Zschokkella
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4269.1.3 |
|
publication LSID |
lsid:zoobank.org:pub:1266D96E-57FC-4768-A347-A9D395FCDBCF |
|
DOI |
https://doi.org/10.5281/zenodo.6028601 |
|
persistent identifier |
https://treatment.plazi.org/id/007687A5-ED0F-FFF5-AFFC-DE5DFD61FDDF |
|
treatment provided by |
Plazi |
|
scientific name |
Zschokkella |
| status |
|
Zschokkella sp.
Host: Serranus scriba Linnaeus, 1758 painted comber ( Perciformes : Serranidae ).
Locality: Mediterranean off Tunisia, Sidi Daoud , Gulf of Tunis (37° 01’ N 10° 55’ E) GoogleMaps .
Site of infection: Within gall bladder.
Prevalence: The overall prevalence is 5.6% (10/180). The frequency of infection is distributed as following, 03/2012: 10% (3/30); 04/2012: 3.3% (1/30); 05/2012: 16.7% (5/30); 06/2012: 3.3% (1/30); 07/2012: 0% (0/30); 08/2012: 0% (0/30) ( Table 10).
Mean intensity: 53±15 spores/20µl bile/infected fish (+++++) ( Table 10).
Vouchers: Digitized photos of spores are deposited in the parasitological collection of the Museum National d’Histoire Naturelle (MNHN), Paris, Coll. No. ZS 139.
Morphological description. Vegetative stages. No vegetative stages are observed for this parasite.
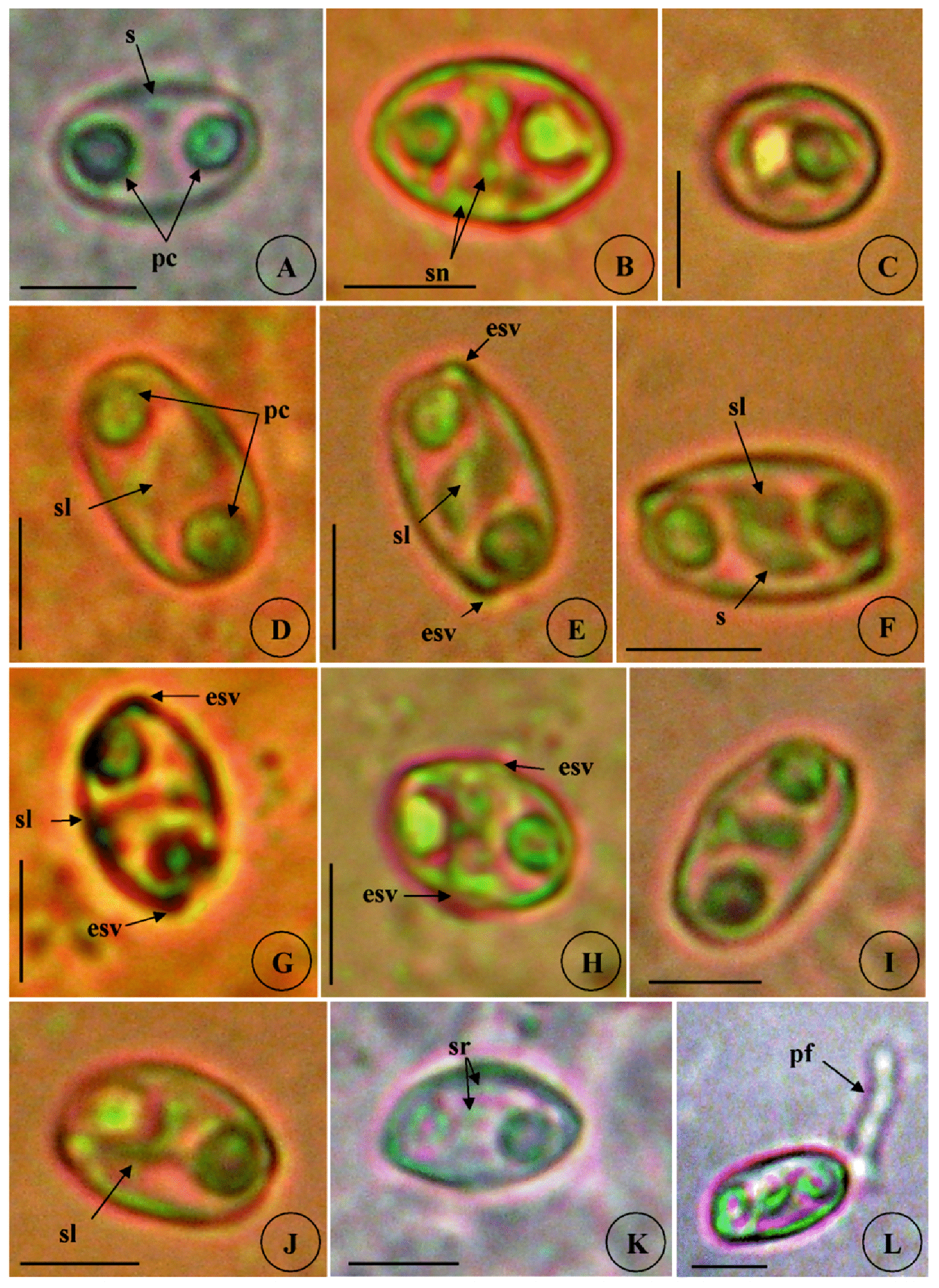
Myxospores. Spores typical of the genus Zschokkella (n = 30 fresh spores). Mature spores are cylindroellipsoidal with regular rounded edges in frontal ( Figs. 6 View FIGURE 6 A,D, 8G), lateral ( Fig. 6 View FIGURE 6 C), and sutural views ( Figs. 6 View FIGURE 6 D–E, 8I) and ovoid in valvular view ( Figs. 6 View FIGURE 6 B, 8H), measuring 16.8±0.9 (15.2–17.8) µm in length and 10.9±0.8 (9.8–12.1) µm in width. Suture line is obvious and mostly curved, circulates between the polar capsules and crosses the rounded ends of the spore ( Figs. 6 View FIGURE 6 D–E, 8I) or sometimes runs quite close to the mid-spore length ( Fig. 6 View FIGURE 6 G). Spores valves characterize with 7 to 8 surface ridges arranged parallel and follow the suture contour ( Figs. 6 View FIGURE 6 K, 8H). A binucleate sporoplasm is confined to a small portion between the capsules ( Figs. 6 View FIGURE 6 A–B,E–F, 8G). Two polar capsules are equal in size, sub-spherical to spherical locate at the two opposite poles of the spore and measuring 4.3±0.2 (4.0–4.5) µm in length and 4.2±0.2 (4.0–4.5) in width µm (n = 30). The discharge canals open sublaterally in two pores which are located in opposite angles and inserts into a fine elevation of the shell valve surface ( Figs. 6 View FIGURE 6 E–H, 8I). The polar filament coils are six turns slightly oblique and measuring 22.4 µm in length when is fully released ( Fig. 6 View FIGURE 6 L).
Taxonomic affinities. Over the Mediterranean Sea, after the transfer of Z. mugilis Sitjà-Bobadilla & Alvarez- Pellitero, 1993e to Ellipsomyxa under new name E. mugilis by Køie & Karlsbakk (2009), a few species of Zschokkella are outwardly similar to the present species that is considered as the first Zschokkella infecting a host from Serranidae and only the third species that has been reported from Tunisian waters after Z. soleae Yemmen, Marton, Bahri & Eszterbauer, 2013 and Z. trachini Azizi, Rangel, Castro, Santos & Bahri, 2016 ( Table 8). Z. soleae separates from the recent finding in having shorter spores (13.8 µm vs 16.75 µm), spherical and smaller polar capsules with lower number of polar filament coils (4–5 vs 6). In addition, the filament discharge pore has never inserted an elevation at any side of the shell valves of Z. soleae conversely to the present form. For Z. trachini , it differs from our species by having a different shape and a smaller size. Its shell valves are more pointed in the two sides and, is therefore a different species.
Among all the Zschokkella spp. identified from different localities in the world, the under studied Z. sp. is superficially similar to several species, Z. nova klokasheva, 1914 (cited from Lom & Dyková 1992), Z. acheilognathi Kudo, 1916 (cited from Kudo 1920), Z. tetrafluvi and Z. pleomorpha Lom & Dyková, 1995 , Z. mugilidae Kpatcha, Diebakate & Toguebaye, 1996 , Z. egyptica Ali, Abdel-Baki & Abdel-Ghaffar, 2007 , Z. helmii Abdel-Ghaffar, Ali, Al-Quraishy, Entzeroth, Abdel-Baki, Al-Farraj & Bashtar, 2008b , Z. nilei Abdel-Ghaffar , El- Toukhy, Al-Quraishy Al-Rasheid, Abdel-Baki, Hegazy & Bashtar, 2008c, Z. auratis Rocha, Casal, Rangel, Severino, Castro, Azevedo & Santos, 2013 and Z. cardinalis Heiniger & Adlard, 2014 ( Table 8).
Eventually, no range measurements, in both spores and polar capsules, overlap between Z. nova , Z. acheilognathi and the finding species. Considering surface ridges as a reliable feature for morphological comparison ( Lom & Dyková 1993, 2006; Bartošová & Fiala 2011), both Z. tetrafluvi and Z. pleomorpha are designed dissimilar to the current species. In addition, the spores and the polar capsules of Z. tetrafluvi are both smaller with a lower number of polar filament coils (4–5 vs 6), also this parasite has been reported from freshwater host. Likewise, the spores and spherical polar capsules of Z. pleomorpha are both smaller and possess a higher number of polar filament coils (7–9 vs 6). Besides, this parasite has been identified in different site of infection, kidney. All the available morphometric measurements between Z. mugilidae and the current species match, however the shape of both species are quite different. In fact, spores of Z. mugilidae are characterized by unequal rounded sides which one was larger than the other. Moreover, the filament discharge pore has never inserted an elevation at any side of the shell valves quite the opposite to our species. The spores of Z. egyptica are more globular and shorter in form. Besides, they possess a lower number of filament coils (4 vs 6), and a higher number, Gall blađđer; UR, Urinary blađđer; K, Kiđney; SL, Spore Length; ST, Spore Thickness; SW, Spore Wiđth; AL, Appenđages Length; PCL, Polar Capsule Length; PCW, Polar Capsule
Wiđth; S, sinuous; SS, slightly sinuous; SC, slight curve; C, curveđ; T, straight; ND, Not đetermineđ
Species Host Site Locality Spore AL Polar capsule Coils Suture
SL ST SW PCL PCW
Myxodavisia sp. (Present Serranus scriba GB Tunisia (Gulf of Tunis) 8.9 21.5 8.6 21 3.2 3 4/5 T/SC
stuđy) (7.6/11.1) (19.3/23.5) (7.6/10.5) (17.4/26.6) (2.8/3.6) (2.8/3.2)
spinosa Davis (1917) Paralichthys View in CoL UB USA (Atlantic ocean) 7 13 ND 70 4 4 ND T
albigutta
cella Jameson 1931 Zhao Porichthys UB USA (Atlantic ocean) (8/10)* ND (9/13) * (25/35) ND ND ND SC
al. (2008) notatus
brachiophora Lairđ Paralichthys UB USA (Atlantic ocean) (9/11) 9 ND (18/22) 3.5 3.5 ND SC
1953) albigutta
diplocrepis Lairđ (1953) Diplocrepis UB New Zealanđ 10.7 ND 13 12.7 3.7 3.4 (5/9) T/SC
puniceus (9/12) (12.1/14) (10/14.2) (3.4/3.8) (3.2/3.5)
ophidioni (Zaika, 1966) Ophidion rochei UB Black Sea (9.1/11.7) 13 ND (21/29) (3.9/5.2) (3.9/5.2) ND SC et al. 2008
anoplopoma Moser & Anoplopoma UB USA (Atlantic ocean) 12.75 15.2 ND 34.7 4.95 4.95 (5/7) SS
Noble (1975) fimbria K (12/14) (13/17) (30/38) (4/6) (4/6)
cynoglossi Cynoglossus sp. GB Inđia (Bay of Bengal) 14.24 12.6(11.4/ ND (20.8/36.4) 6.2 3.4 ND SS
Narasimhamurti et al. 1990) (12.6/16.8) 15.8) (5.8/6.8) (2.8/4.2)
et al. 2008
sauridae Saurida tumbil GB Inđia (Bay of Bengal) 9.6 18.2(14.8/ ND 3.52 1.8 1.6 (2/3) S
Narasimhamurti et al. 1990) (7.24/10.8) 22.6) (2.6/5.4) (1.6/3.2) (1.2/2.4)
et al. 2008
filiformis Pađma Dorothy Thryssa GB Inđia (Bay of Bengal) 12.3 19.73 ND 73.5 8.73 7.77 (2/3) T
al. 1998 setirostris (12/14) (16/20) (60/88) (8/10) (6/8)
narvi Aseeva (2002) Myoxocephalus View in CoL UB Japan Sea (10/11.5) ND (11.5/14.4) (20/27) (4/4.5) (4/4.5) 6 SC
brandtii
longifilus Aseeva (2003) Hippoglossoides View in CoL UB Japan Sea (10.5/13.5) (11.7/13) ND (20/23) (3/4.5) (2.5/3) ND T/SC
dubius
murtii Nanđi et al. 2004 Liza macrolepis View in CoL GB Inđia (Bay of Bengal) 7.31 ND 13.86 6.6 4.56 ND 4-5 S
(4.8/11.2)* (11.2/ (4.2/9.6) (3.2/4.8) 17.6)*
haldarae Sarkar (2010) Sardinella View in CoL GB Inđia (Bay of Bengal) 13.2 8.4 ND 32.5 4(3/4.5) 4(3/4.5) ND SS
longiceps View in CoL (12/15) (7.5/9) (28.5/34.5)
In đescriptions where measurements were not assigneđ to a particular đimension (i.e., length, thickness, or wiđth) we mađe the assignment baseđ to the đescription of Zhao et al. (2008).
of spore striations (9–11 vs 7–8). Z. helmii is the only histozoic cyst species from the gall bladder’s wall and occurs both smaller spores and polar capsules with less number of polar filament coils (5 vs 6) and great number of striations. The spores of Z. nilei have been described in different site of infection, Kidney, from a freshwater fish as well. In addition, they are smaller and contain a higher number of surface ridges than those of present species. Besides, it irregular suture line has never crossed the rounded ends of the spore. The species under study differentiates from Z. auratis in having both spores and polar capsules that are bigger in size with more filament turns and much less number of surface ridges. In addition, this latter species has more ovoid shape and its polar capsules are pyriform. Z. cardinalis resembles superficially to the recent finding species only in shape, however no measurement ranges overlap between both species, and is therefore a distinct species. So, as the first Zschokkella records infecting Serranidae host fishes and in light of the revealed differences with closely related congeneric species, the present myxosporean Zschokkella sp. is designated as a discriminate and different species, has not previously been recorded in Mediterranean Sea.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Zschokkella
| Laamiri, Sayef 2017 |
haldarae
| Sarkar 2010 |
murtii Nanđi et al. 2004
| Nandi et al. 2004 |
longifilus
| Aseeva 2003 |
narvi
| Aseeva 2002 |
spinosa
| Davis 1917 |