Scorpiops lowei, Tang, 2022
|
publication ID |
https://doi.org/ 10.5281/zenodo.7300502 |
|
publication LSID |
lsid:zoobank.org:pub:343250AD-5602-440C-92C3-9FED8EFB77BE |
|
persistent identifier |
https://treatment.plazi.org/id/AB38D259-34A3-4302-BC1B-867B85D6A656 |
|
taxon LSID |
lsid:zoobank.org:act:AB38D259-34A3-4302-BC1B-867B85D6A656 |
|
treatment provided by |
Felipe |
|
scientific name |
Scorpiops lowei |
| status |
sp. nov. |
Scorpiops lowei sp. n.
( Figures 12–84 View Figure 11–18 View Figures 19–22 View Figures 23–24 View Figures 25–26 View Figures 27–30 View Figures 31–32 View Figures 33–45 View Figures 46–58 View Figures 59–66 View Figures 67–74 View Figures 75–76 View Figure 77–83 View Figure 84 , Tables 1–3) http://zoobank.org/urn:lsid:zoobank.org:act:AB38D259-
34A3-4302-BC1B-867B85D6A656
Euscorpiops kubani (misidentification): Di et al., 2011: 1, 5–11, 13–15, 20–21, 23–24, 28, 30–31; Ythier, 2019: 190, 195 (fig. 11) (listed as a species that occurs in China); Di & Qiao, 2020a: 72, 80, 81.
Scorpiops kubani (misidentification): Kovařík et al., 2020: 127, fig. 799 (listed as a species that occurs in China); Tang, 2022a: 77 (listed as a species that occurs in China, marked by the symbol ☆); Lourenço & Ythier, 2022: 3, 6 (listed as a species that occurs in China); Tang, 2022b: 4 (fig. 2), 14 (listed as a species that occurs in China); Tang, 2022c: 3, 5, 13–16, 21, 24, 30–32, 34, 39–43.
TYPE LOCALITY AND TYPE REPOSITORY. China, Yunnan Province, Xishuangbanna Prefecture, Menghai County, Menghai Town , 21°57'35"N 100°27'49"E, 1183 m a. s. l; VT GoogleMaps .
TYPE MATERIAL. Holotype: China, Yunnan Province, Xishuangbanna Prefecture, Menghai County, Menghai Town , 21°57’35”N 100°27’49”E, 1183 m a. s. l, 15 August 2022, 1♂, leg. Hang Qiu, VT; paratypes: 1♂ 2♀ (including allotype ♀), same data as the holotype; Menghai Town , 21°58’18”N 100°27’44”E, 1193 m a. s. l, 21 July 2022, 7♂ 8♀ leg. Hang Qiu, VT (for figures and data, see Tang, 2022c; misidentified as S. kubani ). GoogleMaps
ETYMOLOGY. The specific epithet is named in honor of Dr. Graeme Lowe, a patient, kind and knowledgeable scorpiologist who offered plentiful assistance both in research techniques and scorpiology to the author.
Chinese equivalent. 洛氏ŔH (roughly as “Lowe’s resemblant scorpion” in English; see Tang (2022a) for the rules of designation).
DIAGNOSIS (based on all 19 specimens studied). Total length 37.9–46.0 mm in males and 39.7–50.9 mm in females. Base color uniformly reddish brown to brownish black, telson brown to brownish yellow and aculeus reddish with tip black. Pectinal teeth number 7–8 in males and 5–7 in females; fulcra present; marginal lamellae I and III delimited, sulcus that usually separates the marginal lamella II from the middle lamella often incomplete. Patella of pedipalp with 17–20 external and 8–10 ventral trichobothria. Chela of pedipalp with 4 V series trichobothria located on ventral surface. Chelal trichobothrium Eb 3 located in distal half of manus between trichobothria Dt and Est. Dorsal edge of movable fingers of pedipalps undulate in males, weakly undulate in females. Chela length to width ratio 3.24–3.59 in males, 3.05–3.45 in females. Pedipalp movable finger with ca. 45–59 IAD, which have the same size as MD (ca. 76–92 in number) and create a second row of denticles; there are also 4–8 ID and 14–16 OD present. Tarsomere II of leg III with 5–6 stout median ventral spinules in a single row, and two pairs of flanking setae. Metasoma I with 10 carinae, metasoma II–IV with 8 carinae. Telson elongate and very finely granulate, length to depth ratio 3.21–3.45 in males, 3.34–3.74 in females; annular ring developed in both sexes.
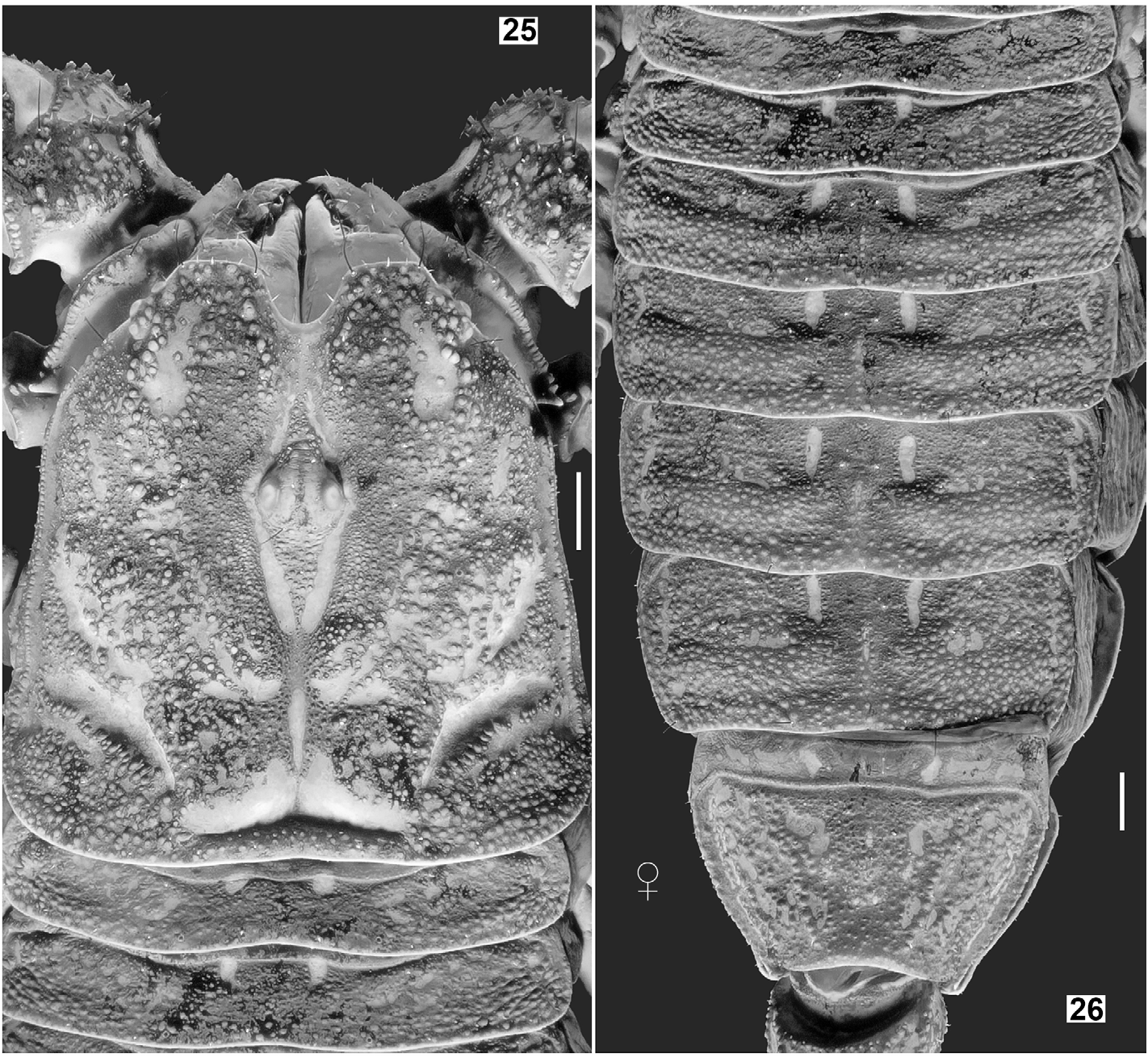
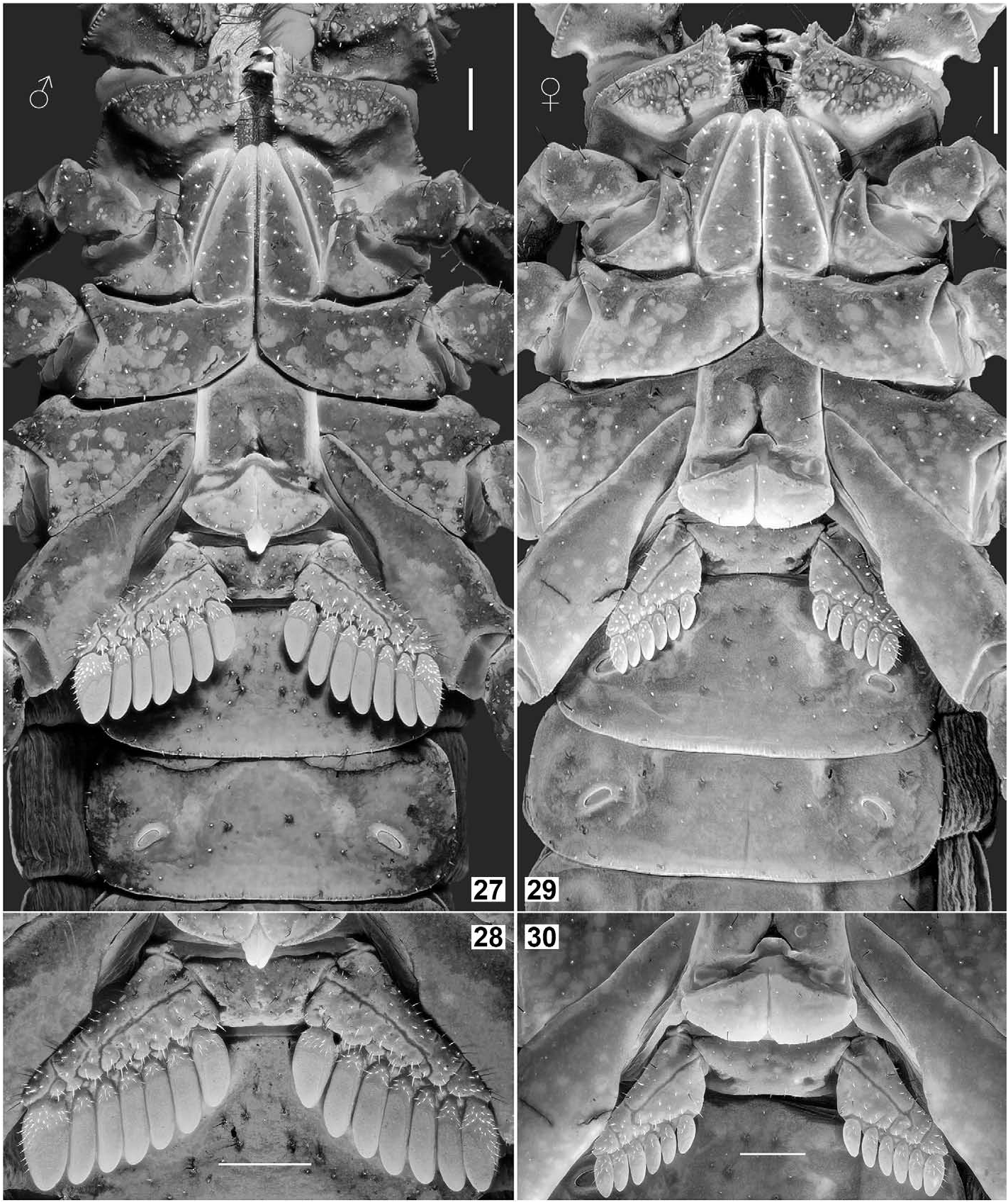
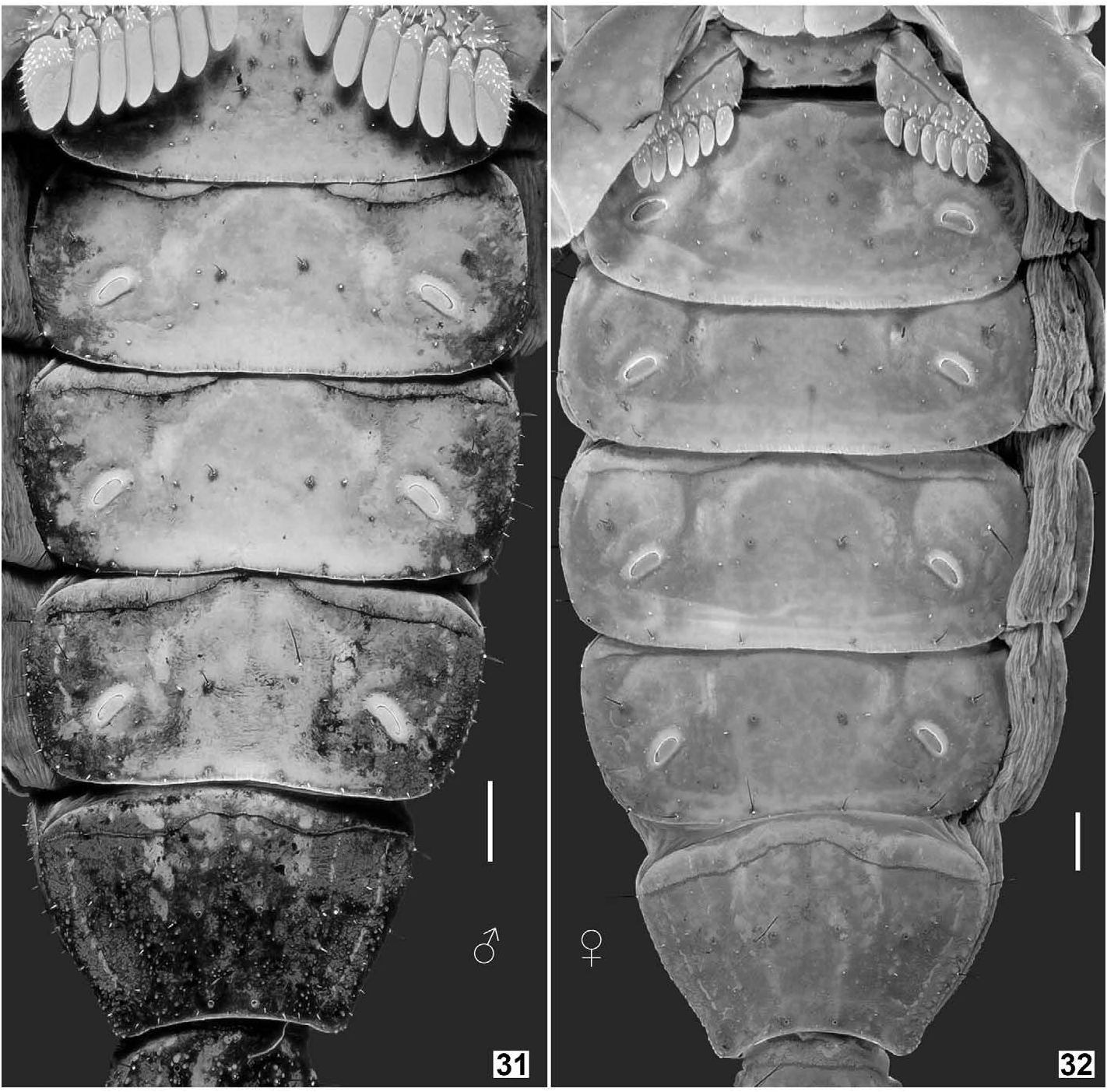
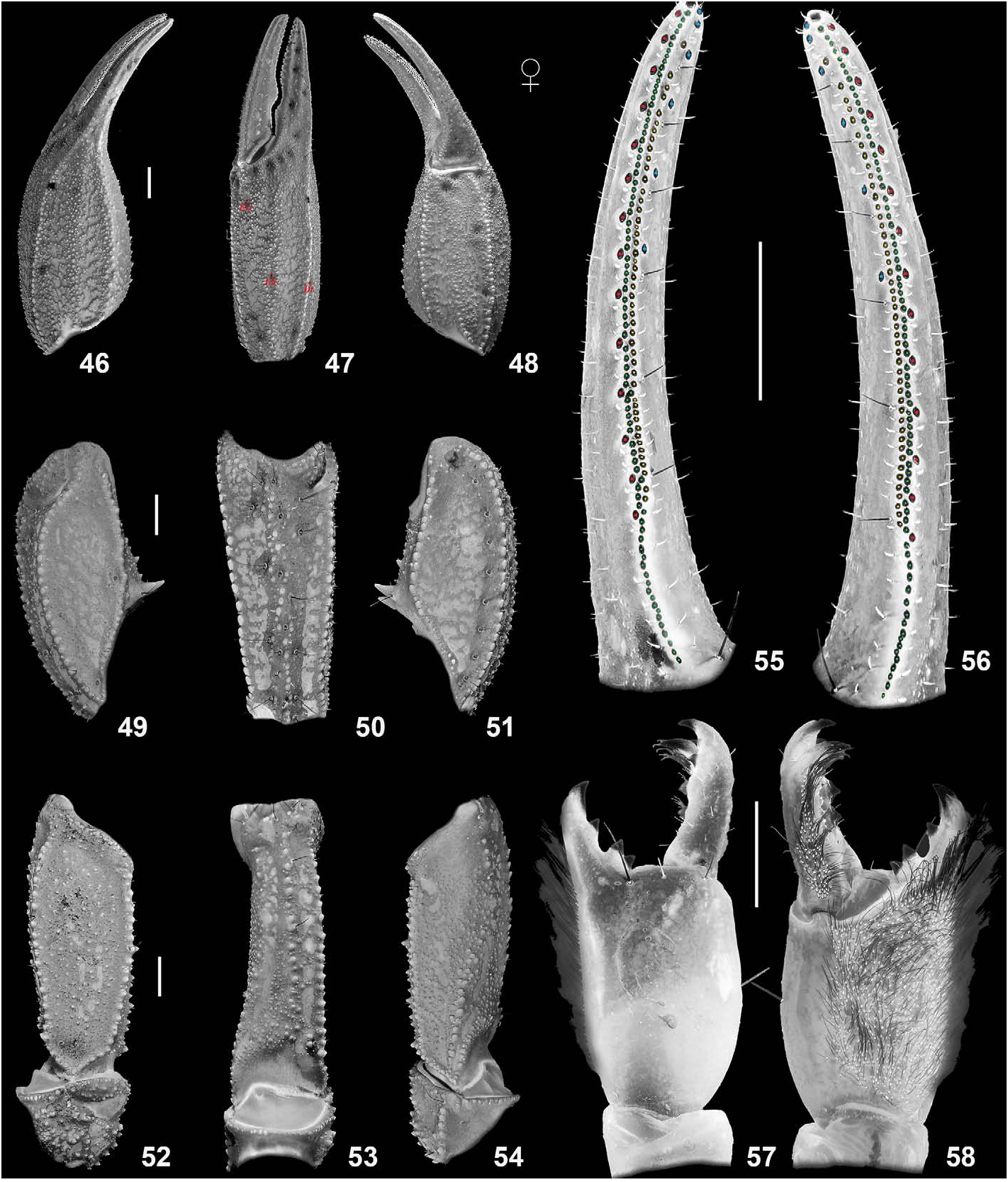
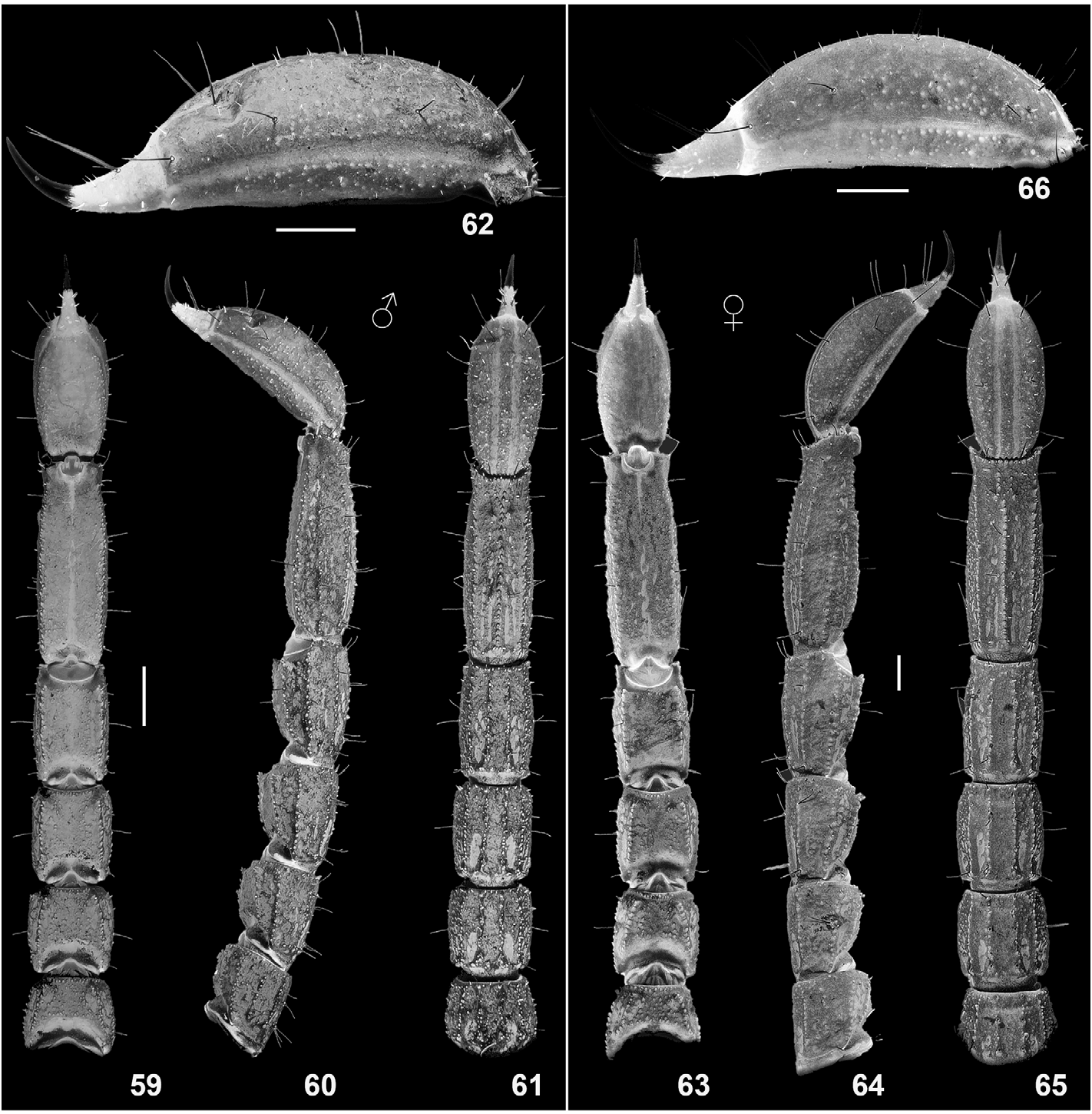
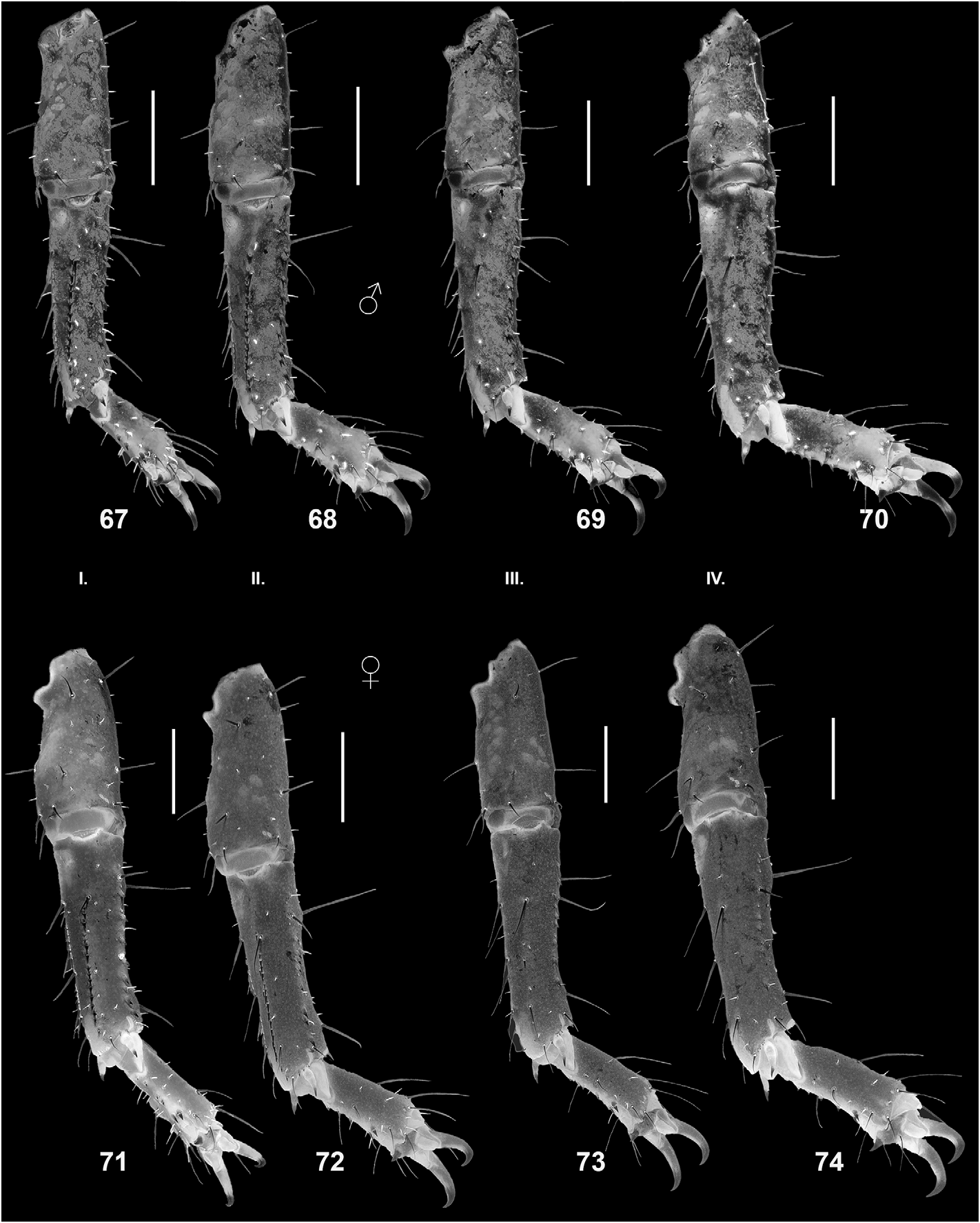
DESCRIPTION (based on male holotype and female allotype). Photos of the specimens in Figs. 19–22 View Figures 19–22 were taken under white light (specimen in vivo habitus is shown in Fig. 75–76 View Figures 75–76 ). Photos of the specimens in Figs. 23–74 View Figures 23–24 View Figures 25–26 View Figures 27–30 View Figures 31–32 View Figures 33–45 View Figures 46–58 View Figures 59–66 View Figures 67–74 were taken under UV light to depict cuticular morphosculpture (granulation and carinae). Morphometric data are given in Tables 1–3.
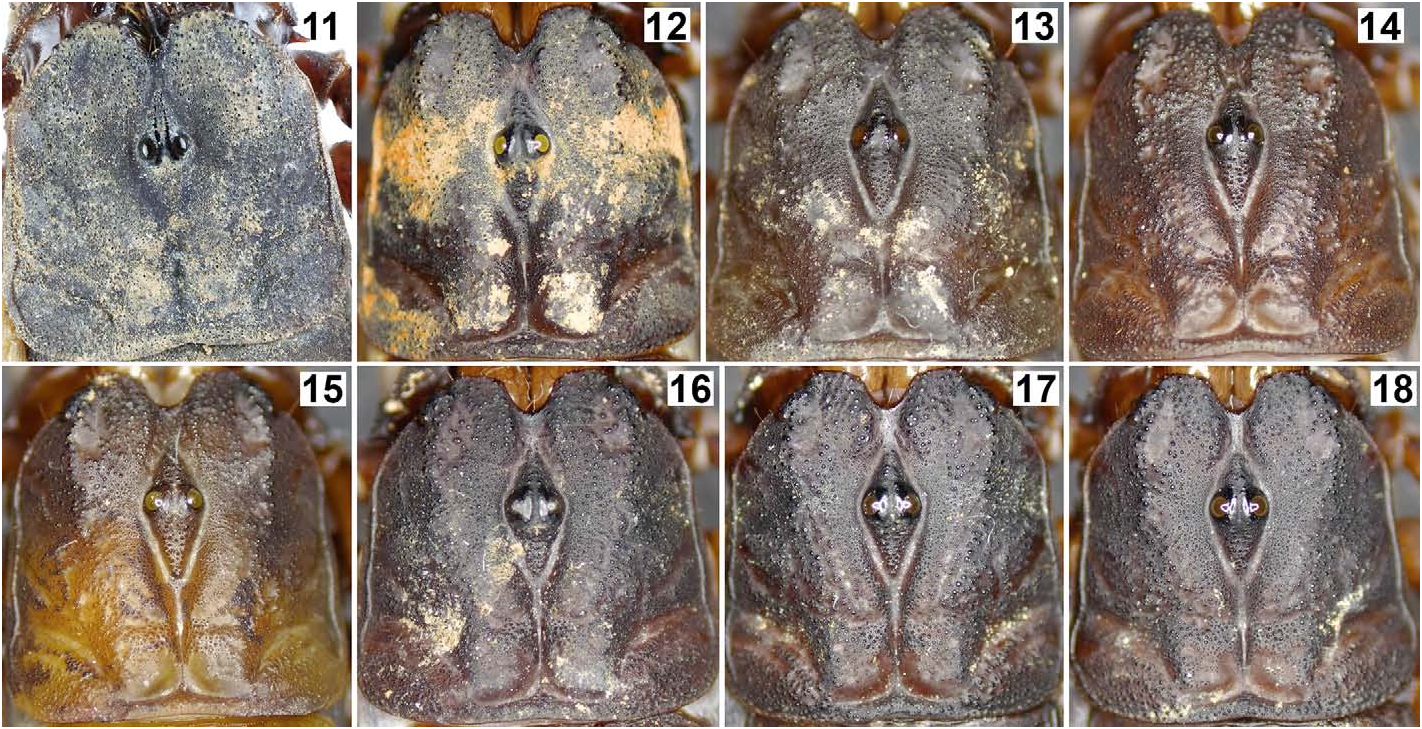
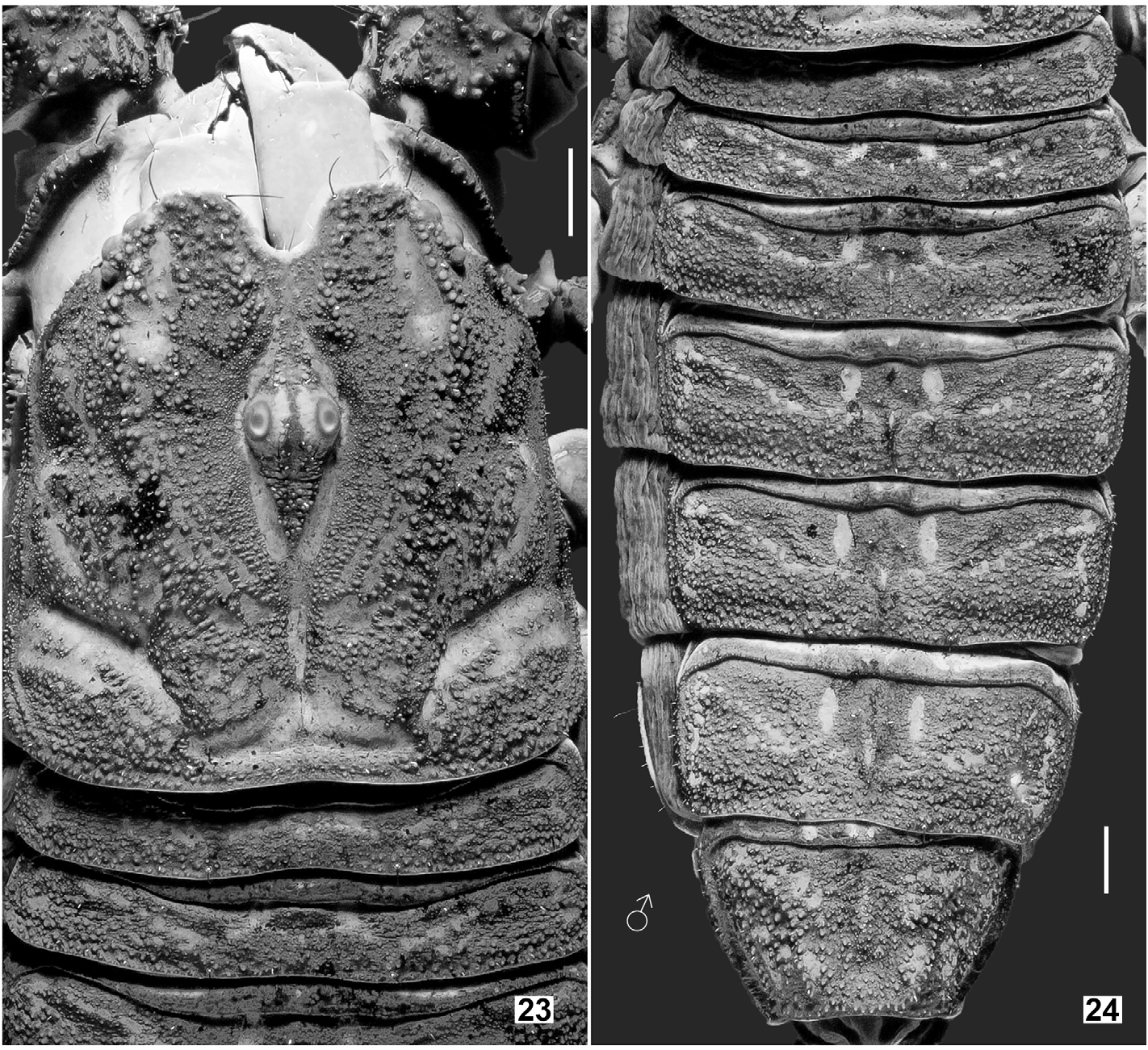
Coloration (in vivo) ( Fig. 75–76 View Figures 75–76 ). Base color uniformly reddish brown to brownish black (female paratype probably just finished the final ecdysis and exhibits lighter coloration). Color darker on strongest carinae and pedipalp (except for the tip which is reddish), with intercarinal surfaces slightly reddish. Mesosoma essentially brownish black, with posterior margin of each tergite reddish brown (except for VII); granules slightly darker. Metasoma the same as mesosoma, but with several carinal granules conspicuously darker. Telson tawny, becoming lighter towards the vesicle/aculeus juncture and abruptly reddened at the aculeus, eventually turning black. Legs vaguely variegated with different degrees of brown, becoming reddish brown distally towards telotarsi. Chelicerae base color yellow with brown reticulation on dorsal surface, becoming reddish brown at anterior margin of manus and on both fingers. Ventral coloration of prosoma light brown; anterior area of coxapophyses brownish yellow. Genital operculum and pectines brownish yellow to whitish yellow; sternites III–VI semi-translucent with posterior area whitish yellow; sternite VII dark brown; areas of book lungs anterior to each respiratory spiracle brownish yellow to whitish yellow. Prosoma and mesosoma ( Figs. 23–32 View Figures 23–24 View Figures 25–26 View Figures 27–30 View Figures 31–32 ). Prosoma: Carapace with 3 pairs of lateral eyes of which two are larger and one is smaller. Median ocular tubercle raised. Superciliary carinae of the median ocelli granulated anteriorly and posteriorly, long and separated by a smooth, conspicuous sulci. Median dorsal part the carapace (ca. half of total area) unevenly planar, from anterior to posterior margins; lateral surfaces slanting downwards. Lateral surfaces with a pair of shallow central lateral sulci and a pair of deep posterior lateral sulci, both are smooth. Entire carapace densely covered with small to large size granules; carinae absent. Anterior margin of carapace with a prominent median notch (emargination) leading to a shallow, wide, smooth anteromedian sulcus. Circumocular sulcus is connected with anteromedian sulcus anteriorly, and with posteromedian sulcus posteriorly, which successively leads to a deep posterior marginal sulcus; all sulci smooth. Larger granules concentrated at edges flanking anteromedian sulcus, above and posterior to lateral ocelli, lateral surfaces and along the posterior margin. Chelicerae with dorsal surface smooth and ventral surface setose; macrosetae localized on fixed finger. Dorsal distal, ventral distal and distal denticles of cheliceral fingers very long. Mesosoma : tergites densely and evenly covered with small to moderate granules, with one median carina indicated, flanked with a pair of depressions (conspicuous on tergites IV, V and VI). Tergite VII is pentacarinate (carinae are only indicated). Sternites smooth with sparse macrosetae and fluorescent microsetae (except sternite VII which has four granulate carinae but not clearly defined); respiratory spiracles suboval. Pectines with marginal lamellae I and III delimited, sulcus that usually separates the marginal lamella II from the middle lamella often incomplete; pectine teeth number 7– 7 in holotype male and 6– 6 in allotype female; sensory area covers the majority of each pectinal tooth in males but only about a half in females; fulcra present; fluorescent microsetae stout.
Metasoma and telson ( Figs. 59–66 View Figures 59–66 ). Metasoma sparsely hirsute and granulated; all carinae evenly with moderatesized granules. Metasomal segment I with 10 carinae, II–IV with 8 carinae, and V with 7 carinae. Median lateral carina of metasoma II–IV obsolete, presented by several granules. Granules relatively larger and rounded on the median lateral carinae of segment I and dorsolateral carinae of segments II–IV, but smaller and thinner on the remaining carinae. Anal arch armed with multiple small, blunt granules (lobes). Telson relatively elongate for the genus, surface essentially smooth with fine granules on the lateral surfaces which become larger when approaching the dorsal surface; annular ring developed. Vesicle sparsely covered with fluorescent microsetae and few macrosetae, with a pair of smooth, shallow sulci on lateral surfaces.
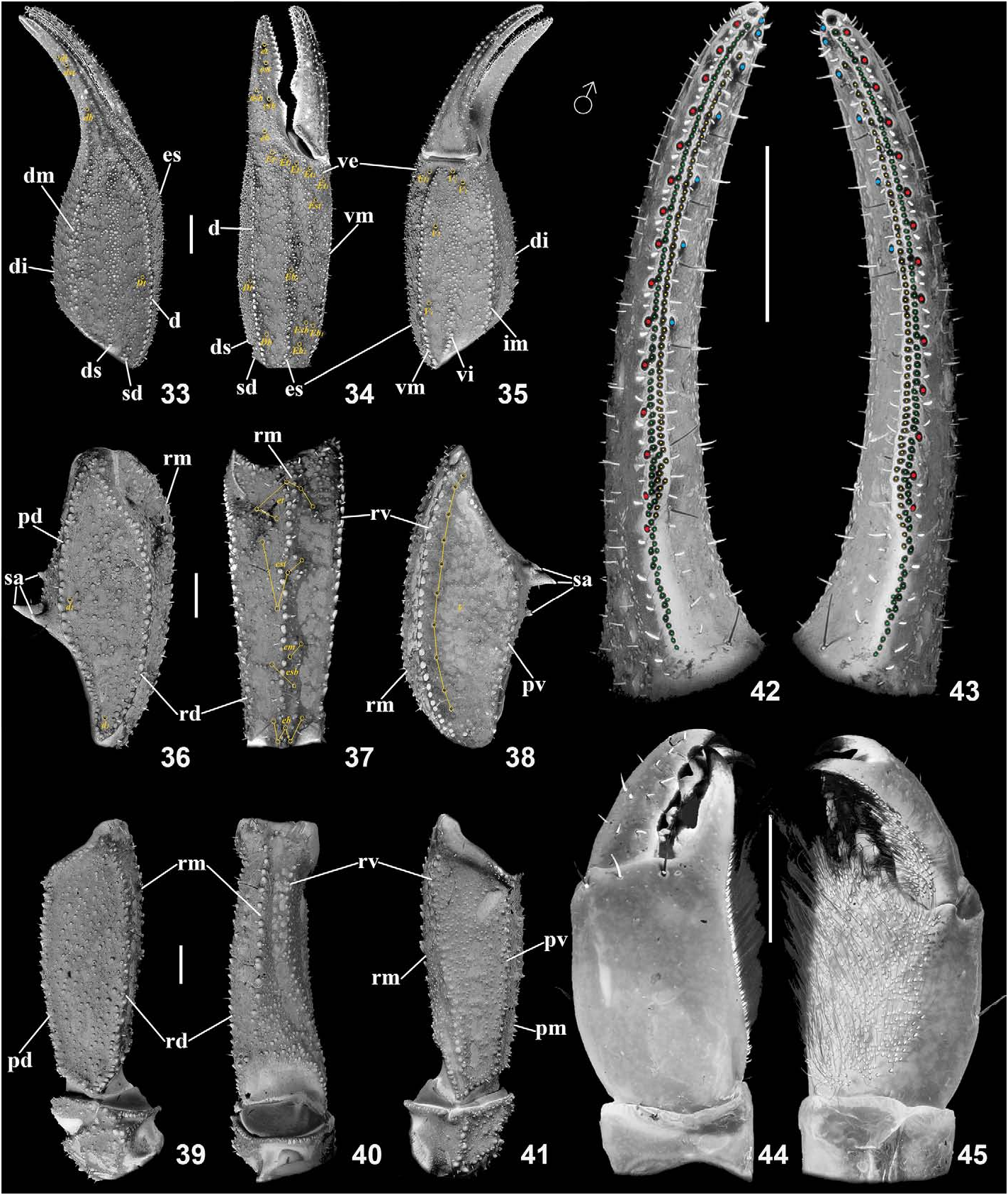
Pedipalps ( Figs. 33–58 View Figures 33–45 View Figures 46–58 ). Pedipalps very sparsely setose. Patella with 18 external and 9–10 ventral trichobothria. Chela with 4 V series trichobothria located on ventral surface. Chelal trichobothrium Eb 3 located in middle of manus between trichobothria Dt and Est. Femur and patella granulated. Intercarinal surfaces densely scattered with small to moderate granules. Femur with 6 granulose carinae; promedian and retromedian carinae almost complete, composed of large granules; retroventral carina only indicated, reduced to small to moderate granules, similar to those between carinae; granules moderate on prodorsal, retrodorsal and proventral carinae. Patella with 5 granulose carinae, composed of moderate to large granules with evident spacing (not costate); prolateral surface with two large and one small spiniform apophyses, the small one on the same level with the lower large one. Manus dorsally with rounded, small to moderate granules, which form a reticulate pattern; dorsal internal carinae formed by spiniform granules; largest granules constitute dorsal marginal, interomedian and ventrointernal (only the proximal part) carinae; moderate size granules present on digital and interomedian carinae; dorsal secondary, ventrointernal and external secondary gradually weakening to dispersed granules; subdigital and ventroexternal carinae obsolete. Movable fingers with 56–59 IAD in holotype male and 47 in allotype female, which have the same size as MD (87 in holotype male and 76–82 in allotype female) and form a second row; 6–7 (in holotype male) or 6 (in allotype female) ID and 14–15 (in holotype male) or 14 (in allotype female) OD also present.
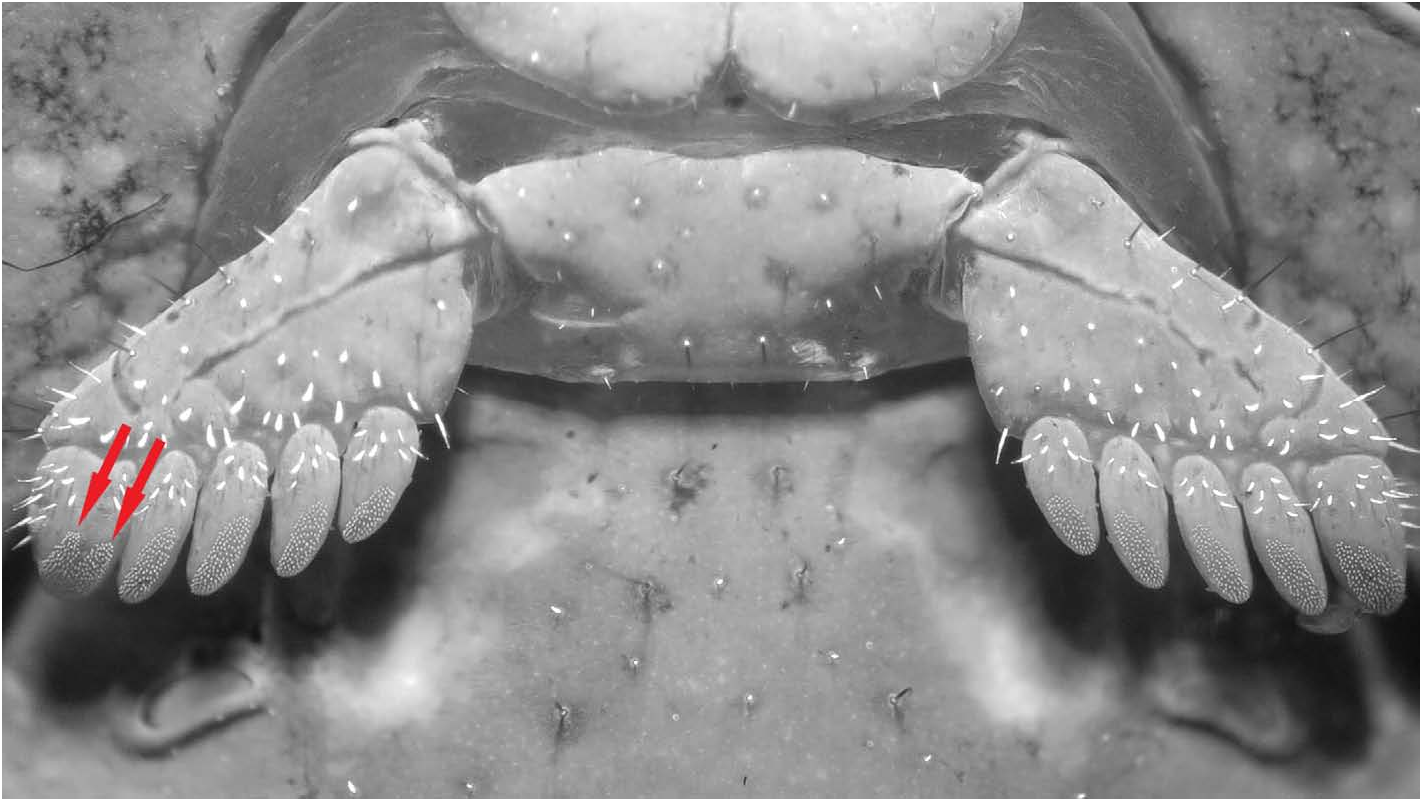
Legs ( Figs. 67–64 View Figures 67–74 ). Tibia and tarsomeres of legs with several macrosetae and fluorescent microsetae not arranged into bristle combsondorsalsurfaces, butwithrowsofspinulesondorsolateral surfaces and on legs I–II also on ventrolateral surfaces (spinules on ventrolateral surfaces of legs I–II denser and longer, forming conspicuous rows). Tarsomere II of legs I–IV with row of 5–8 short, stout median ventral spinules, and two pairs of flanking setae. Femur with 3–4 and patella 4–5 carinae; both femur and patella finely granulated, with sparse setae.
Measurements. See Tables 1–3.
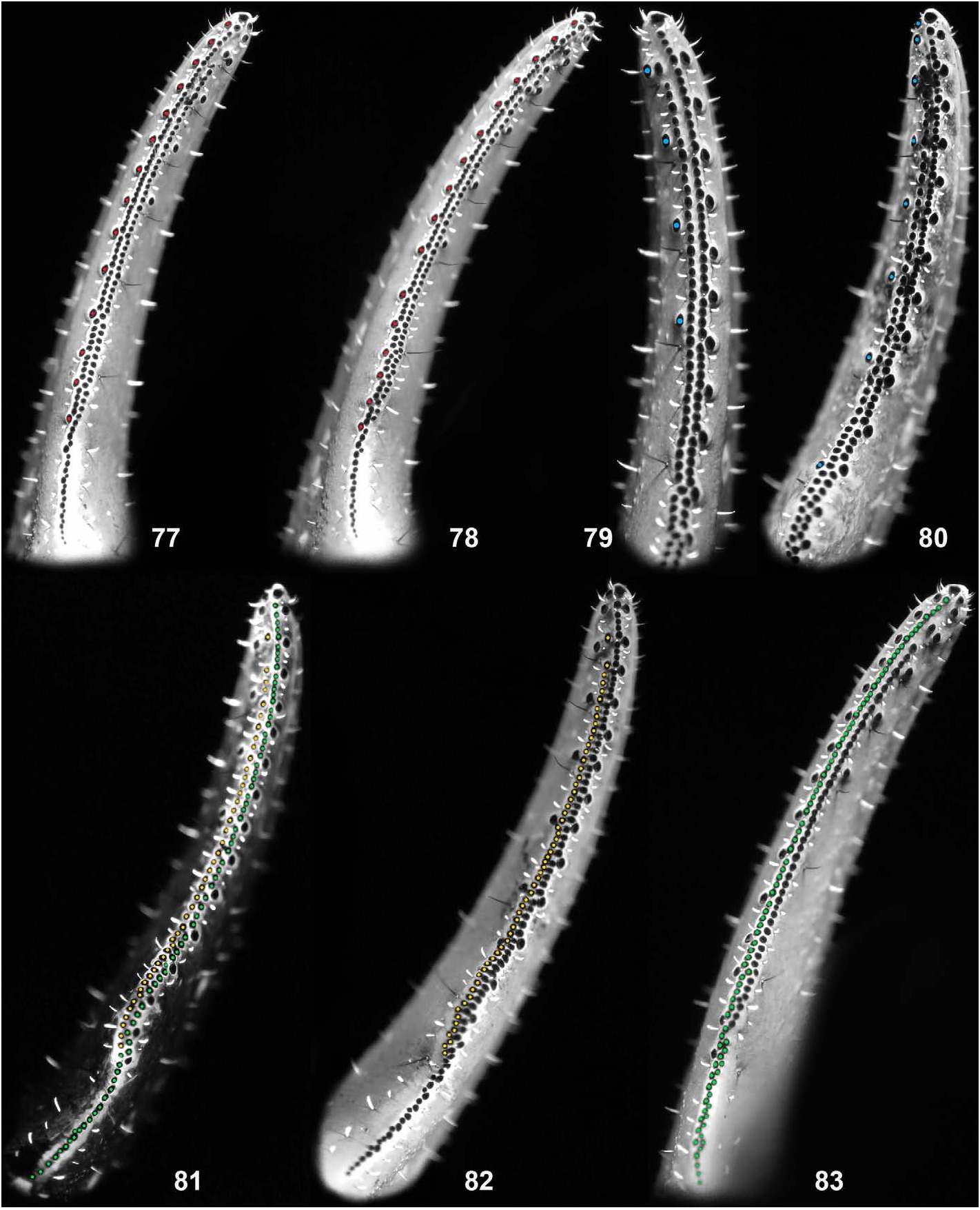
Variation. As reported in Tang (2022c), based on 15 specimens collected in July 2022 and designated here as paratypes, there are two size classes of adult males and females, supported by the presence of hemispermatophores and embryos, respectively. The normal numerical variation range of the external trichobothria on the pedipalp patella is 18–19, but the lowest (17) and highest (20) values were both recorded only once. Higher numerical variation is presented in the IAD and MD of the pedipalp movable finger (Tang, 2022c: table 3). As shown in Tang (2022c: 15, fig. 85), an abnormal dentition arrangement was exhibited on the right pedipalp movable finger of a male. This anomaly has only been observed once (1/19 individuals). The maximum/ minimum values of the four types of denticle recorded in the paratypes reported in Tang (2022c) are shown in Fig. 77–83 View Figure 77–83 ; the four new specimens, collected in August 2022, generally fall into that range. In terms of the morphosculpture, there are variations in the development degree (size) of pectinal fulcra and the division of pectinal lamellae within the same individual. In most cases, marginal lamellae I (basal) and III are delimited, while the sulcus that usually separates marginal lamella II from the middle lamella is often incomplete. The middle lamella may form a single compact unit but may also break into small plates next to where the lamella II is usually delimited. For more evident examples, see Tang (2022c: 13–14, figs. 54–60, 77–84). Other variations including L/W ratio of pedipalp chela, pedipalp finger lobe development, ventral trichobothria on pedipalp patella and PTC, have all been given in Tang (2022c: fig. 32, table 3). In this study, the female paratype was found to have only 5 pectinal teeth on both sides, although the most distal tooth was conspicuously larger and that on the left side appeared to be an incompletely separated tooth ( Fig. 84 View Figure 84 ). Both new paratypes were smaller than the specimens collected previously, but their maturity was determined by the development degree of the proximal lobe on the pedipalp movable finger (i.e., not smaller than those of the previous paratypes in respective sexes). The male was further confirmed mature by the presence of hemispermatophores but neither eggs nor embryos were retrieved from the female. However, according to the collector, the paratype female was carrying offspring when it was collected.
AffINITIES. The new species has long been misidentified as Scorpiops kubani ( Kovařík, 2004) , however, it can be differentiated from that species as follows: (1) carapace with much denser and coarser granules in S. lowei sp. n.; (2) a consistently wider median notch on the anterior margin of carapace in S. lowei sp. n.; (3) higher numbers of OD on the pedipalp movable finger in S. lowei sp. n. (14–16 vs. 11–13); (4) pectines with more developed fulcra in S. lowei sp. n.. It is also important to note that, Di et al. (2011) described the pectinal fulcra of their materials (based on specimens: Ar.- MWHU-YNMH1001–02) as “... small, reduced or absent in E. kubani , but well developed in E. shidian ...”. However, based on the specimens studied by Tang (2022c: figs. 32, 34), the two species possess fulcra of similar development degree, and small (“reduced”) and large (“well developed”) fulcra can both be found in the same individual of the two species. Therefore, the authors may have interpreted their observations to comply with the original diagnosis of S. kubani . For the comparisons with its congeners in Yunnan, see Tang (2022c).
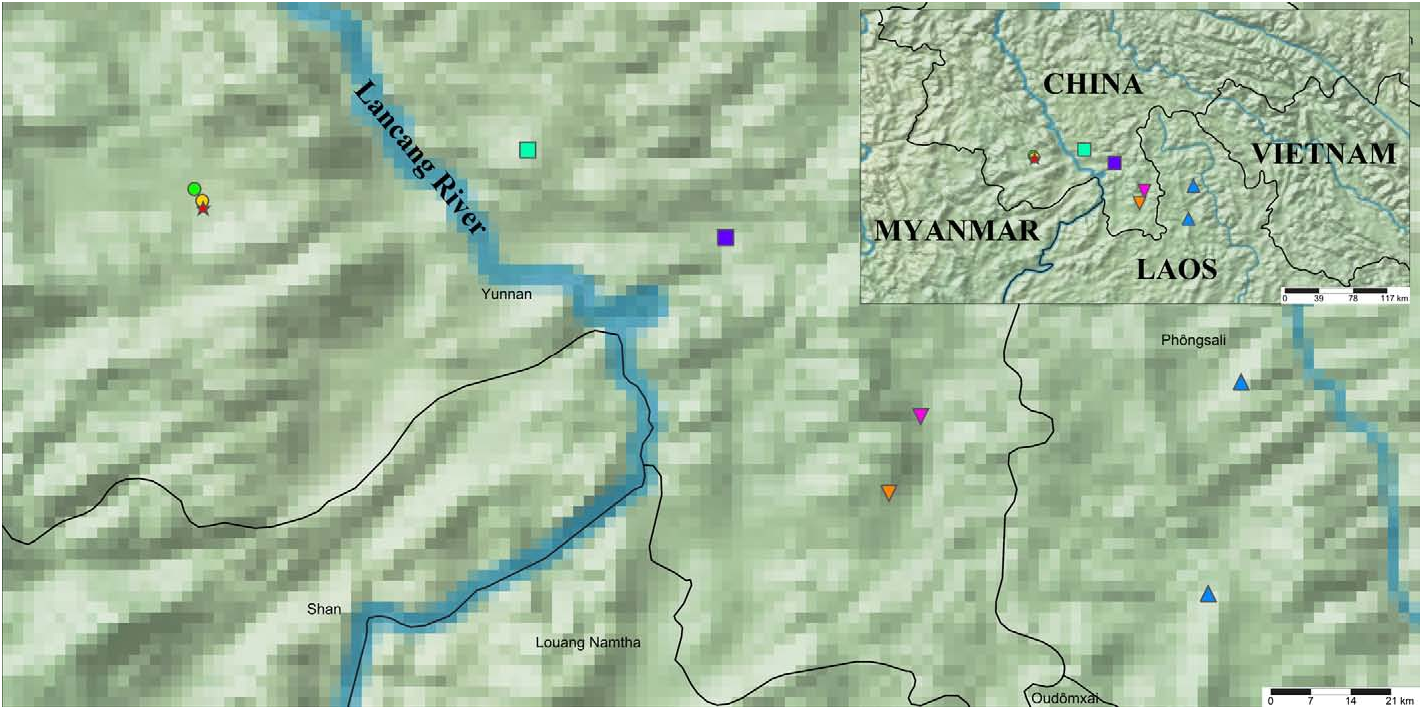
DISTRIBUTION. The new species is known only from the Yunnan Province of China, and so far is confirmed only from the Menghai County. Anecdotal observations of unidentified individuals were recorded from Jinghong City (Tang, 2022c: 29, figs. 226–229; iNaturalist, obs. ID 131523930) and Menglun Town of Mengla County (iNaturalist, obs. ID 111002550, 128697510, 128751713, 8951809 and 132377728) and these populations are separated from the new species by the Lancang River (= Mekong River outside the borders), a river that has been estimated to established in the middle Miocene ( Nie et al., 2018). In order to test whether this river acts as a geographical barrier, in this study, we obtained several individuals from Jinghong City and Menglun Town. However, based on the external morphology alone, a confident conclusion of which species those population represent cannot be reached as there is the possibility of character gradients (see below).
Comparison of the new species with specimens from nearby regions
In terms of the Jinghong population, the adult males appear to have a more pronounced proximal lobe on the pedipalp movable finger, similar to that of S. puerensis , as well as a more robust pedipalp chela ( Table 3). Furthermore, a higher pectinal tooth count is observed in this population. Among the pectines of all males studied (12 pectines), 6 pectines were found to have 8 pectinal teeth, 3 to have 9, 2 to have 7 and 1 to have 6. However, the single 6 pectinal teeth was only found in the juvenile male which appeared to have incompletely separated distal teeth on both sides (dilated significantly). In contrast, among the pectines of all male S. lowei sp. n. studied (28 pectines, including the materials studied by Di et al. (2011)), 15 pectines were found to have 8 pectinal teeth, 12 to have 7 and 1 to have 6. No specimens were found to have 9 pectinal teeth. The average number of ventral trichobothria on the pedipalp patella is also higher than that of S. lowei sp. n. in males. Among the patellae of all males studied (12 patellae), 9 patellae were found to have 11 ventral trichobothria and 3 to have 10. On the contrary, 10 ventral trichobothria is a common state in S. lowei sp. n. (22/28 patellae; the rest of the patellae have 9 ventral trichobothria). Females of this population are very similar to S. lowei sp. n., but the average number of the ventral trichobothria on the pedipalp patella is still 11 and one female was recorded to have 12 trichobothria on its left pedipalp. Although Di et al. (2011: table 2) recorded an immature female to have 11 trichobothria on both of its patellae, it was an isolated case. The pectinal tooth count also goes up to 8 at maximum (though most are 7), which is unprecedented in all the Menghai specimens (as S. kubani ) studied by researchers (18 specimens in total; Di et al. (2011) and this study). Additionally, the pectinal fulcra are more developed in the Jinghong population. No obvious difference was confirmed for the number of denticles on the pedipalp movable finger, but it tends to be slightly higher in the Jinghong population. In all the fingers examined (23; one finger was abnormal, thus not considered), only 2/23 were found to have 13 OD, while 7/23 have 16 and 11/23 have 15. In terms of ID, 3/24 fingers have 6, 15/24 have 7 and 6/24 have 8. The number of IAD and MD are higher, despite the smaller sample size (12 vs. 19). The single adult male examined in Tang (2022c) from Mt. Jinuo (Jinghong City) falls into the variation range recorded in this study.
The data recorded for the Menglun population did not differ significantly from those recorded for the Jinghong population, and it was hence assumed to be a conspecific population of the latter ( Table 3). One male was found to have only 7 ventral trichobothria on its right pedipalp patella and 15 external trichobothria on the left; both of these counts were considered abnormal. An additional exception was the discovery of 19 external trichobothria on the left pedipalp patella in one male (none of the Jinghong specimens had been recorded with this value). To sum up, in the studied males of the Menglun population, 5/8 examined pedipalp patellae had 11 ventral trichobothria and 2/8 had 10; 6/8 examined pedipalp patellae had 18 external trichobothria; 3/8 examined pectines had 7 teeth, 4/8 had 8 and 1/8 had 9. In the females of the Menglun population, two anomalies were confirmed in a juvenile on its right pedipalp: it had an extra ventral trichobothrium (rendered 12 in total) on the patella, and a proliferation of denticles again appeared, on the tip of its right movable finger. Similarly, one adult female was observed to have only 9 ventral trichobothria on its right pedipalp but considering the normal count (9/16 of the patellae of the females had 11 and 6/16 had 10; the case of 12 trichobothria mentioned above is here included under the count of 11 trichobothria) and the spacing between adjacent trichobothria, a trichobothrium was assumed to be missing. A weak proliferation of finger denticles was observed on both fingers in an adult female: it had extra denticles next to the two ID and one OD on the left pedipalp and a proliferation at the proximal part of the right pedipalp. On the contrary, one adult female was presumed to be lack of an OD on its left pedipalp due to the unusual spacing between the two adjacent OD.
The single female collected from Wangtianshu (approximately 17.32 km from the type locality of S. vachoni ) showed a range of morphometrics closer to S. vachoni than to either the Jinghong population or the Mengla population in terms of the finger lobe L/D ratio and counts of MD. However, as asserted in Tang (2022c), a credible identification of Scorpiops species from Yunnan may depend on a sufficient sample size from the targeted population. Therefore, this specimen is not explicitly assigned to any of the species/ populations studied, pending a DNA analysis to reveal its identity.
Both the Jinghong and the Menglun populations resembled S. vachoni in terms of the pedipalp patellar trichobothrial number and PTC ( Table 3). Their total length is slightly smaller than that of S. vachoni and their L/D ratio of the finger lobe is slightly greater (i.e., lobe less pronounced) than that of S. vachoni . On the other hand, their pedipalp chelae are slightly more robust than those of S. vachoni . However, as asserted in Tang (2022c), all these linear morphometrics measured manually are not (and may never be) accurate but instead referential, and large quantities of specimens are required to reduce the variance arising from slight errors. The sample size of each population studied here was not statistically sufficient (less than 30 specimens). Additionally, available adult specimens of each sex were far fewer than what was required for statistical analysis (ratiometrics regarding the chela ratio and lobe ratio are maturity-dependent). Therefore, more explicit and ontogenetically invariant characters were chosen as parameters to visualize the relationship between these populations ( S. lowei sp. n., Jinghong, Menglun and S. vachoni ), i.e., PTC, and the counts for IAD and MD. To avoid ambiguity, it is clarified hereby that all fingers (except for the juveniles of S. vachoni ; see Tang (2022c, “Methods”)) were analyzed, except for the fingers with extremely abnormal dentition. If one pectinal tooth was obviously dilated, it was enumerated twice.
The females of S. lowei sp. n. exhibited a clear trend of possessing fewer pectinal teeth than the females of other populations (Fig. 85). The PTC for Jinghong and Menglun populations was very similar, while females of S. vachoni revealed a higher possibility of showing 8 pectinal teeth despite their smaller sample size (10 pectines vs. 12 (Jinghong) and 16 (Menglun) pectines). The PTC for the males of these four populations mostly covered the range of 7–8; the sole exception was a pectine observed with 6 teeth in a male S. lowei sp. n. (Fig. 86). Most of the males of Jinghong population, Menglun population and S.vachoni possess 8 teeth, few possess 9, and in S. vachoni , only one male was observed to possess 7 teeth, which was unusual considering the sample size (32 pectines). The trend for the IAD and MD count across the four populations manifested an interesting correlation with the distributional pattern (Figs. 87–88, cf. Fig. 89 View Figure 89 ). Among the three populations east to the Lancang River, S. vachoni , which is distributed in the southernmost region showed the highest count for both denticles; on the other hand, S. lowei sp. n. which occurs on the west side of the river, had the lowest count. The Jinghong population is located north of the Menglun population, and the counts increased when moving southward. The counts for the Jinghong population were closer to those for S. lowei sp. n., and the counts for the Menglun population were closer to those for S. vachoni . The observed trend in the external morphology may potentially be associated with intrinsic (e.g., genetic) factors, and may also indicate a dispersal or evolutionary process.
The discovered differences of the Jinghong population (and Menglun population, which is assumed to be conspecific) from S. lowei sp. n. could merit its own species. However, their overall resemblance in some taxonomically informative characters (e.g., carapace) to S. vachoni prevents a premature description of the former, and it is currently considered cryptic until DNA analysis (our work in progress) is accomplished to clarify its relationship with S. lowei sp. n., as well as that with S. vachoni and S. kubani . Nevertheless, the differences found demonstrated that S. lowei sp. n. is not found further east, and the populations from Jinghong and Menglun do not belong to this species.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |