Cambridgea reinga Forster & Wilton 1973
|
publication ID |
https://doi.org/ 10.5281/zenodo.276582 |
|
DOI |
https://doi.org/10.5281/zenodo.6191872 |
|
persistent identifier |
https://treatment.plazi.org/id/03DA6905-FFE8-FF86-FF19-8E58FBCFFA10 |
|
treatment provided by |
Plazi |
|
scientific name |
Cambridgea reinga Forster & Wilton 1973 |
| status |
|
Cambridgea reinga Forster & Wilton 1973
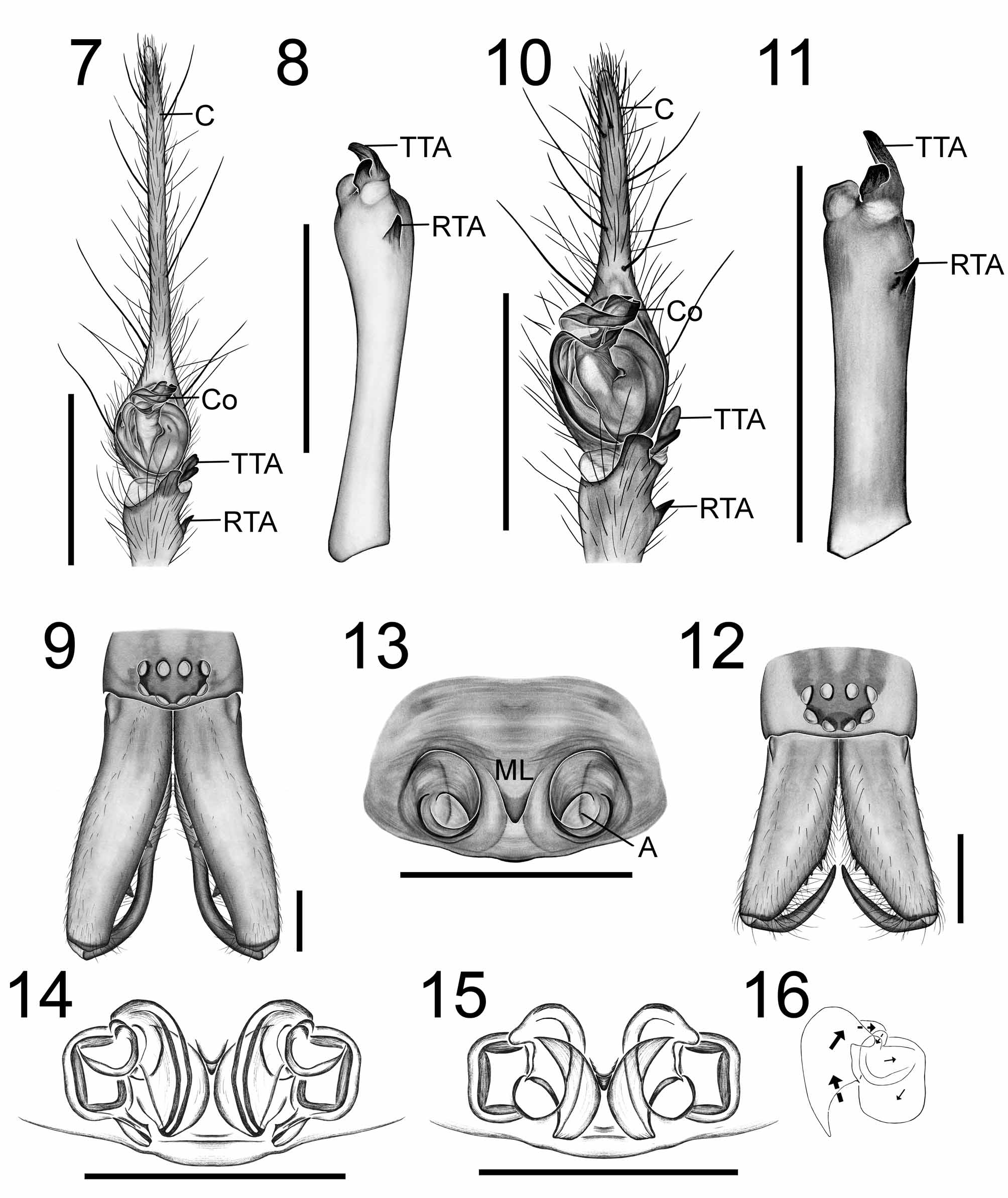
( Figs 7–16 View FIGURES 7 – 16 )
Cambridgea reinga Forster & Wilton 1973: 151 , figs 456–457 (description of female).
Nanocambridgea grandis Blest & Vink 2000: 21 , figs 57–58 (description of male). NEW SYNONYMY.
Type specimens. Holotype Ψ, not examined, NEW ZEALAND: Northland: Cape Reinga [34°26'S, 172°41'E], 7 January 1967, R.R. Forster leg., Otago Museum, New Zealand.
Holotype ɗ, Nanocambridgea grandis , examined. NEW ZEALAND: Northland: Cape Reinga [34°26'S, 172°41'E,], 10–13 December 1995, J.W. Early & R.F. Gilbert leg., ( AMNZ 5031), 2 male paratypes, same locality, date and collector ( AMNZ 6568).
We did not examine the type of Cambridgea reinga ; the illustrations and descriptions by Forster & Wilton (1973) are clear enough to be sure of the species identity.
Other material examined. NEW ZEALAND: Northland: Shenstone Block [34°31.540'S, 172°46.674'E], 17 July–15 August 2008, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001607) (GenBank HQ316174 View Materials ); 1 Ψ ( MONZ AS.001608) (GenBank HQ332444 View Materials ); 22 October–21 November 2008, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001604); 9 April–7 May 2009, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001605) (GenBank HM439089 View Materials ). Spirits Bay [34°28.657'S, 172°52.715'E], 12 January–12 February 2007, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001590). Kerr Point [34°27.726'S, 172°52.962'E], 12 January–12 February 2007, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001609). Taputaputa Site B [34°26.732'S, 172°43.359'E], 17 July–15 August 2008, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001614); 1 ɗ ( MONZ AS.001615); 22 October–21 November 2008, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001612); 1 ɗ ( MONZ AS.001611); 1 ɗ ( MONZ AS.001613). Taputaputa Site A [34°26.527'S, 172°42.162'E], 17 July–15 August 2008, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001632); 22 October–21 November 2008, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001631); 15 January–13 February 2009, O.J.-P. Ball leg., 2 ɗ ( MONZ AS.001640). Unuwhao Site A [34°26.139'S, 172°53.279'E], 12 January–12 February 2007, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001610); 1 immature ( MONZ AS.001629). Cape Reinga [34°26'S, 172°41'E], 10–13 December 1995, J.W. Early & R.F. Gilbert leg., 2 ɗ ( AMNZ 6568). North Cape [34°24.947'S, 173°01.446'E], 22 October–21 November 2008, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001630); 1 ɗ ( MONZ AS.001605) (GenBank HQ316173 View Materials ); 15 January–13 February 2009, O.J.-P. Ball leg., 1 ɗ ( MONZ AS.001606); 1 ɗ ( MONZ AS.001601) (GenBank HM439090 View Materials ); 1 ɗ ( MONZ AS.001603) (GenBank HM439091 View Materials ). Kohuronaki B [34°29.922'S, 172°50.586'E], 22 October–21 November 2008, O.J.-P. Ball leg., 1 ɗ, 1 Ψ ( MONZ AS. 001600); 15 January–13 February 2009, O.J.-P. Ball leg., 1 Ψ ( MONZ AS.001856) (GenBank HQ316175 View Materials ).
Diagnosis. Cambridgea reinga can be distinguished from other Cambridgea species by the arrangement of the male pedipalp ( Figs 7, 8, 10, 11 View FIGURES 7 – 16 ), particularly the terminal tibial apophysis ( Figs 8, 11 View FIGURES 7 – 16 ), which is well sclerotised (weak and foliate in most other Cambridgea species), and the epigynum of the female ( Figs 13–16 View FIGURES 7 – 16 ), which has large, paired atria and a median lobe about half way down the median furrow. The legs (relative to the carapace length) are longer than other Cambridgea species.
Redescription. Colour: carapace orange-brown with contrasting broad, blackish lateral bands and median band divided on midline by a narrow pale stripe; sternum orange-brown; abdomen pale yellow-brown, darker laterally and posteriorly; legs yellow-brown with weak annulations on leg 4.
Two male forms known (see Biology below). Chelicerae of larger male specimens are porrect, those of smaller males and females are not; chelicerae with four promarginal teeth and two retromarginal teeth. Ratio of AME.ALE.PME.PLE = 14.20.15.16; viewed dorsally, anterior eye row recurved, posterior eye row slightly procurved. Colulus large, wider than long, with many hairs. Male pedipalp ( Figs 7, 8, 10, 11 View FIGURES 7 – 16 ) tibia with retrolateral apophysis and tapering terminal apophysis; cymbium extending well beyond bulb (at least twice length of bulb), embolus extending up prolateral margin and across top of bulb, conductor large and curled around embolus; median apophysis absent. Epigynum with large, paired atria, median lobe about half way down median furrow ( Fig. 13 View FIGURES 7 – 16 ); internal genitalia receptaculate with short ducts ( Figs 14–16 View FIGURES 7 – 16 ).
Dimensions. Female Northland, Taputaputa Site A (MONZ AS.001631) (male Northland, Taputaputa Site B (MONZ AS.001611)): total length 9.13 (10.06); carapace length 3.87 (4.64), width 2.94 (3.25), height 1.70 (1.55); abdomen length 4.95 (5.73), width 3.10 (2.94); sternum length 2.01 (2.63), width 1.78 (2.01). Size range: female body length 5.9–9.3 (mean 8.1, n=6), male body length 6.0–11.3 (mean 8.8, n=16). Porrect male body length 6.9– 11.3 (mean 9.7, n=13). Non-porrect male body length 6.0–6.8 (mean 6.5, n=3).
DNA sequences. Mitochondrial COI (GenBank accession numbers HM439089 View Materials – HM439091 View Materials , HQ316173 View Materials – HQ316175 View Materials , HQ332444 View Materials ). The seven sequences varied by only 2%, which is well within intraspecific variation observed in other spiders ( Robinson et al. 2009), and a female and two males from Shenstone Block (GenBank HM439089 View Materials , HQ316174 View Materials , HQ332444 View Materials ) and a male from North Cape (GenBank HM439091 View Materials ) had identical COI sequences. We observed 26 variable nucleotide positions; 25 transitions, one transversion and all were synonymous substitutions. The COI fragment between the primers C1-J-1718-spider and C1-N-2568 is usually 850 bp in other spiders (e.g., Vink et al. 2009; Vink & Dupérré 2010), but for Cambridgea reinga it was only 844 bp. Two sequential codons are absent at 268 bp into the fragment we sequenced in C. reinga ; these codons are present in Stiphidion facetum Simon 1902 ( Spagna & Gillespie 2008), the type species of the family Stiphidiidae . We do not believe we had amplified a pseudogene as there were no stop codons in the sequence.
Biology. Three of the male C. reinga examined (MONZ AS.001590, AS.001605, AS.001607, AS.001608) were smaller (6.0– 6.8 mm body length), had smaller non-porrect chelicerae in which the fangs barely met ( Fig. 12 View FIGURES 7 – 16 ), the distal portion of the cymbium (relative to the bulb) was much shorter ( Fig. 10 View FIGURES 7 – 16 ) and the palpal tibia was shorter relative to its width ( Fig. 11 View FIGURES 7 – 16 ). However, the palpal sclerites ( Figs 7, 10 View FIGURES 7 – 16 ) and tibial apophyses ( Figs 8, 11 View FIGURES 7 – 16 ) are the same shape, position and relative size to one another, and the colour patterns are the same in both forms of the male. The COI sequences of smaller C. reinga specimens (GenBank HQ316173 View Materials , HQ316174 View Materials , HQ332444 View Materials ) matched those of larger specimens (GenBank HM439090 View Materials – HM439091 View Materials ) and were identical for three specimens (GenBank HM439091 View Materials , HQ316174 View Materials , HQ332444 View Materials ). Two forms of male have been noted in other Cambridgea species. In C. antipodiana (White 1849) , the chelicerae are large and porrect in one form of male, while the chelicerae in the other form are similar to those of the female ( Forster & Wilton 1973). In C. annulata Dalmas 1917 , males are either large and the distal portion of the cymbium is relatively elongated, or males are small and have a shorter distal portion of the cymbium (Sirvid, personal observation). The differences in male Cambridgea forms could be due to food availability, which is known to affect size and morphology in some spider species ( Jakob & Dingle 1990).
All specimens collected were from pitfall traps or pan traps in native forest, exotic pine forest ( Pinus spp.), or dense scrub with flax ( Phormium spp.), manuka ( Leptospermum scoparium J.R. Forst. & G. Forst. ) and kanuka ( Kunzea ericoides (A. Rich.) Joy Thomps. ). From this we can assume that C. reinga probably live close to the ground. It is unknown whether this species builds sheet webs typical of other Cambridgea species ( Forster & Wilton 1973; Blest & Taylor 1995). Adults have been found throughout the year.
Distribution. Cambridgea reinga is known only from Te Paki Ecological District at the northern tip of Northland, New Zealand, and is likely to be endemic to that region, which is an area of high endemism (e.g., de Lange et al. 2003; Larochelle & Larivière 2005; Marshall & Barker 2007; Chapple et al. 2008; Buckley & Bradler 2010).
Remarks. The collection of male and female specimens from the same area, the same colour pattern of both sexes and COI sequences that vary by only 2%, lead us to conclude that N. grandis is a junior synonym of C. reinga . We have retained C. reinga in Cambridgea because it has the blackish median and lateral stripes on the carapace that are typical of species of Cambridgea ; this colour pattern is lacking in Nanocambridgea gracilipes , which is the type species of the genus Nanocambridgea Forster & Wilton 1973 . Also, the tibial apophyses of C. reinga are more like those of other Cambridgea and not reduced as they are in N. gracilipes . Forster & Wilton (1973) distinguished Nanocambridgea from Cambridgea by the presence in Nanocambridgea of plumose hairs on the legs, four teeth on the promargin of the chelicerae and a ventral stridulating organ on the pedicel and abdomen of males. Blest & Vink (2000) added to these characters the greater ratio of length of leg 1 to carapace (> 7.0) in male Nanocambridgea . However, all of these characters except the ventral stridulating organ prove not to be diagnostic. Plumose hairs are present on the legs of some species of Cambridgea ( Blest & Vink 2000) , leg 1/carapace ratio of male Cambridgea reinga is greater, not less, than in Nanocambridgea gracilipes , and both genera have four teeth on the promargin and two teeth on the retromargin. Forster and Wilton (1973), in their generic description of Cambridgea , had incorrectly reported two teeth on the promargin and three to five teeth on the retromargin, but in their species descriptions they correctly listed two teeth on the retromargin and between three and five teeth on the promargin. This leaves only the ventral stridulation organ of Nanocambridgea versus the dorsal stridulation organ of Cambridgea as separating the two genera; however, proper assessment of the validity of the genus Nanocambridgea is beyond the scope of this study, but could be resolved by a phylogenetic analysis of New Zealand Stiphidiidae ( Cambridgea , Nanocambridgea and Ischalea L. Koch 1872 ) and other similar genera, such as the Australian Procambridgea Forster & Wilton 1973 , to see if N. gracilipes falls within Cambridgea .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Cambridgea reinga Forster & Wilton 1973
| Vink, Cor J., Fitzgerald, Brian M., Sirvid, Phil J. & Dupérré, Nadine 2011 |
Nanocambridgea grandis
| Blest 2000: 21 |
Cambridgea reinga
| Forster 1973: 151 |