Quedius masuzoi Watanabe, 1989
|
publication ID |
https://doi.org/10.5852/ejt.2020.664 |
|
publication LSID |
lsid:zoobank.org:pub:C3B7A472-8052-46CF-BF62-755FEDDC33AE |
|
persistent identifier |
https://treatment.plazi.org/id/BE518788-FF80-FFF7-195F-FAEFFE28FCB2 |
|
treatment provided by |
Valdenar |
|
scientific name |
Quedius masuzoi Watanabe, 1989 |
| status |
|
Quedius masuzoi Watanabe, 1989 View in CoL
Figs 2 View Fig , 3 View Fig A–B, D–E, G–H, 4 View Fig A, 9 View Fig B (adult); 5 View Fig – 8 View Fig , 9 View Fig C (larva)
Quedius masuzoi Watanabe, 1989: 170 View in CoL , figs 1–4 (description; type locality: Taiwan, Nan-t’ou Hsien, Mt Neng-kao-pei-feng [= 能 高北 峰 = 南華山 = Nanhua Mt], alt. 2870 m).
Quedius chiangi Smetana, 1995: 48 , fig. 54 (description). Syn. nov.
Quedius masuzoi View in CoL – Smetana 1995: 45, figs 49–53 (redescription).
Diagnosis (adult)
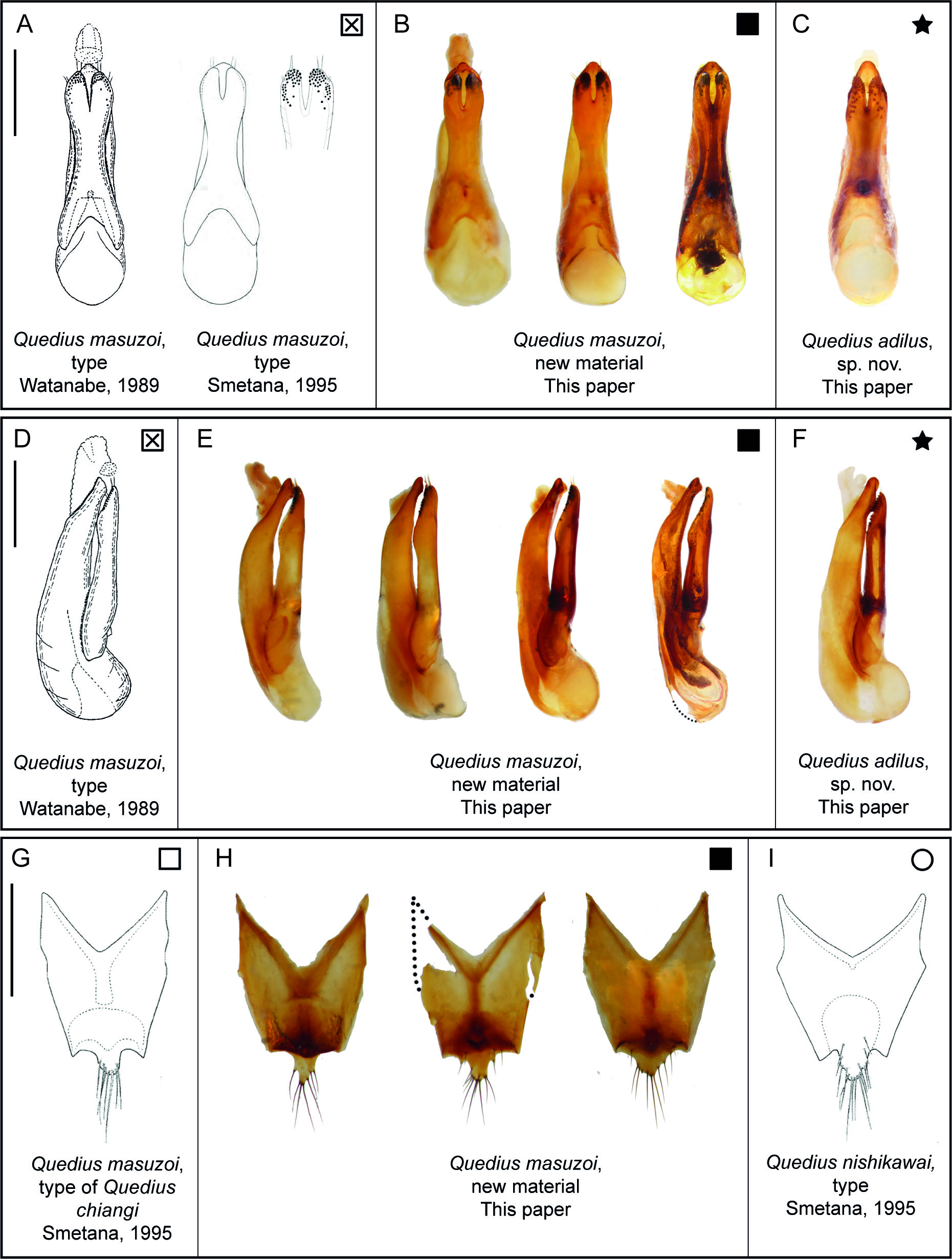
Quedius masuzoi ( Fig. 4A View Fig ) is very similar to the two other species of the abnormalis group in Taiwan. From Q. nishikawai it can be distinguished by the shorter antenna and sclerotization pattern of tergite X, which has a distinctly heavier sclerotized stalk connecting its more sclerotized apical and basal areas (compare Figs 3G, H View Fig and 4I View Fig ). From Q. adilus sp. nov. it can be distinguished by the shorter and wider median lobe of the aedeagus, which is only slightly protruding over the apical margin of the bilobed paramere (in parameral view) (compare Fig. 3 View Fig A–B and 3C).
Diagnosis (larva)
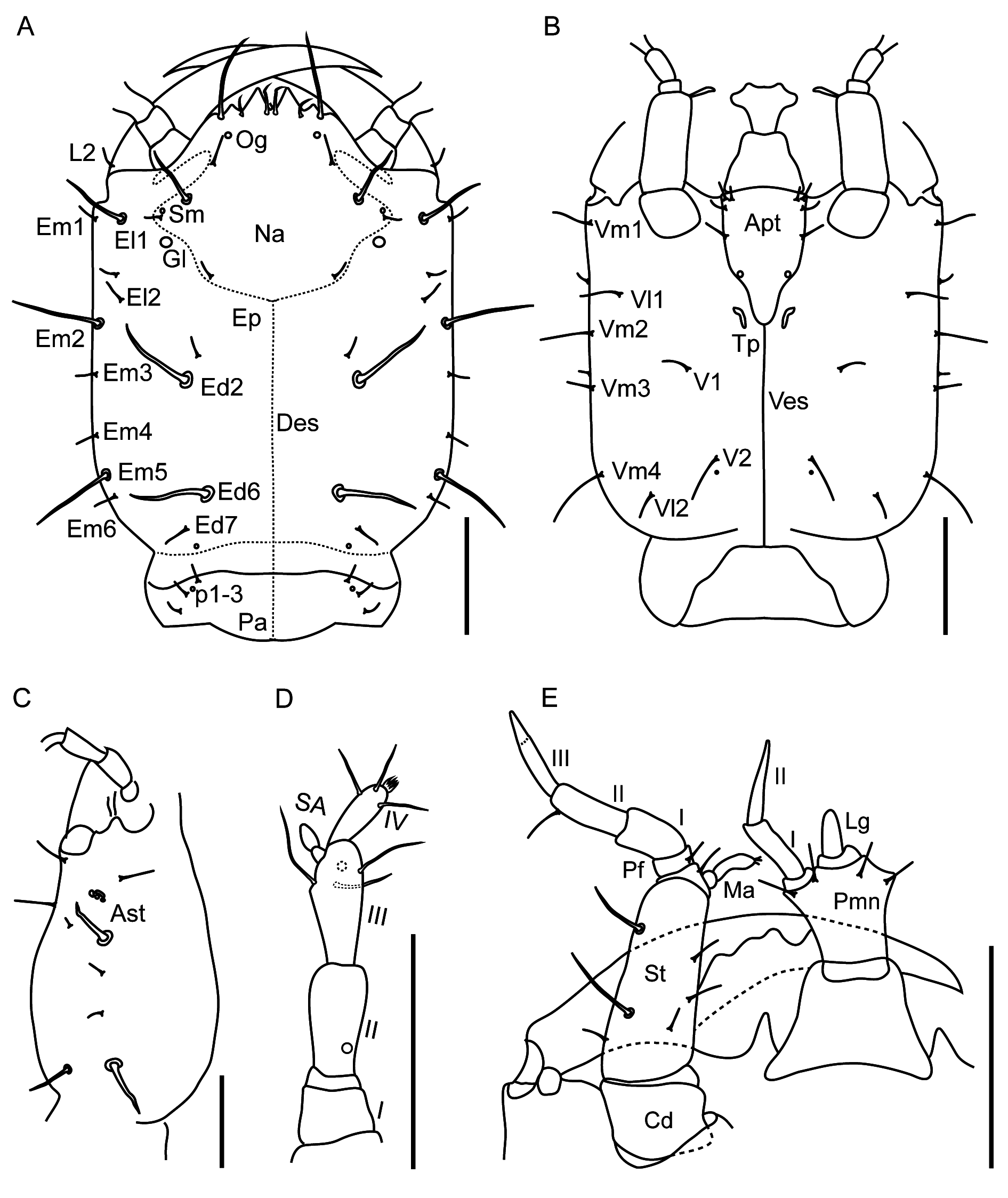
Head capsule parallel-sided, not narrowing anteriad towards bases of mandibles, with four weakly visible white stemmata on each side, without dark pigment spots inside them; frayed setae present from metathorax to apex of abdomen; apotome with short stalk not extending beyond tentorial pits ( Fig. 6B View Fig ); inner margin of mandible without small tooth ( Fig. 6A, E View Fig ); maxillary and labial palp 3- and 2-segmented, respectively, last segment of maxillary palp longer than penultimate segment, weakly divided into two pseudosegments ( Fig. 6E View Fig ); protibial comb with irregular row of 8–10 split (bifid) spines ( Fig. 7C View Fig ).
Material examined (all adults unless indicated as larvae)
Holotype (of Quedius chiangi )
TAIWAN • ♀; Nantou Hsien, Yushan Natonal Park, Pai-Yun Hut [ 玉山 排 雲山莊]; alt. 3528 m; 15 May 1991; “ A. Smetana [T83] / HOLOTYPE ”; “ Quedius chiangi A. Smetana 1993 [red label, hand writing]”; NMNS.
Other material
TAIWAN • 1 ♂, 1 ♀; Chiayi Co., Shi Mt race [ 石山引 水道], Alishan To ; 23.4691° N, 120.8583° E; alt. 2360 m; 11 Feb. 2019; Y.T. Lai leg.; subterranean water diversion construction; NHMD GoogleMaps • 1 ♂; same collection data as for preceding; 23 Feb. 2019; F.S. Hu leg.; FSHc GoogleMaps • 1 ♂; same collection data as for preceding; DNA voucher specimen FS001 (GenBank: MN989304 View Materials ); NMNS GoogleMaps • 1 ♀; same collection data as for preceding; 11 Feb. 2019; Y.T. Lai leg.; DNA voucher specimen FS002 (GenBank: MN989305 View Materials ); FSHc GoogleMaps • 1 ♀; same collection data as for preceding; 3 Feb. 2019; Y.T. Lai leg.; DNA voucher specimen FS003 (GenBank: MN989306 View Materials ); FSHc GoogleMaps • 1 ♀; same collection data as for preceding; 23 Feb. 2019; B.H. Ho leg.; DNA voucher specimen FS004 (GenBank: MN989307 View Materials ); NMNS GoogleMaps • 1 ♂; same collection data as for preceding; F.S. Hu leg.; DNA voucher specimen FS005; NMNS GoogleMaps • 1 ♂; same collection data as for preceding; 24 Feb. 2019; F.S. Hu leg.; DNA voucher specimen FS006; NMNS GoogleMaps • 1 ♀; same collection data as for preceding; DNA voucher specimen FS007; NMNS GoogleMaps • 1 ♂; same collection data as for preceding; 21 Feb. 2020; W.Z. Tseng and H.Y. Lin leg; FSHc GoogleMaps • 2 larvae; same collection data as for preceding; 23 Feb. 2019; F.S. Hu leg.; DNA voucher specimens FS008L (GenBank: MN989308 View Materials ), FS009L [2 specimens in different vials]; FSHc GoogleMaps • 2 larvae; same collection data as for preceding; DNA voucher specimens FS010L (GenBank: MN989309 View Materials ), FS011L [2 specimens in different vials]; NHMD GoogleMaps .
Description (adult)
Male
See Watanabe (1989) and Smetana (1995).
Female
See original description of Q. chiangi in Smetana (1995) .
Character variability and new synonymy
Smetana (1995) described Q. chiangi as a species very similar to Q. masuzoi , but differing from the latter in the position of the posterior frontal puncture and the number of its additional setiferous punctures. Smetana (1995) expressed the position of the frontal puncture as a ratio of the distances between that and the hind margin of the head to the distance between the frontal puncture and the hind margin of the eye. According to this, the posterior frontal puncture is positioned at a ratio of no more than 1.5 in Q. masuzoi , whereas in Q. chiangi this ratio is roughly 3.5, which in effect means that this puncture in Q. chiangi is positioned much closer to the hind margin of the eye. Furthermore, Smetana (1995) mentioned one additional setiferous puncture posteriad of the posterior frontal puncture in Q. masuzoi and two to three punctures postero-mediad of the posterior frontal puncture in Q. chiangi .
Our new material with the sequenced CO1 barcode, however, shows that the mentioned traits are quite variable even within our sample of specimens from the same locality with identical barcodes. We found that the position of the posterior frontal puncture, expressed in the same ratio as Smetana (1995), varies in our sample between 1.46 and 4.90, meaning that the intraspecific variability range encompasses the differences reported as interspecific by Smetana (1995). One of our specimens was completely missing the posterior frontal puncture on one side. Remarkably, the position and number of additional setiferous punctures near the posterior pronotal puncture also vary in our specimens, ranging from missing through having one anterio-mediad to having multiple posterio-mediad.
We also found additional overlap in other ratio values and characters mentioned as differences between the two species by Smetana (1995). Specifically, the head width to length ratio varies in our conspecific material from 1.14 to 1.36 ( Q. masuzoi 1.14 and Q. chiangi 1.10 in Smetana 1995). The ratio of length of temples to length of eyes varies from 4.27 to 7.33 ( Q. masuzoi 4.73 and Q. chiangi 4.58 in Smetana 1995). The ratio of length to width of antennomere 4 varies from 0.75 to 1.6 ( Q. masuzoi 1.09 and Q. chiangi > 1.09 in Smetana 1995). The scutellum varies in the degree of punctation, with one specimen having it totally glabrous.
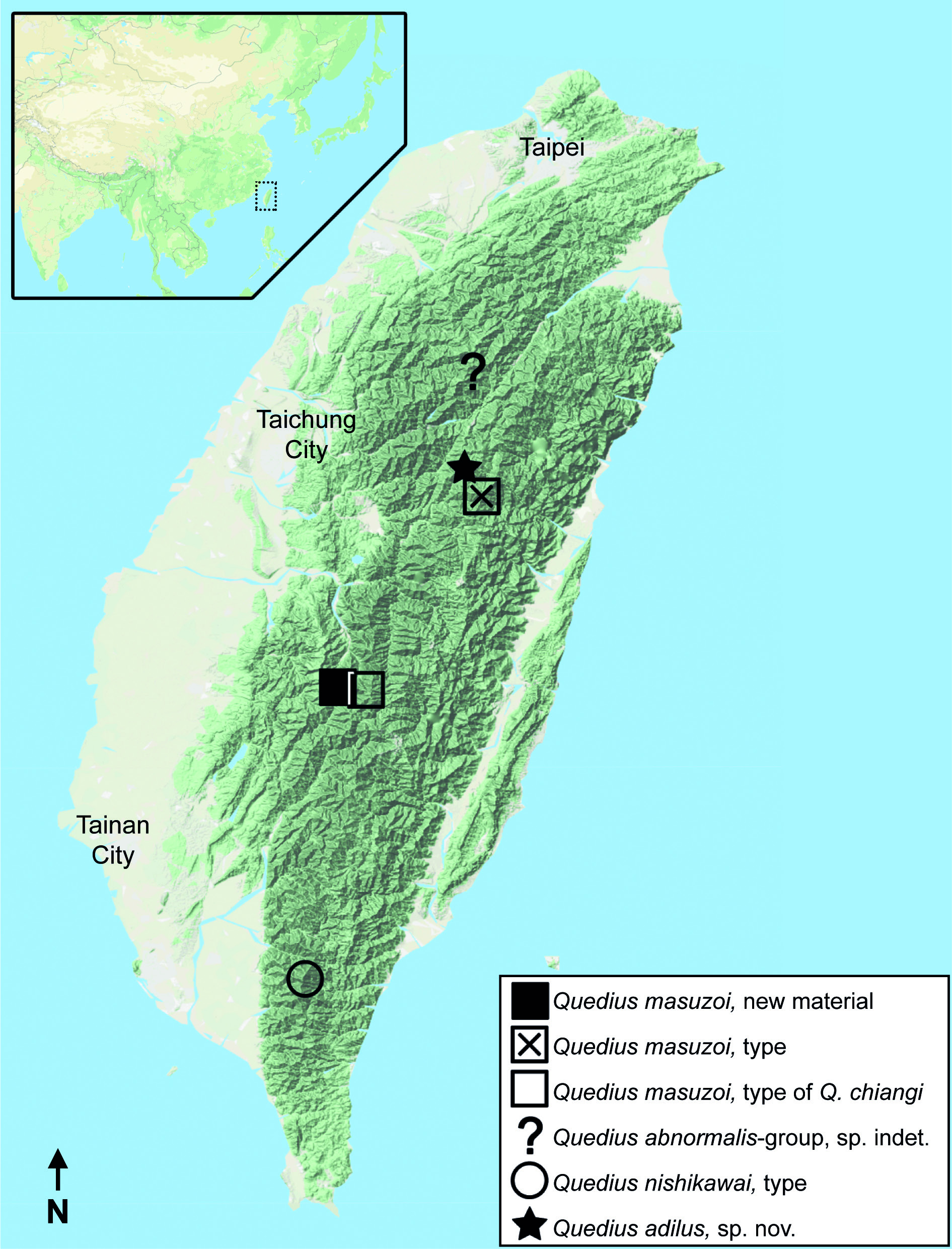
Noteworthy is that the female tergite X of Q. chiangi illustrated in Smetana (1995) has the same sclerotization pattern as in all females of Q. masuzoi that we examined, namely a heavier sclerotized apical area with a similar darker stalk connecting this area with the base of the tergite ( Fig. 3 View Fig G–H). Also, the geographic proximity of the type locality of Q. chiangi to the locality where additional material of Q. masuzoi was collected ( Fig. 2 View Fig ) is noteworthy.
Examination of the holotype of Q. chiangi confirmed our thoughts, with all characters fitting within the explained variability, except for its size being slightly smaller. Based on all these considerations, we place Q. chiangi in synonymy with Q. masuzoi .
Description (larva)
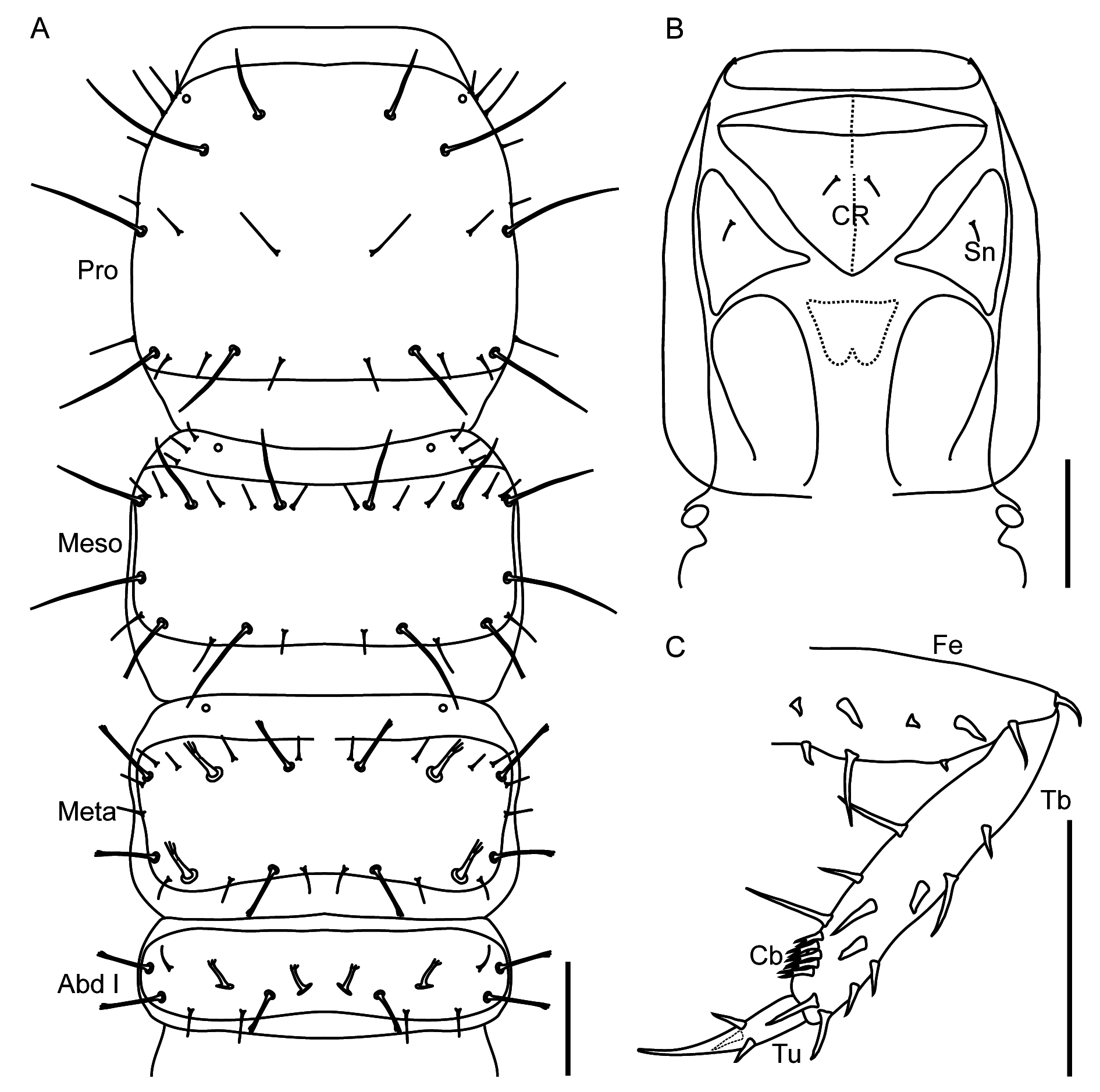
The larva of Q. masuzoi ( Fig. 5 View Fig ) possesses all the characters of the subfamily Staphylininae : toothed nasale ( Fig. 6A View Fig ); non-setose ligula ( Fig. 6E View Fig ); eyes of four stemmata each ( Fig. 6C View Fig ); anterior position of sensory appendage on antennal segment III ( Fig. 6D View Fig ); triangular cervicosternum with its apex projecting between proepisterna ( Fig. 7B View Fig ); and tarsungulus with three spines ( Fig. 7C View Fig ). It has 2-segmented labial palps and 3-segmented maxillary palps ( Fig. 6E View Fig ), relatively short urogomphi (about as long as pygopod) ( Fig. 9C View Fig ), frayed setae on the body ( Figs 7A View Fig , 8 View Fig ) and combs of split (bifid) spines on anterior tibia ( Fig. 7C View Fig ), as in other Quediini . Overall, it fits the larval diagnosis of the genus Quedius given in Pietrykowska-Tudruj et al. (2014), which, however, was based on limited material. Based on the data in Pietrykowska-Tudruj et al. (2014) and our experience with larvae of other Staphylininae , we have above provided a diagnosis which should facilitate the separation of larvae of Q. masuzoi and related hypogean species from those of other Quedius or Quediini , supplemented by the following description.
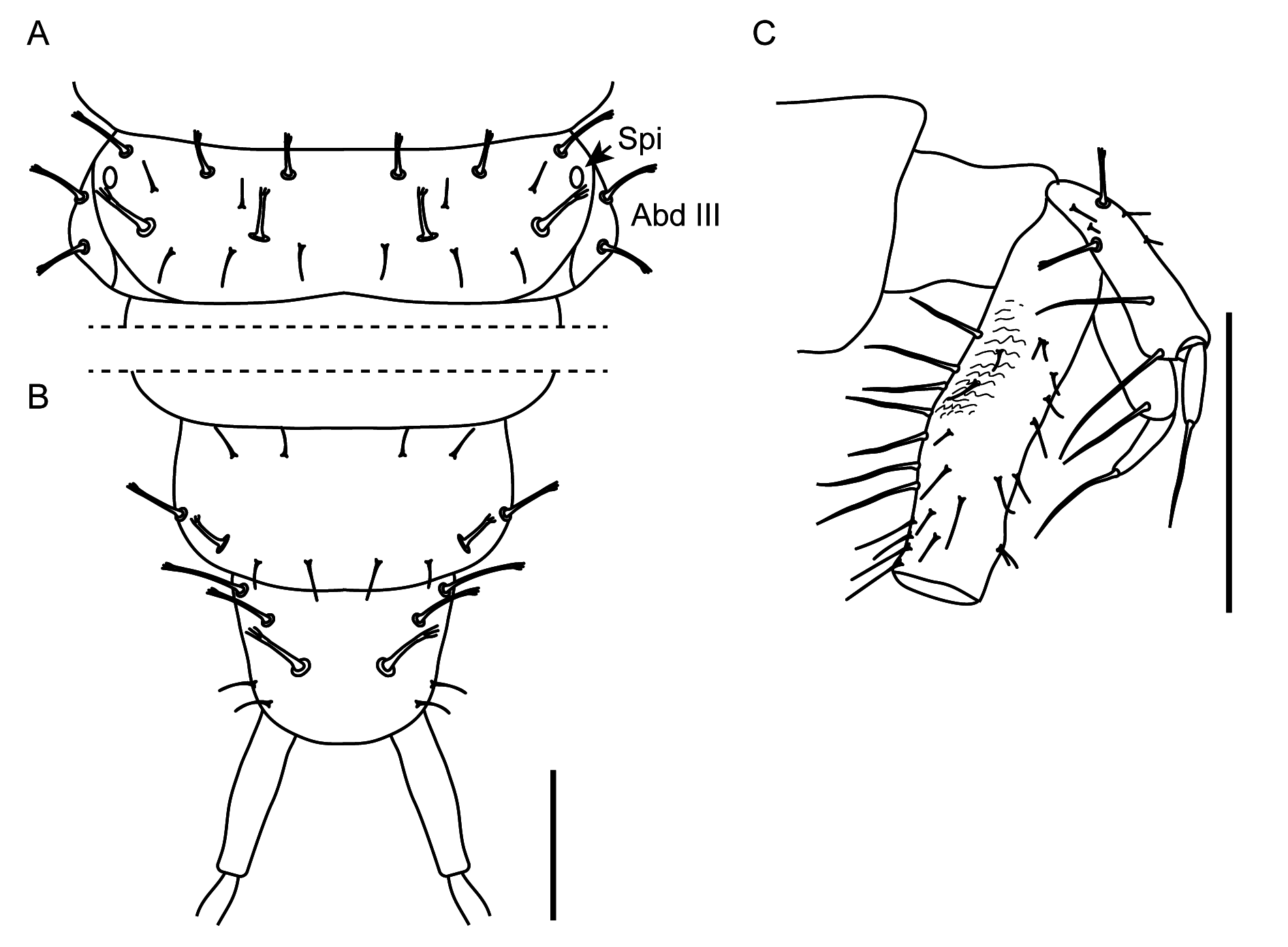
GENERAL. Body length 10–11 mm; head length 1.9–2.1 mm, width 1.5–1.6 mm; pronotum length 1.8– 1.9 mm, width 1.5–1.7 mm. Body slender, with relatively large head and long legs, prothorax as wide as parallel-sided head, meso- and metathorax, as well as basal four segments of abdomen; from segment IV abdomen gradually narrowing posteriad. Habitus as in Fig. 5 View Fig , head capsule brownish, protergum, thorax and legs pale brown, abdomen whitish-grey, setae brown to dark brown. All macro- and microsetae of head, pro- and mesothorax and some setae on abdominal segments simple; few macrosetae on metathorax, the majority of those on abdomen, except pygopod, apically frayed ( Figs 6–8 View Fig View Fig View Fig ).
HEAD. With broad but distinct neck, slightly longer than wide ( Fig. 6 View Fig A–B). Chaetotaxy of dorsal side of head as in Fig. 6A View Fig , as a pattern of larger and smaller simple setae, with distinct pair of epicranial glands (Gl), and poorly visible small olfactory organs (Og) and sensilla (Sm) on nasale. Four pale stemmata (Ast) without pigment spots, in cluster on each side ( Fig. 6C View Fig ). Ventral side of head with chaetotaxy as in Fig 6B View Fig , with clearly visible tentorial pits (Tp) extended along short stalk of apotome (Apt). Antenna ( Fig. 6D View Fig ) 4-segmented, slender, segment III longer than segment II; segment II with one large ‘pore’ dorso-laterally; segment III with three macrosetae, large apically pointed sensory appendage (SA) and one ‘pore’ (ventrolateral); segment IV with three subapical macrosetae and a group of small setae and four solenidia apically. Nasale (Na) with nine teeth at apical margin, with pair of paramedian teeth being the longest. Mandibles ( Fig. 6A View Fig ) slender, apically very sharp, with one large seta externally at base. Stipes of maxilla ( Fig. 6E View Fig ) with several macrosetae, two of them distinctly larger than others. Mala (Ma) finger-like, ca three times as long as wide at base ( Fig. 6E View Fig ). Palpifer (Pf) with one seta ventrally. Maxillary palp 3-segmented, its apical segment the longest, weakly subdivided into two pseudosegments. Labium ( Fig. 6E View Fig ) with well-sclerotized ventral side of prementum (Pmn), with four setae apically. Ligula (Lg) conical, ca two times as long as wide. Labial palps 2-segmented; segment II distinctly thinner and longer than I.
THORAX. Prothorax as long as wide, meso- and metathorax wider than long, of similar structure, metathorax slightly shorter than mesothorax; protergite with posterior carina, meso- and metatergite with anterior and posterior carinae ( Fig. 7A View Fig ). Pronotum and mesonotum with simple (not frayed) setae only; metanotum with larger setae frayed, smaller setae simple. Pro-, meso- and metanotum with smooth, glossy cuticle. Cervicosternum (CR) large, triangular, with pair of setae medially. Sternites of prosternal area (Sn) each with one seta ( Fig. 7B View Fig ).
LEGS. Relatively long, all of about the same length; femora with strong, long spines ventrally, tibia with similar spines dorsally and ventrally; tibial comb (Cb) present on anterior legs only, consists of up to ten split (bifid) spines; rather long, apically very sharp tarsungulus (Tu) with three spines ( Fig. 7C View Fig ).
ABDOMEN. Ten-segmented; only segment I with visible brownish glossy tergite and with distinct anterior carina; other segments without visible darker sclerites, whitish-grey. All abdominal segments with frayed setae, large and small, and with some simple small setae. Tergal chaetotaxy of segment I ( Fig. 7A View Fig ) simpler than on segments II–VIII (e.g., Fig. 8A View Fig of segment III), the latter having the same serially homologous pattern. Segment IX with reduced chaetotaxy ( Fig. 8B View Fig ), without laterosclerites and ecdysial line. Segment X (pygopod) with dense, long simple setae ventrally and short frayed setae dorsally, slightly longer than short urogomphi (measured without apical seta), the latter with few simple, frayed macrosetae, each with one long, frayed apical macroseta ( Fig. 8C View Fig ).
Note
The lack of notable pigmentation of the stemmata and the relatively long legs are apparently adaptations to the hypogean environment during the larval stage.
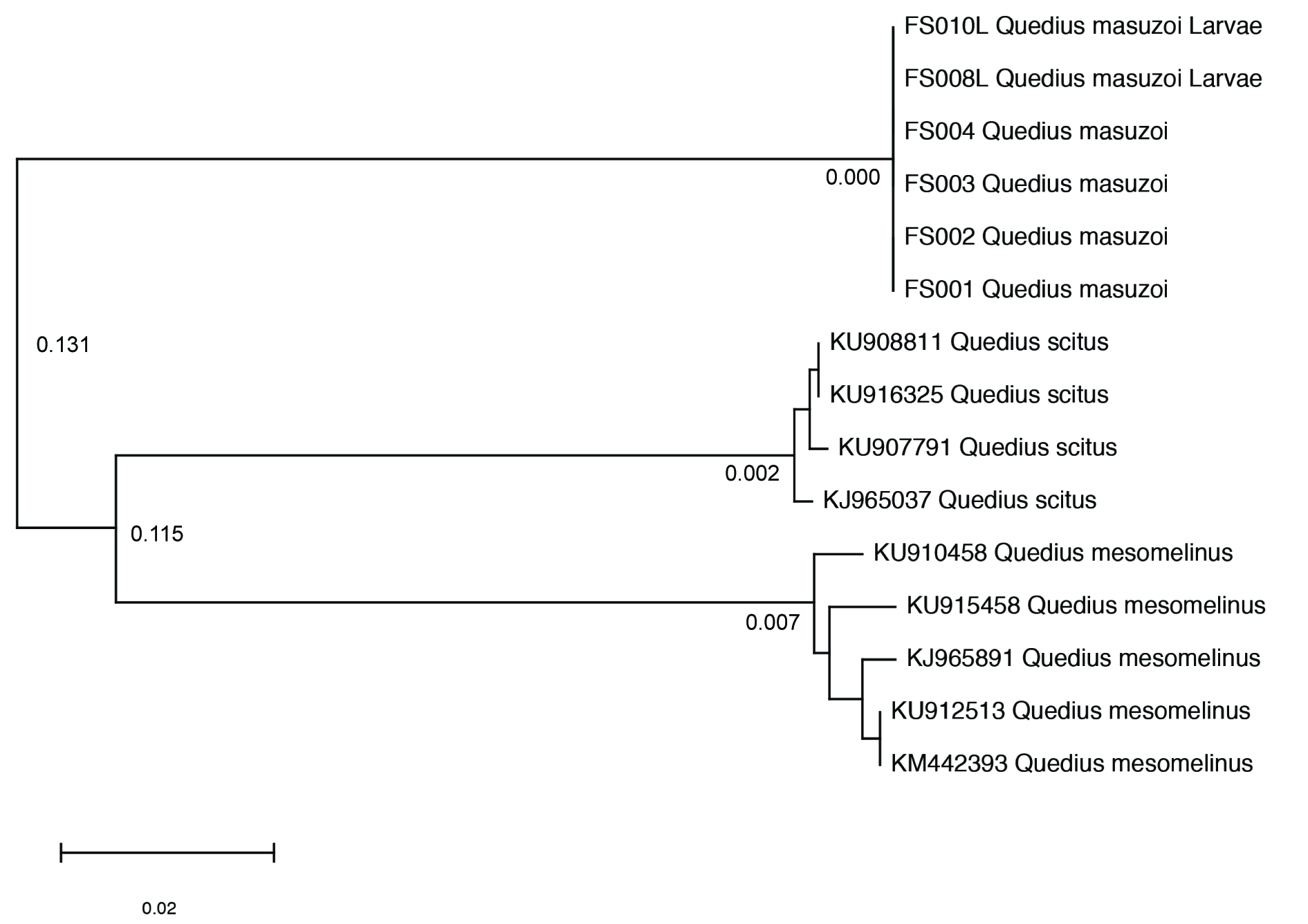
Molecular results and identity of the new material
We sequenced the CO1 barcode for six specimens (four adults and two larvae) of the putative Q. masuzoi from the same locality, a subterranean water diversion channel at Mt Shi ( Fig. 9 View Fig ). All larvae and adults showed matching CO1 barcodes, supporting the idea that they are conspecific. There were no ambiguous sites within the six barcoded individuals, and thus the dissimilarity between them equals 0.000 ( Fig. 10 View Fig ). The two species, Q. mesomelinus and Q. scitus , used as outgroup, showed a 0.131 dissimilarity to our samples of Q. masuzoi and a dissimilarity of 0.115 between each other. They also showed larger within species dissimilarity compared to Q. masuzoi ( 0.007 in Q. mesomelinus and 0.002 in Q. scitus ). The larger within species dissimilarity in the outgroup species is probably due to the larger geographic range of the sampled specimens. All our putative Q. masuzoi samples were from the same site and collecting event, whereas samples for the outgroup species from GenBank come from multiple localities which are geographically remote from one another. Aedeagi were identical in all five males in the sample of our newly collected material and fully match the illustrations of the aedeagus of Q. masuzoi in Watanabe (1989) and Smetana (1995), reliably identifying our specimens as that species ( Fig. 3 View Fig A–B, D–E).
Distribution
This species is known from three localities: from Mt Neng-kao-pei-feng in northeastern Taiwan ( type locality of Q. masuzoi ), as well as from Pai-Yun Hut, Yushan National Park ( type locality of Q. chiangi ) and nearby Mt Shi (newly collected material) in the central part of the island (i.e., from Nantou and Chiayi Counties) ( Fig. 2 View Fig ).
Bionomics
The holotype of Q. masuzoi was dug out from loose rocks with poor vegetation ( Watanabe 1989). For Q. chiangi , the holotype was collected from an old rocky wall, while the collecting circumstances of the paratype remain unknown ( Smetana 1995). The additional specimens of Q. masuzoi reported here were collected from a short subterranean tunnel ( Fig. 9A View Fig ) close to the type locality of Q. chiangi . The rocky walls of the tunnel were covered by bat guano and its entrance opens to a primary forest. Adults were observed both on the surface of the ground and under the rocks of the tunnel ( Fig. 9B View Fig ). Larvae ( Fig. 9C View Fig ) were collected under rocks in the same tunnel, during the same collecting event. Leiodidae spp, other rove beetles belonging to Xantholinini (all Coleoptera ), Rhaphidophoridae sp. ( Orthoptera ) and Nycteribiidae sp. ( Diptera ) were found in the same tunnel. The collected adults and larvae of Q. masuzoi were kept in the laboratory for over two weeks, where they were fed with both live and dead Rhaphidophoridae sp., Blatta lateralis Walker, 1868 (Blattodea) and Odontotermes formosanus (Shiraki, 1909) (Blattodea) . Despite being supplied with live prey, both adults and larvae fed on dead prey only, i.e. they did not display predatory behaviour in the laboratory. Unfortunately, we did not rear any adults from the collected larvae and no other significant observations on their behaviour were made.
Remarks
The holotype of Q. chiangi is in good condition, mounted on a card. Segment 3–4 of the left maxillary palpus, antennomeres 4–11 of left antenna, the left fore claw, and the left hind tibia and femur are missing. The genital segment was dissected and sealed using Euparal, placed on a plastic card pinned underneath the paper card.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Staphylininae |
|
Tribe |
Quediini |
|
Genus |
|
|
SubGenus |
Microsaurus |
Quedius masuzoi Watanabe, 1989
| Hu, Fang-Shuo, Bogri, Amalia, Solodovnikov, Alexey & Hansen, Aslak Kappel 2020 |
Quedius chiangi
| Smetana A. 1995: 48 |
Quedius masuzoi
| Smetana A. 1995: 45 |
Quedius masuzoi Watanabe, 1989: 170
| Watanabe Y. 1989: 170 |