Stygomomonia manchurica Semenchenko
|
publication ID |
https://doi.org/ 10.5281/zenodo.180506 |
|
DOI |
https://doi.org/10.5281/zenodo.6228254 |
|
persistent identifier |
https://treatment.plazi.org/id/03E587AE-6E0C-FFE4-FF59-FE619EFDFF37 |
|
treatment provided by |
Plazi |
|
scientific name |
Stygomomonia manchurica Semenchenko |
| status |
sp. nov. |
Stygomomonia manchurica Semenchenko sp. n.
( Figs. 1–7 View FIGURES 1 – 5 View FIGURES 6 – 7 )
Material examined. Holotype: male (01-kas IBSS), Russia, Primory Territory, Khasansky District, “Kedrovaya Pad National Nature Biosphere Reserve”, the Kedrovaya River, N 43°06’ 056”, E 131°33’ 310”, 18.VII.1993, coll. T.M. Tiunova. Paratypes: 1 male (Tdh-1 ZMMU), 27.VI.1993, 3 females (Tdh-2-4 ZMMU), 18.VII.1993, 9.IX.1993, 9.X.1993 and 1 deutonymph (Tdh-5 ZMMU), 9.X.1993, coll. T.M. Tiunova. Paratypes with same locality as holotype.
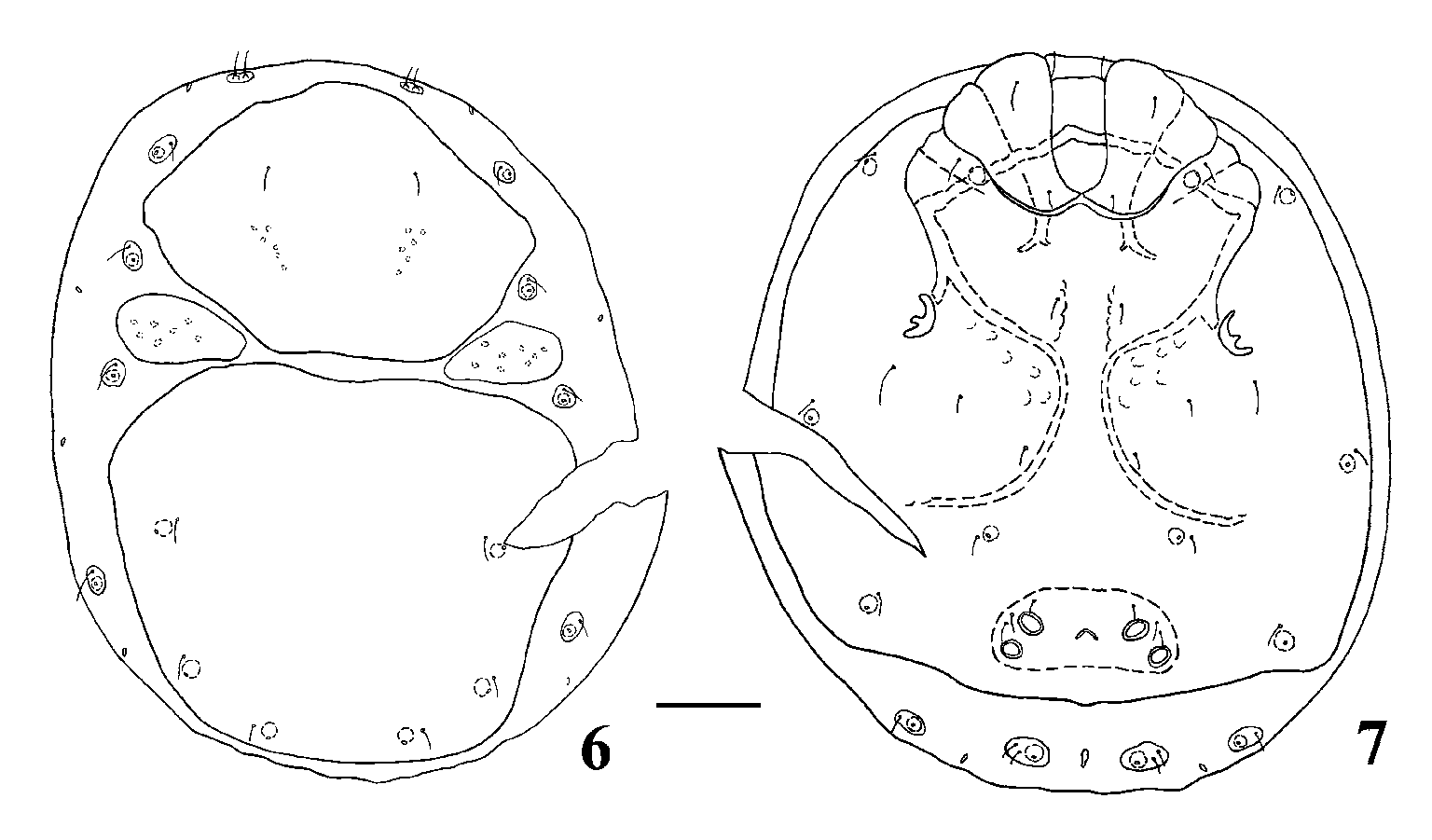
Male ( Figs. 1–2, 5 View FIGURES 1 – 5 ). Dorsal shield elongated, tapered near posterior end; peripheral thickening of shield not extending medially toward the posterior pair of dorsoglandularia ( Fig. 1 View FIGURES 1 – 5 ). The anterior apex of coxal plates I wide and rounded ( Fig. 2 View FIGURES 1 – 5 ). Coxal plates IV bear 5–6 long setae below insertions of legs IV. Width of genital field about twice its length, with 3 pairs of acetabula and 5 pairs of setae, posterior pairs of acetabula widely separated from centre of genital opening. Single pair of platelets with glandularia on each side of excretory pore, posterior pair of slit organs below platelets, platelets slightly constricted at level of slit organs. Pedipalps chaetotaxy similar to that of female ( Fig. 4 View FIGURES 1 – 5 ). I–L–6 of first pair of legs less than half as long as I–L– 5, a pair of proximoventral setae on posterolateral surface of I–L–6 inserted near midlength of segment ( Fig. 5 View FIGURES 1 – 5 ).
Measurements (proportion of paratype indicated in parentheses): Length dorsal shield 490 (530), width dorsal shield 370 (400), length ventral shield 470 (500), length coxal plates 310 (320), width coxal plates (one side, midline to tip of condyle at insertion of leg IV) 84 (94), length genital field 68 (75), width genital field 144 (145). Lengths pedipalp segments: 20 (21), 53 (54), 32 (32), 46 (48), 28 (29) (P 1–5). Lengths leg segments: I – 36 (34), 58 (56), 82 (80), 84 (83), 184 (196), 86 (92); II – 36 (40), 46 (48), 52 (52), 66 (68), 86 (93), 92 (94); III – 44 (48), 48 (50), 49 (52), 72 (76), 104 (108), 92 (94); IV – 60 (61), 62 (68), 68 (72), 96 (104), 128 (130), 104 (114).
Female ( Fig. 3–4 View FIGURES 1 – 5 ). Dorsal shield similar to that of male. Ventral shield with chaetotaxy of coxal plates IV and shape of coxal plates I similar to that of male ( Fig. 3 View FIGURES 1 – 5 ). Genital field bearing 3 pairs of small acetabula and 5 pairs of setae; region posterior to genital field similar to that of male but with large postgenital sclerite. P–5 of pedipalp with 4 thin setae and 3 stout spines – 1 short and 2 long ( Fig. 4 View FIGURES 1 – 5 ). Distal segments of first pair of legs similar to that of male.
Measurements (of 3 paratypes): Length dorsal shield 500–510, width dorsal shield 375–400, length ventral shield 420–445, length coxal plates 308–315, width coxal plates (one side, midline to tip of condyle at insertion of leg IV) 88–110, length genital field 90–104, width genital field 151–152. Lengths pedipalp segments: 15–18, 54–56, 28–30, 47–48, 27–28 (P 1–5). Lengths leg segments: I – 38 –40, 52–60, 81–88, 88–90, 182–206, 92–96; II – 34 –40, 46–52, 51–52, 68–72, 88–92, 95–100; III – 44 –52, 48–53, 48–50, 72–76, 104– 108, 92–100; IV – 56 –60, 68–72, 69–73, 101–104, 132–136, 106–114.
Deutonymph ( Figs. 6–7 View FIGURES 6 – 7 ). Dorsal shield divided into anterior and posterior plates; anterior dorsal plate subdivided into large anterior plate and pair of small posterolateral plates ( Fig. 6 View FIGURES 6 – 7 ). The anterior apex of coxal plates I wide and rounded ( Fig. 7 View FIGURES 6 – 7 ). I–L–6 less than half as long as I–L–5, a pair of proximoventral setae on posterolateral surface of I–L–6 inserted near midlength of segment.
Measurements: Length anterior dorsal plate 138, width anterior dorsal plate 184, length posterior dorsal plate, length ventral shield 328, length coxal plates 224, width coxal plates (one side, midline to tip of condyle at insertion of leg IV) 76, length genital field 36, width genital field 92. Lengths pedipalp segments: 16, 40, 24, 39, 18 (P 1–5). Lengths leg segments: I – 32, 44, 56, 68, 144, 69; II – 30, 44, 36, 50, 66, 78; III – 30, 36, 32, 54, 75, 72; IV – 52, 46, 48, 70, 96, 99.
Etymology. The species epithet manchurica is derived from the name of the East-Manchurian Mountains, the type locality of the new species.
Habitat. Stygomomonia manchurica sp. n. was collected from the interstitial habitat in the Kedrovaya River. The substratum of the river is composed of pebbles, gravel and coarse-grained sand sediments; water temp. 13–14 °C, water depth 22–55 cm and the speed of water flow 0.2–1.0 m/sec.
Differential diagnosis. Adults of S. manchurica sp. n. have the following features in common with S. neomexicana Cook , known from North America: dorsal shield elongated, tapered near posterior end; peripheral thickening of shield not extending medially toward the posterior pair of dorsoglandularia; P–5 of pedipalp with 4 thin setae and 3 stout spines (1 short and 2 long); I–L–6 of first pair of legs less than half as long as I– L–5, pair of proximoventral setae on posterolateral surface of I–L–6 inserted near midlength of segment. Adults of S. manchurica sp. n. can be distinguished from S. neomexicana by the shape of the platelets that are just slightly constricted at level of slit organs, whereas S. neomexicana has strongly constricted platelets at level of slit organs. By the shape of platelets S. manchurica sp. n. is more similar to the North American species S. occidentalis Cook. However , S. manchurica sp. n. differs from S. occidentalis and S. neomexicana in that the anterior apex of coxal plates I is wide and rounded, the genital field in male is broad, its width about twice its length and the posterior pairs of acetabula greatly distanced from the centre of genital opening. The anterior apexes of coxal plates I in S. occidentalis and S. neomexicana are more pointed, the width of genital field of males is less than half as long as its length and the posterior pairs of acetabula are close to the centre of the genital opening.
The deutonymph of S. manchurica sp. n. is very similar to that of S. neomexicana in the character of the division of the dorsal shield into three anterior plates (one large and two small) and one posterior plate. This type of division of the dorsal shield distinguishes these two species from all other known species of the subgenus Stygomomonia . The deutonymph of the new species can be distinguished from S. neomexicana by wide and rounded anterior apex of coxal plates I.
Discussion. The genus Stygomomonia with type-species S. latipes was described by Szalay in 1943 on the basis of males collected from the Dragan River ( Romania), and on the basis of female and deutonymph collected from Iad stream in Rimetea village ( Romania). Later, Walter (1947) described three new species of this genus – S. gracilis from Romania, S. jurassica and S. transversaria from Switzerland. According to Walter the main distinguishing features of these species is the appearance of sutures between the coxal plates and between coxal plates IV and the posterior part of ventral shield. Motas, Tanasachi and Botnariuc (1948) referred S. jurassica and S. transversaria to varieties of S. latipes . However, Viets (1956) considered S. gracilis and S. jurassica as junior synonyms of S. latipes and S. transversaria as subspesies of S. latipes . In 1971 Rensburg suggested that S. latipes transversaria is a junior synonym of S. latipes latipes . One more species S. szalayi Motas et Tanasachi, 1947 was also considered as junior synonym of S. latipes by Viets (1956).
Subsequently, Cook (1968) indicated that the features, on the basis of which Walter described S. gracilis , S. jurassica and S. transversaria , are variable and therefore unsuitable for taxonomic use. Ibidem, Cook also described the subgenus Allomomonia on the basis of the following features: first and second coxae fused with the third coxae and genital field of male located at extreme posterior end of ventral shield, but Smith (1989) demonstrated that adults of one species from the subgenus Allomomonia – S. (A.) antarkicola Smith have the first and second coxae separated from the third coxae, therefore this characteristic is not always suitable for distinguishing the species between the subgenera Stygomomonia and Allomomonia . According to Smith (1991) the most reliable characteristics for the subgenus Stygomomonia are the relatively long, slender tarsi on the first pair of legs (about equal to the genua in thickness and length), the relatively long claws on these segments and the subterminal position of the genital field on the ventral shield.
Describing S. occidentalis Smith (1991) indicated that this species together with two other Nearctic species of the subgenus Stygomomonia View in CoL – S. californiensis Smith and S. plana Cook and two Palaearctic species – S. latipes Szalay and S. rotunda Imamura are morphologically similar and probably belong to a monophyletic group, since the deutonymphs of these species have the dorsal shield subdivided into anterior and posterior plates, neither of which is further subdivided and glandularia-bearing sclerites in the genital field of adults fused into a single pair of large platelets. However, S. manchurica sp. n. does not belong to this group, because, as shown above, the deutonymph of S. manchurica sp. n. has another type of division of the dorsal shield, and it is possible that this species shares a common ancestry with S. neomexicana .
Determination of features that characterize species of the genus Stygomomonia View in CoL as representatives of the interstitial fauna is required for an understanding of their origin. Mitchell (1959) indicated that Stygomomonia View in CoL species have a hydrophilic integument with a lack of pigmentation (a feature that is typical for most interstitial water mites species) and modified leg I, which carries out sensory and mechanical functions (feeding or fixing by its claw). Smith (1991) indicated that the genus Stygomomonia View in CoL together with the Nearctic genus Cyclomomonia Smith and the Neotropical genera – Notomomonia Cook , Neomomonia Cook and Partidomomonia Cook share apotypical adaptations for the interstitial habitat, but nevertheless without indication of detail specifying these features. However, a dorso-ventrally flattened, sclerotised body as well as reduced eyes can indicate Stygomomonia View in CoL species as interstitial dwellers. It is possible, that putative ancestors of the genus Stygomomonia View in CoL inhabited swift waterways and were preadapted to the interstitial habitat.
The modern Holarctic distribution of the genus Stygomomonia has been attributed to an origin on the Laurasian palaeocontinent, which was completely separated from Gondwanaland in Late Cretaceous. The repeated glaciations of different Holarctic regions occurred over a whole Pleistocene period, resulting in restricted surviving populations to disjunct protected refugial areas south of the ice sheets ( Smith 1991). Colonization of interstitial habitats by putative ancestors, apparently, may be explained by a paradigm similar to the climatic-relict model (sometimes called relictual or Pleistocene effect model), which was proposed by Vandel (1964) for terrestrial troglobites of unglaciated karstic areas of temperate latitudes (see Holsinger 2000). Following Sidorov & Holsinger (2007), who adopted “Vandel’s rule” for troglomorphic dwellers of undergroundwaters in the Far East region, colonization of the interstitial habitats in the north-eastern Asia by vicariant remnants of Stygomomonia ancestors may be explained in the same way.
More complete investigations of Palaearctic species of the genus Stygomomonia , especially from northeastern Asia, are required to reconstruct dispersal pathways of these species through Eurasia. Finding a new species of the genus Stygomomonia in eastern Eurasia is of great significance not only for biogeography, but for understanding processes of origin and evolution of interstitial fauna in the Far East.
A detailed map of the distribution of Nearctic species of the subgenus Stygomomonia was compiled by Smith (1991), therefore a map with distribution of only Palaearctic species is given below.
An annotated checklist of the water mite species of the subgenus Stygomomonia Szalay *
* For detailed distributional data of Nearctic Stygomomonia (s. str.) species see Smith (1991). The distribution of S. latipes is given on the basis of available literature.
1. S. californiensis Smith, 1991
Type locality: Ventura River, Ventura County, California, U.S.A. ( Smith 1991).
Other localities: Moccasin Creek, Tuolumne County, California, U.S.A. ( Smith 1991). 2. S. cooki Smith, 1991
Type locality: stream in Perry City, Schuyler County, New York, U.S.A. ( Smith 1991). 3. S. hachiojiensis Imamura, 1959
Type locality: well in Hachiōji City, Tokyo Pref., Japan ( Imamura 1959).
Other localities: well in Kanazawa City, Ishikawa Pref., Japan ( Imamura 1959). 4. S. imamurai Smith, 1991
Type locality: Rancheria Creek, Mendocino County, California, U.S.A. ( Smith 1991). 5. S. latipes Szalay, 1943
Syn.: S. gracilis Walter, 1947
S. latipes transversaria Walter, 1947
S. szalayi Motas et Tanasachi, 1947
Type locality: Dragan River, Cluj County, Romania ( Szalay 1943).
Other localities: Great Britain: Kent River ( Gledhill 1979); Spain: Moros River (Valdecasas & Camacho 1984); France: Tech River basin ( Angelier 1953), Casaluna River, Golo River basin, Taravo River ( Angelier 1959), stream flowing into Longemer lake ( Schwoerbel 1965); Luxemburg: Wark River, Sauer River, Our River, Rhine River basin ( Gerecke et al. 2005); Switzerland: streams in Cantons Jura and Ticino ( Rensburg 1971); Italy: Sant' Ágata River basin, Rio Flumineddu River basin, Franco River basin, Imera River basin, Belice River basin, Pollina River basin, Cassibile River basin, Asinaro River basin ( Gerecke 1991); Germany: Innerste River, Gose River, Oker River, Radau River, Ecker River, Ilse River, Fulda River, Weser River basin ( Husmann 1956), Eschbach River, Wagensteinbach River, Ibenbach River, Dreisam River, Haslach River, Gutach River, Rhine River basin ( Schwoerbel 1959), Krebsbach River, Elbe River basin ( Schwoerbel 1965), Wutach River, Rhine River basin ( Schwoerbel 1971); Austria: Ibss River ( Husmann 1962); Poland: Raba River, Vistula River basin ( Biesiadka 1973), Kamionka stream, Vistula River basin ( Biesiadka 1979); Bosnia and Herzegovina: Bosna River, Danube River basin ( Motas et al. 1948); Montenegro: Zena River, Morača River basin ( Pesic 2003); Macedonia ( Schwoerbel 1963); Romania: Iad stream ( Szalay 1943), Barnar River, Bicaz River, Bistricioara River, Bistriţa River, Bârsa River, Bogata River, Valea Neagră River, Cerna River, Pecinişca River, Someşul Rece River, Tîrlungul River, Danube River basin (Motas et al. 1947); Doftăniţa River, Ialomiţa River basin ( Motas & Botea 1961); Azuga River, Ialomiţa River basin ( Motas & Botea 1962), Prisăcii River, Danube River basin ( Motas & Tanasachi 1963), Crisul Repede River basin ( Botea & Plesa 1968); Bulgaria: Loucovska River ( Petrova 1965), Mladezhka River ( Deltshev et al. 1998); Iran: nameless stream in Simk village, Shahrestanak River ( Pesic et al. 2005).
6. S. manchurica sp. n.
Type locality: Kedrovaya River, Kedrovaya Pad National Nature Biosphere Reserve, Khasansky District, Primory Territory, Russia.
Type locality: Whitewater Creek, Catron County, New Mexico, U.S.A. ( Cook 1968).
Other localities: British Columbia, Canada; Arizona, California, Idaho, Montana, Nevada, Oregon, Washington, Wyoming, U.S. A ( Smith 1991).
Type locality: Rogue River, Jackson County, Oregon, U.S.A. ( Cook 1968).
Other localities: California, U.S.A. ( Smith 1991).
Type locality: stream in Delaware County, Oklahoma, U.S.A. ( Cook 1968). 10. S. pallida Cook, 1968
Type locality: Jackson River, Bath County, Virginia, U.S.A. ( Cook 1968).
Other localities: New Brunswick, Nova Scotia, Canada; Maine, New York, Vermont, U.S. A ( Smith 1991).§
Type locality: Trinity River, Trinity County, California, U.S.A. ( Cook 1968).
Other localities: Oregon, Washington, U.S.A. ( Smith 1991). 12. S. riparia Habeeb, 1957
Type locality: Salmon River, Victoria County, California, U.S.A. ( Habeeb 1957).
Other localities: New Brunswick, Nova Scotia, Ontario, Canada; Alabama, Arkansas, Georgia, Indiana, Kentucky, Maine, Missouri, New Hampshire, New Jersey, New York, North Carolina, Ohio, Oklahoma, Pennsylvania, Vermont, Virginia, U.S.A. ( Smith 1991).
Type locality: well in Aioi City, Hyôgo Pref., Japan ( Imamura 1956).
Other localities: well in Hachiōji City, Tokyo Pref., Japan ( Imamura 1959), stream in Kangnung, South Korea ( Chung & Kim 1997).
Type locality: Satus Creek, Klickiitat County, Washington, U.S.A. ( Cook 1968).
Other localities: British Columbia, Canada; California, Montana, Nevada, Oregon, U.S.A. ( Smith 1991).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Stygomomonia manchurica Semenchenko
| Semenchenko, Ksenia A. 2008 |
S. occidentalis
| Smith 1991 |
S. jurassica
| Walter 1947 |
S. latipes transversaria
| Walter 1947 |
S. transversaria
| Walter 1947 |
S. szalayi
| Motas et Tanasachi 1947 |