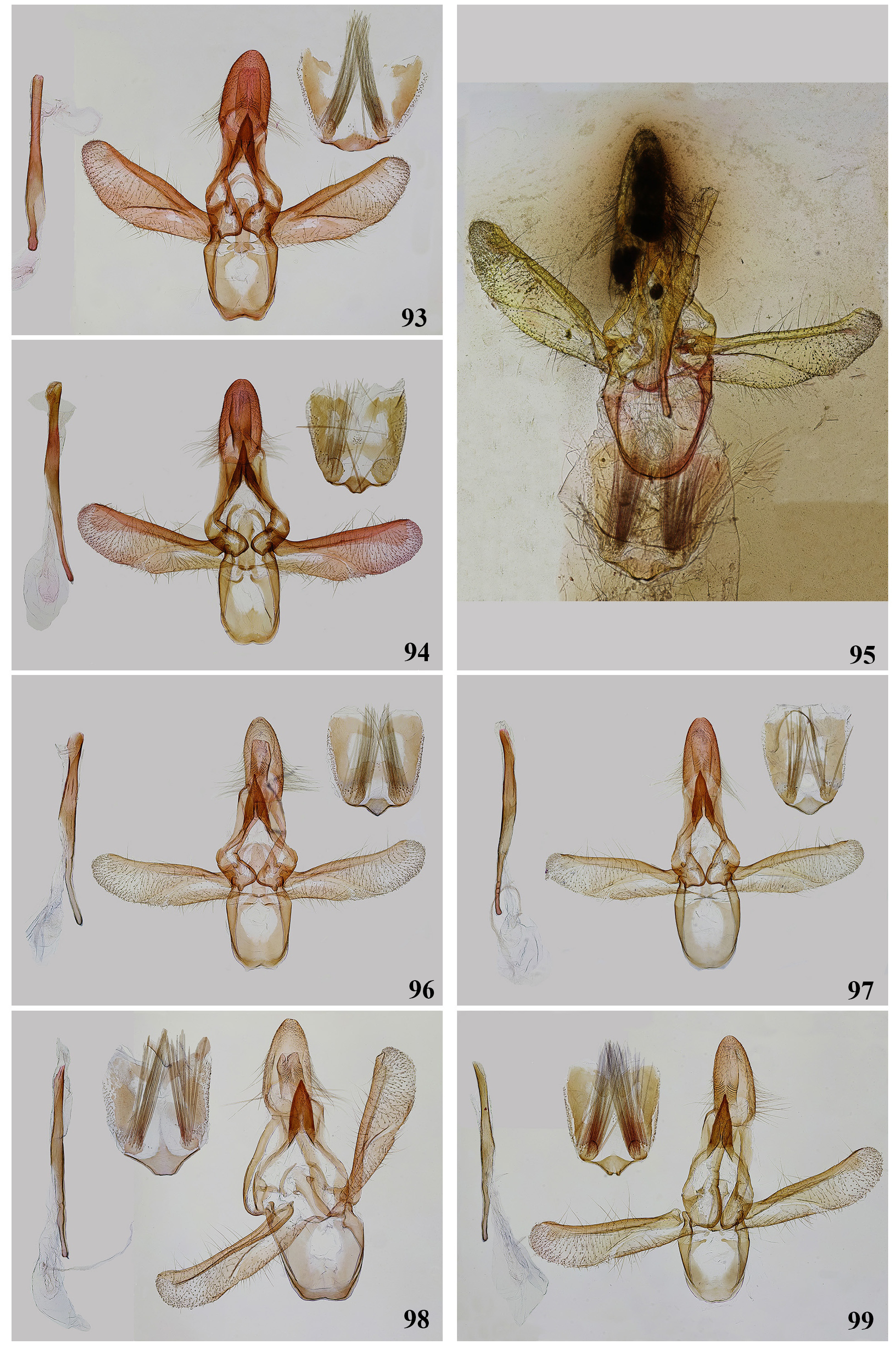
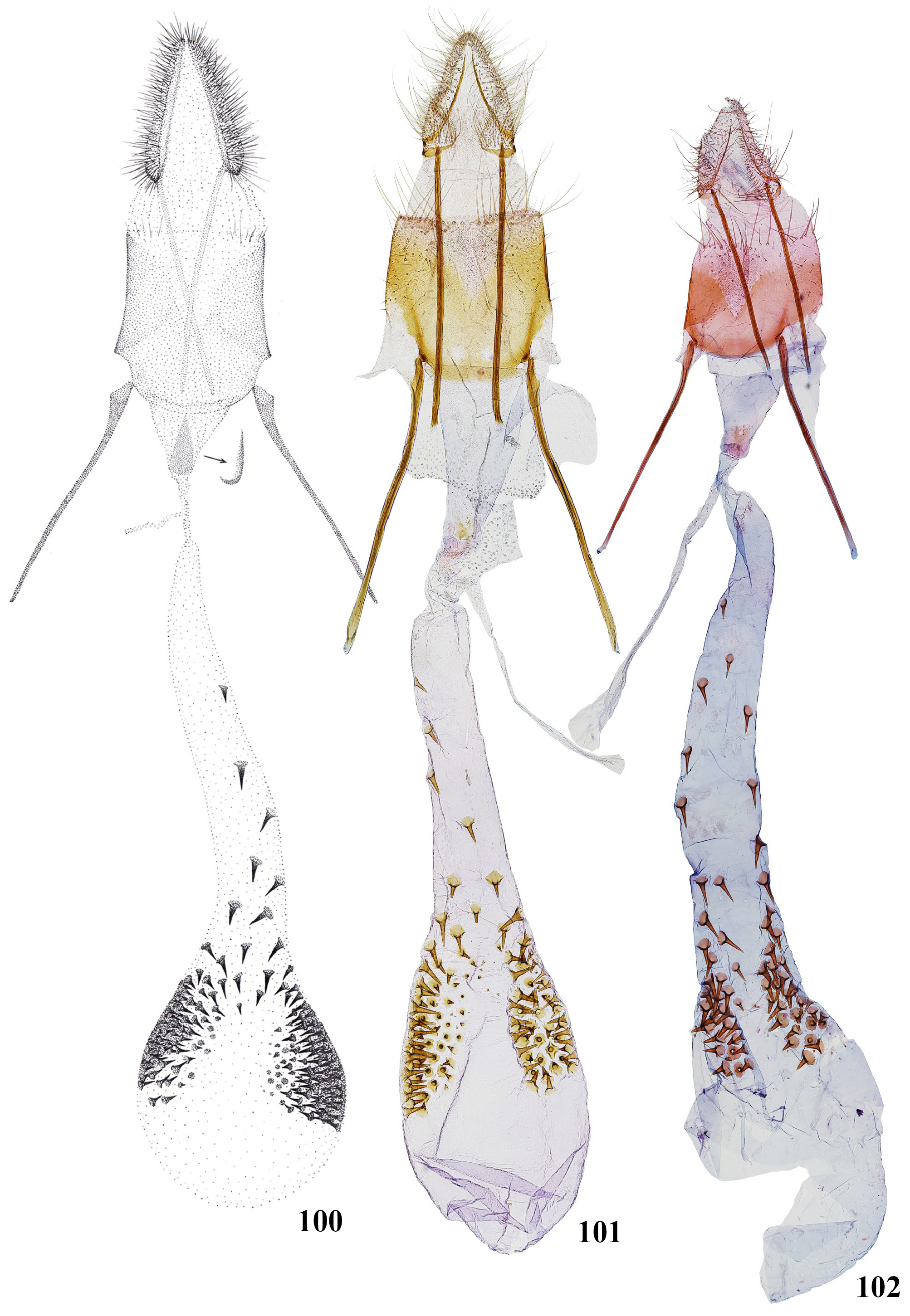
Figs 78–81 View FIGURES 78–92 , 93 View FIGURES 93–99 , 100 View FIGURES 100–102
Myel. [ois] ciliatella Zeller, 1872 — Verhandlungen der Zoologisch-Botanischen Gesellschaft in Wien, 22: 557. TL: Volgograd [Sarepta], Russia.
Type material examined. Holotype of ciliatella : ♂ “ ciliatella Z., Sarepta Chr. 69” | “mihi ighnota”, Stdg. Lit.14/3 71” | “Zell. Coll., 1884”| “Type” ( NHM).
Additional material examined. 2 ♂, 1 ♀, Russia: Astrakhan reg., W bank of Lake Baskunchak , 7.v.2018 ; 2 ♂, 3 ♀, Kazakhstan : 1 ♂, Emba, Uralsk | 954 | Heterographis pectinatella (gen. slide 284/19, O. Bidzilya) ( NHMW); Mangistau reg., 43 km SE of Shetpe, sands, 4.v.2019 ; 2 ♂, 2 ♀, Kazakhstan: Turkestan reg., 33 km NE of vill. Sozak, sands, 25.vi.2019 (all ET).
Comparative material examined. Ancylosis labeculella : 1 ♂, 1 ♀, Tunisia, Djedeida , 30.v.1913 (gen. slide 280/ 19♂; 279/ 19♀, O. Bidzilya) ; 1 ♂, 1 ♀, Maknassy ( Tunisia ), 2.vi.1927 (Dumont) ; 1 ♂, 1 ♀, Hung. Zool. Miss., Tunisia: Sfax, Thyna; sea coast: Juncus , Salsola , Phragmites ass., 7.iv.1977; lamp, No. 85 (Gozmány & Mahunka) (all NHMB) .
Ancylosis pectinatella : lectotype ♂, “Alai, 85 Hbh.” | “953” | ” Heterographis pectinatella Rag , type, orig., pl. XXVIII, fig. 16” | “ U. Roesler, ♂, GU: 5091” ( MfN) .
Ancylosis pr. pectinatella 1 ♂, 1 ♀, Hung. Zool. Miss., Tunisia: Tozeur, 14.iv.1977; lamp, No. 109 (Debreszy et Mahunka) (gen. slide 347/ 19♂; 348/ 19♀, O. Bidzilya) ( NHMB) .
Diagnosis. Ancylosis ciliatella is a rather variable species ( Figs 78–81 View FIGURES 78–92 ). The forewing is varying from pale, light brown to reddish-brown and magenta, but white costal strip is usually distinct. This forewing pattern in combination with narrow elongated forewing with distinct apical and tornal angles and long cilia on the male antennae are most constant characters for separating of A. ciliatella from related species of Ancylosis .
Ancylosis ciliatella seems most related to Ancylosis pectinatella ( Ragonot, 1887) , A. yerburii ( Butler, 1884) and A. labeculella ( Ragonot, 1887) both in the genitalia and externally sharing long cilia on the male antennae. Ancylosis pectinatella differs in a broader forewing with more rounded apical and tornal angles, pale and less contrasting ground colour and more diffuse white costal strip ( Figs 82–84 View FIGURES 78–92 ). Ancylosis labeculella is distinguished by the absence of white costal strip and presence of dark discal spot ( Figs 85, 86 View FIGURES 78–92 ). Ancylosis yerburii is usually darker, greyish-brown rather than light brown, sometimes with white suffusion on the forewing ( Figs 88–92 View FIGURES 78–92 ).
Long cilia on the male antennae (more than twice as long as the width of flagellum) is a reliable character for separating A. ciliatella from externally somewhat similar A. larissae , A. delicatella , A. amseli and A. albicosta . Two first differ additionally in the absence of short hump on white costal streak, the absence of white suffusion in basal 1/4–1/5 of dorsal margin and uniformly reddish-brown rather than white edged in A. ciliatella tegulae. Ancylosis amseli has no pink shade on the forewing, contrary to A. ciliatella . However, this character is rather variable and we cannot exclude the discovery of specimens mottled with pink among A. amseli . The specimens of A. ciliatella without pink scales on the forewing can be distinguished from A. albicosta by their uniformly light brown colouration, in A. albicosta the forewing pattern is usually more contrasting.
Male genitalia of A. ciliatella are recognized by medially broadened valva with distinct medial fold, broad sacculus, narrow, long apically connected trastilla lobes in combination with long (twice longer than broad) uncus and basally narrowed aedeagus ( Fig. 93 View FIGURES 93–99 ). We can hardly specify characters for reliable separating A. ciliatella from A. pectinatella , A. yerburii and A. labeculella in the genitalia alone, until this group remains unrevised. However, based on our study of available material, we consider that A. ciliatella can be separated from A. yerburii and A. labeculella ( Figs 96, 97 View FIGURES 93–99 ) by more convex ventral edge of valva, presence of paired ventral sclerites of juxta near base of aedeagus, and ductus ejaculatorius placed much closer to proximal end of the aedeagus in the male genitalia. Additionally, basal sclerite of sternum VIII in A. ciliatella is more prolonged that this of A. labeculella ( Figs 96, 97 View FIGURES 93–99 ) and A. yerburii ( Figs 98, 99 View FIGURES 93–99 ). In females of A. yerburii thorn-like signae are larger and less numerous ( Fig. 102 View FIGURES 100–102 ). In A. labeculella signae are grouped in one rather than in two clusters, and ductus bursae is broader with gradual transition to corpus bursae ( Figs 103, 104 View FIGURES 103, 104 ). Ancylosis pectinatella is an insufficiently known species. The examined male lectotype from Alai differs from A. ciliatella in broader uniformly light brown forewing ( Fig. 82 View FIGURES 78–92 ). The male genitalia ( Figs 94, 95 View FIGURES 93–99 ) are very similar to A. ciliatella but ductus ejaculatorius is closer to base of aedegus. Ancylosis albicosta has somewhat similar valva (with well-defined broad sacculus), but it is of equal width without medial broadening and transtilla lobes are shorter.
Redescription. Adult ( Figs 78–81 View FIGURES 78–92 ). Wingspan 19–21 mm. Head smooth. Frons widely convex. Proboscis well developed. Labial palps nearly 1.3× diameter of eye, pointed up. First segment bended, second segment twice as long as the first and of the same width. Third segment very small, ovate, it is 1/5–1/6 of the second segment in length, and 1/3 of the latter in width. Maxillary palps extremely small with ovate segments. Antennae about 3/5 of the forewing in length, scape almost ovate with its wide part approximately 1.5× as long as wide. Flagellum without basal sinus. Cilia rather long in males (2–2.5× of flagellomere diameter) and shorter (1/2) in females. Frons white, vertex whitish-creamy or yellowish-creamy, labial palps white, often with creamy elongate area on the outer surface, maxillary palps in short white scales, antennae whitish, whitish-creamy or brown. Tegulae and patagia varying in colouration: light brown, creamy, yellowish-creamy or magenta. Abdominal terga yellowish-creamy or greyish, sterna whitish. Legs whitish, partly yellowish-brown. Forewing elongate triangular with slightly convex termen; costa almost straight, convex apically, as a rule. Ground colour of the forewing can be ash grey, brownish-grey, brownish-creamy, grey with pink tinge or magenta. Sometimes whitish suffusion expressed. White costal strip with straight edges, smooth and usually sharply pointed in apical 1/3, much varying in width. Costal strip fading in distal 1/ 2 in some cases. A small streak-like white marking present along the hind margin basally. Fringe grey, creamy or grey with pink tinge. Underside of the forewing brown with narrow white costal strip and whitish area along the hind margin, occupying 1/3–1/2 of the wing. Hindwing varying from light brown to whitish with darkened (light brown or yellowish-brown) marginal area. Fringe whitish or light brown, usually with darker brown basal 1/3. Underside of the hindwing light brown or whitish-brown.
Female of A. ciliatella . Females of A. ciliatella have no external differences from males except for much shorter cilia, which is nearly 1/3 of flagellomere. Like in males, ground colour of the forewing is ash grey with more or less extensive pink tinge.
Male genitalia ( Fig. 93 View FIGURES 93–99 ). Uncus twice as long as borad, parallel-sided to 2/3, then narrowed towards rounded apex. Distal sclerite of gnathos short, triangular, pointed, extending to 1/5 length of uncus. Tuba analis subrectangular, with transverse folds before middle, extending to 3/4 length of uncus. Tegumen slightly longer than uncus, broadly constricted in distal portion. Vinculum band-shaped, saccus subrectangular, twice as long as broad, apex weakly emarginated in middle. Juxta long, subrectangular, lobes with distinctly pointed apex. Transtilla lobes long, narrow, curved inwards, connected medially. Valva broad, with rounded apex; dorsal margin straight with short medial hump; costa moderately broad, extending nearly to the top of valva; ventral margin distinctly bended in middle; sacculus weakly sclerotised, about 1/3 width of valva, extending to 1/2 length of valva; distinct narrow oblique medial fold from the base of costa to mid of dorsal margin. Aedeagus as long as costa of valva, basal 1/4–1/3 narrow, then broadened, distal half narrow, parallel-sided.
Tergum VIII rounded, stronger sclerotized in posterior portion, anterior margin with deep, narrow, triangular medial emargination; sternum VIII strongly sclerotized laterally, subtrapezoidal, anterior margin straight, posterior margin with medial triangular hump, lateral tufts of scales distinctly longer than the length of sternum VIII.
Female genitalia ( Fig. 100 View FIGURES 100–102 ). Papillae anales elongate, slightly tapering to apex, densely covered with bristles. Posterior apophyses rather thin, about 3/2 of papillae anales and nearly equal to anterior apophyses.Anterior apophyses some more stout, angulate and broadened at the base. Posterior margin of tergum VIII with deep V-shaped membranous hollow, anterior margin convex. Antrum membranous conical with small curved sclerite located anteriorly. The sclerite varies in shape, it is more often petal like. Ductus bursae very thin and short. Ductus seminalis arises from nearly 1/2 of ductus bursae. Corpus bursae pear-shaped with elongate posterior part. Thorn like signae form two clusters of usually rounded shape on the surface of corpus bursae. Clusters are located laterally and connected with each other. A group of signae is present as a continuation of two clusters in posterior part of corpus bursae. Each signa is on a weakly sclerotized rounded base. Signae are variable in size, the smaller ones are located at the edges of the two clusters.
Remarks. Myelois ciliatella was described from a single male from Sarepta. We studied six males and six females from South of European Russia and Kazakhstan which match externally to the photograph of holotype in all details and, hence, are conspecific with latter. The study of the genitalia of these specimens justified A. ciliatella a separate species, which genitalia differ considerably from those of A. rhodochrella . We therefore reinstate A. ciliatella as valid species: A. ciliatella ( Zeller, 1872) spec. rev., stat. nov.
Ancylosis pectinatella was described from unspecified number of specimens from Margilan ( Uzbekistan). Roesler (1973: 419) described in detail the specimen from MfN labelled “ Heterographis pectinatella Rag , type, orig., pl. XXVIII, fig. 16” collected in “Alai” (not in Margilan as it states in the original description) and indicated it as a “Type”. According to ICZN (1999: article 74.6) this specimen is considered as a lectotype. The male genitalia of A. pectinatella are extremely similar to those of A. ciliatella (see above for details). However, taking in account the external differences in wing pattern and in the shape of the forewing we suggest A. pectinatella as a separate species until additional material from type locality including females becomes available. We studied additional specimens from Tunisia identified as A. pectinatella . They differ externally from the lectotype by light grey mixed with dark brown forewing ( Figs 83, 84 View FIGURES 78–92 ). The male genitalia are nearly indistinguishable from those of A. pectinatella from Alai except for valva that is stronger narrowed apically in the latter ( Fig. 95 View FIGURES 93–99 ).
Ancylosis labeculella was described from Gabès, Tunisia. Roesler (1973) considered A. labeculella as a synonym of A. yerburii . Leraut (2014: 415) re-instated A. labeculella as a separate species. The current status of A. labeculella remains uncertain as type specimens are unrevised. We examined two males and three females from Tunisia ( Figs 85, 86 View FIGURES 78–92 ) which match externally to specimens of A. labeculella figured by Leraut (2014: pl. 65, figs 8–10). These specimens are undoubtedly closely related in the genitalia to A. yerburii , A. ciliatella and A. pectinatella , though valva in A. ciliatella and A. pectinatella is broader in the middle and ventral sclerites near aedeagus are present. Additional differences in the shape of the aedeagus are discussed above. For the differences from A. yerburii see below under that species. Signae grouped in one rather than in two clusters ( Figs 103, 104 View FIGURES 103, 104 ) seem the most constant character for separating female genitalia of A. labeculella from related species.
Distribution. The species is known with certainty from Russia (Volgograd and Astrakhan regions) and Kazakhstan (Mangistau and Turkestan regions). Records from other regions ( Roesler 1973 (as A. rhodochrella )) need verification.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |