Alpheus perlas
View in CoL
sp. nov.
Figures 1–4
View FIGURE 1
View FIGURE 2
View FIGURE 3
View FIGURE 4
Type material.
Holotype: male (cl 6.7 mm), MNHN-IU- 2019-2301, Panama,
Pacific
coast,
Bay
of Panama, Las Perlas Archipelago, Isla Casayeta, 8°31’14.6”N 79°01’26.4”W, small shallow bay with mudflat fringed by rocks and mangroves, depth: less than 0.5 m at low tide, in burrow, suction pump, leg. A. Anker, 20 April 2015.
GoogleMaps
Description. Small-sized species of
Alpheus
(holotype at cl 6.7 mm). Carapace smooth, glabrous, not setose, not pubescent. Rostrum very short, subtriangular, as long as broad at base, subacute distally, not reaching mid-length of first article of antennular peduncle; rostral carina feebly developed, rounded dorsally, gradually fading posteriorly to eye level ( Fig. 1A, B
View FIGURE 1
). Orbital hoods moderately developed, not particularly swollen, semi-open frontally, with anterior margin broadly rounded; frontal margin between rostrum and orbital hoods slightly concave; adrostral furrows absent ( Fig. 1A, B
View FIGURE 1
). Pterygostomial angle rounded, not protruding anteriorly ( Fig. 1B
View FIGURE 1
); cardiac notch well developed.
Telson very broad, subrectangular, tapering distally, about 1.8 times as long as maximal width, with lateral margins slightly convex, not constricted; dorsal surface with two pairs of short cuspidate setae both inserted far from lateral margin, first pair at about mid-length, second pair at about 0.8 length of telson; posterior margin broadly rounded, without spiniform setae; each posterolateral angle with pair of slender spiniform setae, mesial about three times as long as lateral ( Fig. 1C
View FIGURE 1
).
Antennular peduncle moderately stout; stylocerite broad, inflated, with small sharp point almost reaching distal margin of first article; ventromesial carina with strong, anteriorly directed tooth; second article moderately elongate, almost twice as long as wide; lateral antennular flagellum with secondary ramus fused to main ramus over most of its length, with numerous groups of aesthetascs starting at eighth joint ( Fig. 1A, B, D
View FIGURE 1
). Antenna with basicerite moderately stout, its distoventral margin bearing small acute tooth; scaphocerite with well-developed blade, straight to faintly concave lateral margin and strong broad distolateral tooth overreaching blade but not distal margin of antennular peduncle; carpocerite reaching far beyond both scaphocerite and end of antennular peduncle ( Fig. 1A, B
View FIGURE 1
).
Mouthparts typical for genus in external observation. Third maxilliped moderately stout; coxa with projecting, distally subacute lateral plate; antepenultimate article flattened ventrolaterally, with straight mesial margin; penultimate article relatively short, widening distally, about twice as long as maximal (distal) width; ultimate article tapering distally, with numerous rows of short serrulate setae and longer stiff setae, distally unarmed; arthrobranch well developed, with smaller accessory branch reminiscent of small pleurobranch ( Fig. 1E, F
View FIGURE 1
).
Male major cheliped overall moderately robust for species of
A. edwardsii
group; ischium very short, stout, unarmed; merus relatively stout, about 2.4 times as long as wide, distodorsal margin ending bluntly, mesial margin smooth, without spiniform setae, unarmed (without tooth) on distomesial margin; carpus very short, cup-shaped; chela elongate, slightly compressed; palm oval in cross-section, about 1.5 times as long as fingers; palm surface mostly smooth, without tubercles, not excessively setose; dorsal transverse groove broad, adjacent shoulder sloping smoothly, not overhanging groove; ventral transverse groove well marked, however, not significantly extending onto mesial surface, adjacent shoulder broadly rounded, not protruding anteriorly to overhang groove; mesial face of palm with deep longitudinal groove stretching from mesial extension of dorsal transverse groove posteriorly, almost reaching linea impressa; lateral face of palm with similar longitudinal groove stretching from lateral extension of dorsal transverse groove towards posterior half of palm, reaching linea impressa; dactylus slightly longer than pollex, distally rounded, not twisted, with short, moderately stout, distally truncate plunger, latter with numerous stamen-shaped sensillae; adhesive disks small ( Fig. 2
View FIGURE 2
A–D).
Male minor cheliped significantly smaller than major cheliped, with much weaker, shorter chela; ischium short, stout, unarmed; merus somewhat slenderer, about 2.8 times as long as wide, otherwise similar to that of major cheliped; carpus also noticeably longer, cup-shaped; chela not particularly elongate, moderately slender, with palm subcylindrical in cross-section, somewhat shorter than fingers; palm surface smooth, without grooves or notches; fingers subequal in length, crossing distally, simple, not balaeniceps; dactylus not expanded, more conical distally, with simple blade-like cutting edge; pollex shallowly excavated along lateral side cutting edge; adhesive disks not discernable ( Fig. 2E, F
View FIGURE 2
). Female minor cheliped unknown.
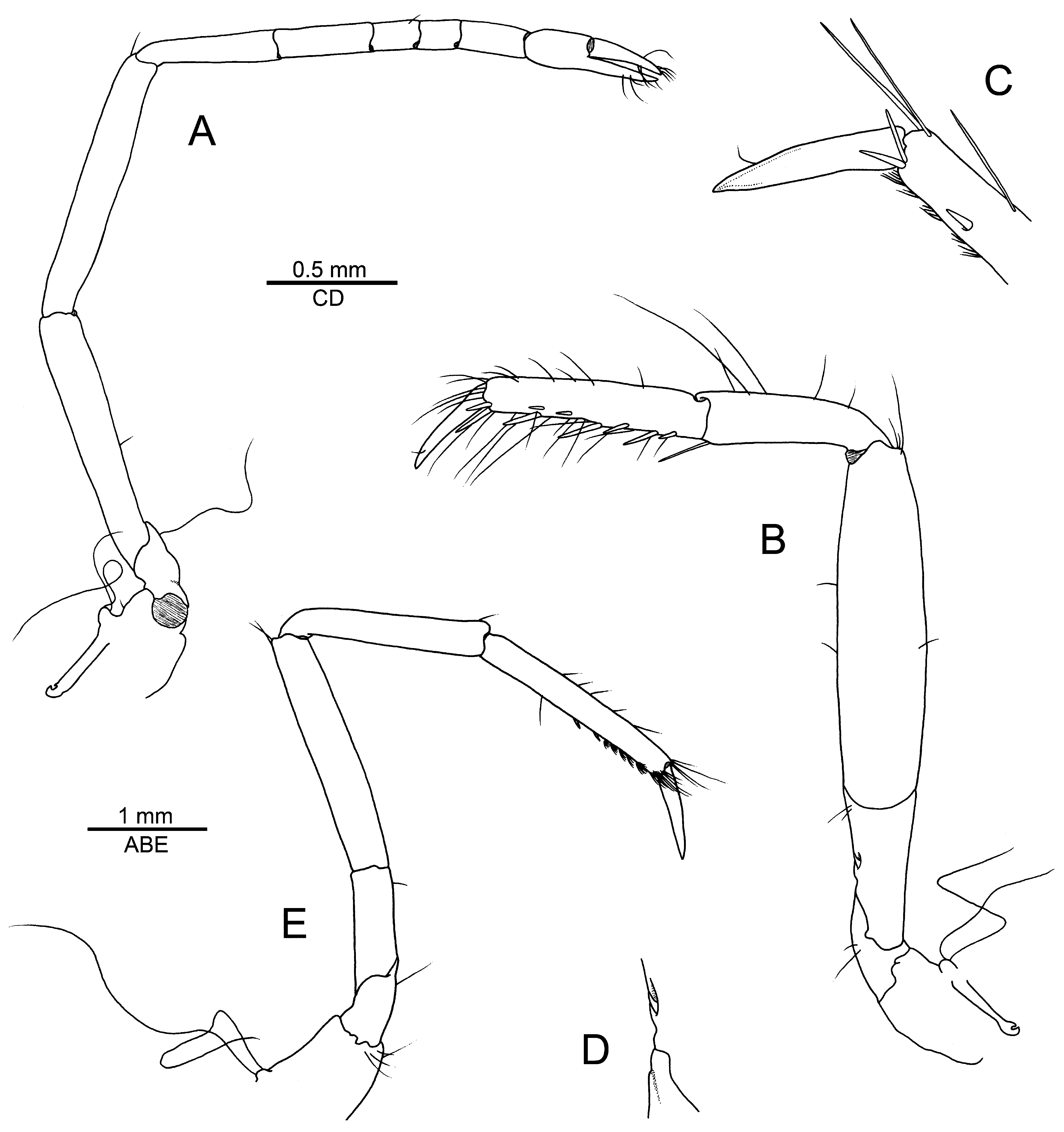
Second pereiopod moderately slender; ischium and merus subequal in length; carpus with five subdivisions, first longest, ratio of carpal subdivisions approximately equal to 3.2/2.4/1.2/1.0/1.8; chela as long as two distal-most carpal subdivisions combined ( Fig. 3A
View FIGURE 3
). Third pereiopod rather stout; ischium with very small cuspidate seta on ventrolateral surface; merus about 3.5 times as long as maximal width, slightly inflated, unarmed; carpus about half- length of merus, much slenderer, with long stiff distoventral seta; propodus noticeably longer than carpus but shorter than merus, with seven or so irregularly inserted, stout spiniform setae along ventral and ventrolateral margin, in addition to one pair of spiniform setae near dactylar base; dactylus about half-length of propodus, stout, conical, very slightly expanded and curved ( Fig. 3
View FIGURE 3
B–D). Fourth pereiopod generally similar to third, slightly slenderer. Fifth pereiopod much slenderer than third and fourth; ischium unarmed; merus not inflated, about six times as long as wide; carpus slightly less than 0.8 times length of merus; propodus with three spiniform setae along ventromesial margin and one distal pair of spiniform setae near articulation with dactylus; distal third of propodus with welldeveloped rows of serrulate setae on ventrolateral surface; dactylus about 0.4 length of propodus, conical, simple, almost straight ( Fig. 3E
View FIGURE 3
).
Male first pleopod with protopod bearing conspicuous subacute process on subdistal ventral (posterior) margin, distally carrying single thickened seta; endopod very small, fringed with setae ( Fig. 1G
View FIGURE 1
). Male second pleopod with protopod bearing similar subacute process, but situated slightly more distally on ventral (posterior) margin and without thickened seta; appendix interna longer than appendix masculina, latter furnished with numerous stiff setae on apex and with some stiff setae also on subapical surface and distal half ( Fig. 1H, I
View FIGURE 1
).
Uropod with both mesial and lateral lobes of protopod ending in subacute tooth; exopod and endopod very broad, ovoid; exopod with fairly stout distolateral tooth adjacent to strong distolateral spiniform seta and unarmed distal margin; diaeresis somewhat sinuous, with broadly triangular lobe adjacent and mesial to distolateral spiniform seta; endopod with short row of small spiniform setae on distal margin ( Fig. 1J
View FIGURE 1
).
Colour pattern. General body background semitransparent creamy with straw-yellow tinge; carapace with fields of reddish chromatophores, e.g. in postrostral area, along anterolateral margin and on posterodorsal surface; pleon with reddish chromatophores forming broad, transverse, rather diffuse bands, less conspicuous on last pleonite; telson and uropods also with some reddish chromatophores; chelipeds creamy-yellowish, tips of major chela fingers pale pinkish; walking legs semitransparent ( Fig. 4
View FIGURE 4
).
Etymology. The new species is named after the type locality, Las Perlas Archipelago; used as a plural noun (
perlas
, from the Spanish word perla = pearl) in apposition.
Ecology.
Alpheus perlas
sp. nov. appears to be associated with burrows of unknown hosts on shallow subtidal mudflats. Sympatric burrowing animals collected at the type locality included
Axianassa christyi Anker & Pachelle, 2016
,
Neocallichirus
sp. [in study], as well as several unidentified echiurans (possibly
Ochetostoma
sp. or
Listriolobus
sp.).
Remarks.
Alpheus perlas
sp. nov. plainly belongs to the
A. edwardsii
species group, as redefined by Anker et al. (2009) for the dorsal notch of the major chela palm extends posteriorly on the mesial face ( Fig. 2A
View FIGURE 2
). Within the
A. edwardsii
group, the new species belongs to a non-monophyletic assemblage of species, in which (1) the male minor chela is not balaeniceps (= fingers do not possess the so-called “balaeniceps ridges” furnished with rows of densely inserted thick plumose setae); (2) the rostral carina does not form a sharply delimited, V- or U-shaped, flattened post-rostral plate; (3) the ventral shoulder of the major chela is not protruding anteriorly, overhanging the ventral notch; (4) the distoventral margin of the third pereiopod merus is blunt, i.e. not ending in a sharp tooth; and (5) the antepaenultimate article of the third maxilliped is not significantly expanded. In the eastern Pacific, the only species of the
A. edwardsii
group that have this combination of characters are:
A. latus Kim & Abele, 1988
,
A. burukovskyi Anker & Pachelle, 2015
,
A. galapagensis Sivertsen, 1933
(=
A. canalis Kim & Abele, 1988
),
A. millsae Anker, Hurt & Knowlton, 2007
,
A. mazatlanicus Wicksten, 1983
,
A. colombiensis Wicksten, 1988
(=
A. hamus Kim & Abele, 1988
),
A. agrogon Ramos, 1997
, and
A. spinicaudus Lockington, 1878
[nomen dubium] ( Kim & Abele 1988; Anker et al. 2007; Anker & Pachelle 2015). All other eastern Pacific species of this group can be separated from
A. perlas
sp. nov. by at least one of the five characteristics mentioned above, with most of these taxa having either strongly balaeniceps minor chelae in males or an abruptly delimited post-rostral plate ( Kim & Abele 1988; Anker & Pachelle 2015; Salgado-Barragán et al. 2017).
Alpheus latus Kim & Abele, 1988
View in CoL
and
A. burukovskyi Anker & Pachelle, 2015
View in CoL
are two eastern Pacific species that are superficially most similar to the new species; both are also associated with muddy bottoms ( Kim & Abele 1988; Anker & Pachelle 2015; A. Anker, pers. obs.). However,
A. perlas
View in CoL
sp. nov. differs from each of them by a series of morphological characters. For instance,
A. perlas
View in CoL
sp. nov. differs from
A. latus
View in CoL
by (1) the shorter, less marked rostral carina (vs. longer and more marked in
A. latus
View in CoL
); (2) the shallower adrostral furrows (vs. deeper ones in
A. latus
View in CoL
); (3) the major chela dactylus rounded (vs. angular in
A. latus
View in CoL
); (4) the major chela palm with a moderately deep ventral notch, only slightly extending into the mesial surface of the palm (vs. with a deeper notch, more sharply delimited proximally and extending further into the mesial surface of the palm, in
A. latus
View in CoL
); (5) the minor chela palm stouter, about 1.6 times as long as high (vs. slenderer, almost twice as long as high, in
A. latus
View in CoL
); (6) the first article of the second pereiopod carpus about four times as long as wide (vs. almost six times in
A. latus
View in CoL
); and (7) the third pereiopod dactylus only slightly expanded, subconical (vs. spatulate in
A. latus
View in CoL
) (cf. Kim & Abele 1988: fig. 37).
Similarly,
A. perlas
View in CoL
sp. nov. differs from
A. burukovskyi
View in CoL
by (1) the distinctly longer second article of the antennular peduncle, about 1.7 times as long as wide (vs. shorter, about 1.3 times as long as wide, in
A. burukovskyi
View in CoL
); (2) the scaphocerite not reaching the distal margin of the antennular peduncle (vs. slightly overreaching it in
A. burukovskyi
View in CoL
); (3) the penultimate article of the third maxilliped without elongate thickened setae on its mesial surface (vs. with these setae in
A. burukovskyi
View in CoL
); (4) the dactylus plunger of the major chela very short, subtriangular, distally truncate, with the anterior angle not well marked, not at the same level as the posterior angle (vs. much more robust, longer, more square-shaped, not truncate distally, with the anterior angle well marked, at the same level as the posterior angle, in
A. burukovskyi
View in CoL
); (5) the posterior margin of the telson without row of spiniform setae (vs. with spiniform setae in
A. burukovskyi
View in CoL
); and (6) the uropodal endopod with just a few spiniform setae on the distal margin (vs. with a complete row of spiniform setae in
A burukovskyi
View in CoL
) (cf. Anker & Pachelle 2015: figs. 1, 2).
In addition,
A. perlas
sp. nov. differs from both
A. latus
and
A. burukovskyi
by its bland, pale-yellow and reddish-banded colour pattern, contrasting to the much darker olive-greenish colouration of
A. latus
(A. Anker, unpublished data) and the uniform bright red-orange colouration of
A. burukovskyi
( Anker & Pachelle 2015: fig. 3).
Alpheus perlas
View in CoL
sp. nov. is not closely related to
A. galapagensis
View in CoL
,
A. millsae
View in CoL
,
A. mazatlanicus
View in CoL
,
A. colombiensis
View in CoL
and
A. agrogon
View in CoL
, differing from each of them by a great number morphological characters, mainly on (1) the frontal region of the carapace (e.g. both
A. galapagensis
View in CoL
and
A. millsae
View in CoL
with a strong sharp rostral carina,
A. agrogon
View in CoL
with a short post-rostral crest, see below); (2) the major chela (e.g.,
A. galapagensis
View in CoL
and
A. millsae
View in CoL
both with a very stout dactylus plunger,
A. mazatlanicus
View in CoL
and
A. colombiensis
View in CoL
both with the distally angular dactylus,
A. agrogon
View in CoL
with the major chela dactylus reaching well beyond the pollex); (3) the minor chela (especially
A. mazatlanicus
View in CoL
and
A. colombiensis
View in CoL
, both with elongate fingers of the minor chela); and (4) the third pereiopod (especially
A. mazatlanicus
View in CoL
and
A. colombiensis
View in CoL
, both with spatulate dactyli (cf. Wicksten 1988; Kim & Abele 1988; Ramos 1997; Anker et al. 2007). The new species also differs from
A. galapagensis
View in CoL
,
A. millsae
View in CoL
,
A. mazatlanicus
View in CoL
, and
A. colombiensis
View in CoL
by its colour pattern (cf. Anker et al. 2007: fig. 7, for colour patterns of
A. galapagensis
View in CoL
and
A. millsae
View in CoL
; A. Anker, unpublished data, for
A. mazatlanicus
View in CoL
and
A. colombiensis
View in CoL
); the colour pattern of
A. agrogon
View in CoL
remains unknown.
It must be noted here that Ramos’ (1997) description of
A. agrogon
contains some inaccuracies, especially in the illustration of the rostro-orbital area of the carapace. Ramos (1997: fig. 1B) illustrated an interrupted rostral carina followed by a V-shaped post-rostral plate, similar to the configuration seen in some species of the
A. armillatus
complex. The first author (AA) examined the holotype and the only known specimen of
A. agrogon
, deposited in the National Museum of Natural History, Smithsonian Institution, Washington DC, USA ( USNM). The holotype of
A. agrogon
indeed has a marked rostral carina interrupted between the orbital hoods, i.e. separating the proper rostral carina from a short post-rostral (in fact, post-orbital) crest, gradually flattening posteriorly; however, it does not possess a triangular V-shaped plate, as figured by Ramos (1997: fig. 1B). Collection of more material in the shallow intertidal areas of Gorgona Island off Colombia, the type locality of
A. agrogon
, will be necessary to confirm the validity and uniqueness of this character and to elucidate the phylogenetic relationships of this species.
Alpheus spinicaudus Lockington, 1878
from the Gulf of California, known only from its extremely inadequate original description by Lockington (1878), was listed as a nomen dubium by De Grave & Fransen (2011). The major chela of this species was described as having a short dactylus, “moving obliquely”, whereas the posterior margin of the telson was described as bearing “posterior spinules” ( Lockington 1878; Kim & Abele 1988); both of these features are not observed in
A. perlas
View in CoL
sp. nov. Importantly, the obliquely moving major chela dactylus is more typical to the members of the
A. macrocheles Hailstone, 1835
View in CoL
species group ( Coutière 1899; Banner & Banner 1982). Unfortunately, Lockington’s alpheid specimens were lost in the fire of the old California Academy of Sciences building following a major earthquake, in 1906 ( Wicksten 1994).
None of the Atlantic species of the
A. edwardsii
View in CoL
group with the five characteristics mentioned above, e.g.
A. viridari Armstrong 1949
View in CoL
,
A. nuttingi ( Schmitt, 1924)
View in CoL
and
A. estuariensis Christoffersen, 1984
View in CoL
, seem to be closely related to
A. perlas
View in CoL
sp. nov. (cf. Schmitt 1924; Armstrong 1949; Christoffersen 1984; Anker et al. 2007; Anker 2012). Among the Indo-West Pacific species of the
A. edwardsii
View in CoL
group, only
A. dolerus Banner, 1956
View in CoL
has some superficial similarities with the new species, but can be easily separated by the configuration of the rostro-orbital region and frontal appendages (cf. Banner & Banner 1982). Remarkably,
A. perlas
View in CoL
sp. nov. is also superficially similar to
A. echiurophilus Anker, Komai & Marin, 2015
View in CoL
, a recently described species associated with burrowing echiurans across the Indo-West Pacific. These similarities include the general shape of the body, frontal region (including rostro-orbital area), chelipeds, walking legs, as well as colour pattern (Anker et al. 2015). However, this resemblance seems to be purely convergent since
A. echiurophilus
View in CoL
does not belong to the
A. edwardsii
View in CoL
group, but to the
A. leviusculus Dana, 1852
View in CoL
group (“L clade” in Anker et al. 2009), as discussed by Anker et al. (2015). This means that in
A. echiurophilus
View in CoL
, the transverse groove does not extend posteriorly on the mesial surface of the major chela palm (Anker et al. 2015: figs. 2B, 4B), as it does in the new species from Panama ( Fig. 2A
View FIGURE 2
). Whether
A. perlas
View in CoL
sp. nov. is also associated with echiurans remains to be demonstrated by a more intensive sampling of echiuran burrows in the eastern Pacific.
The presence of a subacute process on the subdistal-ventral margin of the protopod of the first and second pleopods in
A. perlas
sp. nov. ( Fig. 1G, H
View FIGURE 1
) may be the only true autopomorphic feature of the new taxon. However, the absence of such a process in other species of
Alpheus
needs confirmation as the protopods are not always carefully observed and/or drawn; this is especially true for the protopod of the first pleopod. In the authors’ personal experience, most American species of
Alpheus
do not have such a process on the first and second pleopods. The only other conspicuous modification of the pleopodal protopods is observed in
A. colombiensis
, in which they are armed with row(s) of stout sharp teeth, especially in ovigerous females ( Wicksten 1988; Kim & Abele 1988).